Abstract
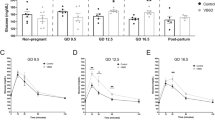
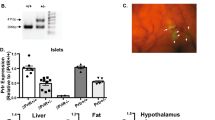
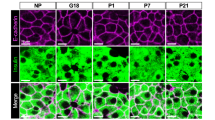
During pregnancy, the energy requirements of the fetus impose changes in maternal metabolism. Increasing insulin resistance in the mother maintains nutrient flow to the growing fetus, whereas prolactin and placental lactogen counterbalance this resistance and prevent maternal hyperglycemia by driving expansion of the maternal population of insulin-producing beta cells1,2,3. However, the exact mechanisms by which the lactogenic hormones drive beta cell expansion remain uncertain. Here we show that serotonin acts downstream of lactogen signaling to stimulate beta cell proliferation. Expression of serotonin synthetic enzyme tryptophan hydroxylase-1 (Tph1) and serotonin production rose sharply in beta cells during pregnancy or after treatment with lactogens in vitro. Inhibition of serotonin synthesis by dietary tryptophan restriction or Tph inhibition blocked beta cell expansion and induced glucose intolerance in pregnant mice without affecting insulin sensitivity. Expression of the Gαq-linked serotonin receptor 5-hydroxytryptamine receptor-2b (Htr2b) in maternal islets increased during pregnancy and normalized just before parturition, whereas expression of the Gαi-linked receptor Htr1d increased at the end of pregnancy and postpartum. Blocking Htr2b signaling in pregnant mice also blocked beta cell expansion and caused glucose intolerance. These studies reveal an integrated signaling pathway linking beta cell mass to anticipated insulin need during pregnancy. Modulators of this pathway, including medications and diet, may affect the risk of gestational diabetes4.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Van Assche, F.A., Aerts, L. & De Prins, F. A morphological study of the endocrine pancreas in human pregnancy. Br. J. Obstet. Gynaecol. 85, 818–820 (1978).
Parsons, J.A., Brelje, T.C. & Sorenson, R.L. Adaptation of islets of Langerhans to pregnancy: increased islet cell proliferation and insulin secretion correlates with the onset of placental lactogen secretion. Endocrinology 130, 1459–1466 (1992).
Huang, C., Snider, F. & Cross, J.C. Prolactin receptor is required for normal glucose homeostasis and modulation of beta-cell mass during pregnancy. Endocrinology 150, 1618–1626 (2009).
Buchanan, T.A. & Xiang, A.H. Gestational diabetes mellitus. J. Clin. Invest. 115, 485–491 (2005).
Freemark, M. et al. Targeted deletion of the PRL receptor: effects on islet development, insulin production, and glucose tolerance. Endocrinology 143, 1378–1385 (2002).
Vasavada, R.C. et al. Targeted expression of placental lactogen in the beta cells of transgenic mice results in beta cell proliferation, islet mass augmentation and hypoglycemia. J. Biol. Chem. 275, 15399–15406 (2000).
Sorenson, R.L. & Brelje, T.C. Adaptation of islets of Langerhans to pregnancy: beta-cell growth, enhanced insulin secretion and the role of lactogenic hormones. Horm. Metab. Res. 29, 301–307 (1997).
Brelje, T.C. et al. Effect of homologous placental lactogens, prolactins, and growth hormones on islet B-cell division and insulin secretion in rat, mouse, and human islets: implication for placental lactogen regulation of islet function during pregnancy. Endocrinology 132, 879–887 (1993).
Karnik, S.K. et al. Menin controls growth of pancreatic β-cells in pregnant mice and promotes gestational diabetes mellitus. Science 318, 806–809 (2007).
Ben-Jonathan, N., LaPensee, C.R. & LaPensee, E.W. What can we learn from rodents about prolactin in humans? Endocr. Rev. 29, 1–41 (2008).
Sakowski, S.A. et al. Differential tissue distribution of tryptophan hydroxylase isoforms 1 and 2 as revealed with monospecific antibodies. Brain Res. 1085, 11–18 (2006).
Hara, M. et al. Transgenic mice with green fluorescent protein–labeled pancreatic beta cells. Am. J. Physiol. Endocrinol. Metab. 284, E177–E183 (2003).
Fadda, F. Tryptophan-free diets: a physiological tool to study brain serotonin function. News Physiol. Sci. 15, 260–264 (2000).
Hoyer, D. et al. International union of pharmacology classification of receptors for 5-hydroxytryptamine (serotonin). Pharmacol. Rev. 46, 157–203 (1994).
Lesurtel, M. et al. Platelet-derived serotonin mediates liver regeneration. Science 312, 104–107 (2006).
Nebigil, C.G. et al. Serotonin 2B receptor is required for heart development. Proc. Natl. Acad. Sci. USA 97, 9508–9513 (2000).
Wouters, M.M. et al. Exogenous serotonin regulates proliferation of interstitial cells of Cajal in mouse jejunum through 5–HT2B receptors. Gastroenterology 133, 897–906 (2007).
Rothman, R.B. et al. Evidence for possible involvement of 5-HT2B receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications. Circulation 102, 2836–2841 (2000).
Fitzgerald, L.W. et al. Possible role of valvular serotonin 5-HT2B receptors in the cardiopathy associated with fenfluramine. Mol. Pharmacol. 57, 75–81 (2000).
Collet, C. et al. The serotonin 5-HT2B receptor controls bone mass via osteoblast recruitment and proliferation. FASEB J. 22, 418–427 (2008).
De Lucchini, S., Ori, M., Cremisi, F., Nardini, M. & Nardi, I. 5-HT2B–mediated serotonin signaling is required for eye morphogenesis in Xenopus. Mol. Cell. Neurosci. 29, 299–312 (2005).
Gannon, M., Shiota, C., Postic, C., Wright, C.V. & Magnuson, M. Analysis of the Cre-mediated recombination driven by rat insulin promoter in embryonic and adult mouse pancreas. Genesis 26, 139–142 (2000).
Matsuda, M. et al. Serotonin regulates mammary gland development via an autocrine-paracrine loop. Dev. Cell 6, 193–203 (2004).
Møldrup, A., Petersen, E.D. & Nielsen, J.H. Effects of sex and pregnancy hormones on growth hormone and prolactin receptor gene expression in insulin-producing cells. Endocrinology 133, 1165–1172 (1993).
Leahy, J.L., Bonner-Weir, S. & Weir, G.C. Abnormal glucose regulation of insulin secretion in models of reduced B-cell mass. Diabetes 33, 667–673 (1984).
Goodner, C.J., Koerker, D.J., Weigle, D.S. & McCulloch, D.K. Decreased insulin- and glucagon-pulse amplitude accompanying beta-cell deficiency induced by streptozocin in baboons. Diabetes 38, 925–931 (1989).
Butler, A.E. et al. Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 52, 102–110 (2003).
Mezuk, B., Eaton, W.W., Albrecht, S. & Golden, S.H. Depression and type 2 diabetes over the lifespan: a meta-analysis. Diabetes Care 31, 2383–2390 (2008).
Kozhimannil, K.B., Pereira, M.A. & Harlow, B.L. Association between diabetes and perinatal depression among low-income mothers. J. Am. Med. Assoc. 301, 842–847 (2009).
Gianfrancesco, F.D., Grogg, A.L., Mahmoud, R.A., Wang, R.H. & Nasrallah, H.A. Differential effects of risperidone, olanzapine, clozapine, and conventional antipsychotics on type 2 diabetes: findings from a large health plan database. J. Clin. Psychiatry 63, 920–930 (2002).
Acknowledgements
We thank G. Grodsky, W. Rutter and members of the German laboratory for helpful discussions; F. Schaufle and the UCSF Diabetes and Endocrinology Research Center Microscopy Core Laboratory; G. Szot and the UCSF Diabetes and Endocrinology Research Center Islet Core; Y. Zhang, Z. Li and S. Zhao for technical assistance with mouse husbandry and genotyping; N. Daimaru, E. Magoshi and K. Nakamura for technical assistance; K. Takahashi, W. Inaba, M. Tsujii and S. Nakayama for immunohistochemical studies; Y. Katayama (Hirosaki University) and P. Ursell (UCSF) for providing human pancreatic samples; and D. Kuhn (Wayne State University), L. Maroteaux (Institut National de la Santé et de la Recherche Médicale) and M. Hara (University of Chicago) for generously providing the Tph1-specific antisera, Htr2b-targeted mice and MIP-GFP mice, respectively. This work was supported by grants from the Larry L. Hillblom Foundation, the Juvenile Diabetes Research Foundation, the American Diabetes Association, the US National Institutes of Health–National Institute of Diabetes and Digestive and Kidney Diseases and the Ministry of Education, Sports and Culture of Japan.
Author information
Authors and Affiliations
Contributions
H.K., H.W. and M.S.G. designed the research; H.K., Y.T., F.C.L., E.C., T.U., H.M., Y.F., T.M., Y.K., G.H., M.v.d.H., K.Y., N.K. and J.W. performed the research; H.K., F.C.L., R.K., S.Y., H.W., L.H.T. and M.S.G. analyzed the data; and H.K., F.C.L., H.W. and M.S.G. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1 and 2, Supplementary Figures 1–6 and Supplementary Methods (PDF 625 kb)
Rights and permissions
About this article
Cite this article
Kim, H., Toyofuku, Y., Lynn, F. et al. Serotonin regulates pancreatic beta cell mass during pregnancy. Nat Med 16, 804–808 (2010). https://doi.org/10.1038/nm.2173
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2173
This article is cited by
-
Discovery of a peripheral 5HT2A antagonist as a clinical candidate for metabolic dysfunction-associated steatohepatitis
Nature Communications (2024)
-
Antidepressants and type 2 diabetes: highways to knowns and unknowns
Diabetology & Metabolic Syndrome (2023)
-
Variant rs17619600 in the gene encoding serotonin receptor 2B (HTR2B) increases the risk of gestational diabetes mellitus: a case–control study
European Journal of Medical Research (2023)
-
Hyperaminoacidemia induces pancreatic α cell proliferation via synergism between the mTORC1 and CaSR-Gq signaling pathways
Nature Communications (2023)
-
Multiparity increases the risk of diabetes by impairing the proliferative capacity of pancreatic β cells
Experimental & Molecular Medicine (2023)