Abstract
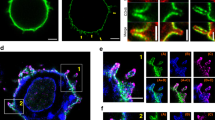
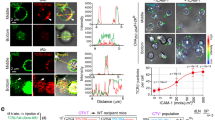
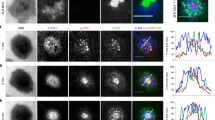
T cell receptor (TCR) activation and signaling precede immunological synapse formation and are sustained for hours after initiation. However, the precise physical sites of the initial and sustained TCR signaling are not definitively known. We report here that T cell activation was initiated and sustained in TCR-containing microclusters generated at the initial contact sites and the periphery of the mature immunological synapse. Microclusters containing TCRs, the tyrosine kinase Zap70 and the adaptor molecule SLP-76 were continuously generated at the periphery. TCR microclusters migrated toward the central supramolecular cluster, whereas Zap70 and SLP-76 dissociated from these microclusters before the microclusters coalesced with the TCR-rich central supramolecular cluster. Tyrosine phosphorylation and calcium influx were induced as microclusters formed at the initial contact sites. Inhibition of signaling prevented recruitment of Zap70 into the microclusters. These results indicated that TCR-rich microclusters initiate and sustain TCR signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Monks, C.R., Freiberg, B.A., Kupfer, H., Sciaky, N. & Kupfer, A. Three-dimensional segregation of supramolecular activation clusters in T cells. Nature 395, 82–86 (1998).
Grakoui, A. et al. The immunological synapse: a molecular machine controlling T cell activation. Science 285, 221–227 (1999).
Huppa, J.B. & Davis, M.M. T-cell-antigen recognition and the immunological synapse. Nat. Rev. Immunol. 3, 973–983 (2003).
Dustin, M.L. & Cooper, J.A. The immunological synapse and the actin cytoskeleton: molecular hardware for T cell signaling. Nat. Immunol. 1, 23–29 (2000).
Vicente-Manzanares, M. & Sanchez-Madrid, F. Role of the cytoskeleton during leukocyte responses. Nat. Rev. Immunol. 4, 110–122 (2004).
Freiberg, B.A. et al. Staging and resetting T cell activation in SMACs. Nat. Immunol. 3, 911–917 (2002).
Allenspach, E.J. et al. ERM-dependent movement of CD43 defines a novel protein complex distal to the immunological synapse. Immunity 15, 739–750 (2001).
Blanchard, N., Di Bartolo, V. & Hivroz, C. In the immune synapse, ZAP-70 controls T cell polarization and recruitment of signaling proteins but not formation of the synaptic pattern. Immunity 17, 389–399 (2002).
Delon, J., Kaibuchi, K. & Germain, R.N. Exclusion of CD43 from the immunological synapse is mediated by phosphorylation-regulated relocation of the cytoskeletal adaptor moesin. Immunity 15, 691–701 (2001).
Lee, K.H. et al. T cell receptor signaling precedes immunological synapse formation. Science 295, 1539–1542 (2002).
Irvine, D.J., Purbhoo, M.A., Krogsgaard, M. & Davis, M.M. Direct observation of ligand recognition by T cells. Nature 419, 845–849 (2002).
Harriague, J. & Bismuth, G. Imaging antigen-induced PI3K activation in T cells. Nat. Immunol. 3, 1090–1096 (2002).
Costello, P.S., Gallagher, M. & Cantrell, D.A. Sustained and dynamic inositol lipid metabolism inside and outside the immunological synapse. Nat. Immunol. 3, 1082–1089 (2002).
Lee, K.H. et al. The immunological synapse balances T cell receptor signaling and degradation. Science 302, 1218–1222 (2003).
Purtic, B., Pitcher, L.A., van Oers, N.S. & Wulfing, C. T cell receptor (TCR) clustering in the immunological synapse integrates TCR and costimulatory signaling in selected T cells. Proc. Natl. Acad. Sci. USA 102, 2904–2909 (2005).
Bunnell, S.C. et al. T cell receptor ligation induces the formation of dynamically regulated signaling assemblies. J. Cell Biol. 158, 1263–1275 (2002).
Huppa, J.B., Gleimer, M., Sumen, C. & Davis, M.M. Continuous T cell receptor signaling required for synapse maintenance and full effector potential. Nat. Immunol. 4, 749–755 (2003).
Dustin, M.L. & Shaw, A.S. Costimulation: building an immunological synapse. Science 283, 649–650 (1999).
Stoll, S., Delon, J., Brotz, T.M. & Germain, R.N. Dynamic imaging of T cell-dendritic cell interactions in lymph nodes. Science 296, 1873–1876 (2002).
Miller, M.J., Wei, S.H., Parker, I. & Cahalan, M.D. Two-photon imaging of lymphocyte motility and antigen response in intact lymph node. Science 296, 1869–1873 (2002).
Hailman, E., Burack, W.R., Shaw, A.S., Dustin, M.L. & Allen, P.M. Immature CD4+CD8+ thymocytes form a multifocal immunological synapse with sustained tyrosine phosphorylation. Immunity 16, 839–848 (2002).
Heissmeyer, V. et al. Calcineurin imposes T cell unresponsiveness through targeted proteolysis of signaling proteins. Nat. Immunol. 5, 255–265 (2004).
Iezzi, G., Karjalainen, K. & Lanzavecchia, A. The duration of antigenic stimulation determines the fate of naive and effector T cells. Immunity 8, 89–95 (1998).
van Stipdonk, M.J. et al. Dynamic programming of CD8+ T lymphocyte responses. Nat. Immunol. 4, 361–365 (2003).
Faroudi, M., Zaru, R., Paulet, P., Muller, S. & Valitutti, S. Cutting edge: T lymphocyte activation by repeated immunological synapse formation and intermittent signaling. J. Immunol. 171, 1128–1132 (2003).
Depoil, D. et al. Immunological synapses are versatile structures enabling selective T cell polarization. Immunity 22, 185–194 (2005).
Bunnell, S.C., Kapoor, V., Trible, R.P., Zhang, W. & Samelson, L.E. Dynamic actin polymerization drives T cell receptor-induced spreading: a role for the signal transduction adaptor LAT. Immunity 14, 315–329 (2001).
Barda-Saad, M. et al. Dynamic molecular interactions linking the T cell antigen receptor to the actin cytoskeleton. Nat. Immunol. 6, 80–89 (2004).
Krummel, M.F. & Davis, M.M. Dynamics of the immunological synapse: finding, establishing and solidifying a connection. Curr. Opin. Immunol. 14, 66–74 (2002).
Krummel, M.F., Sjaastad, M.D., Wulfing, C. & Davis, M.M. Differential clustering of CD4 and CD3ζ during T cell recognition. Science 289, 1349–1352 (2000).
Brossard, C. et al. Multifocal structure of the T cell - dendritic cell synapse. Eur. J. Immunol. 35, 1741–1753 (2005).
Gelkop, S. & Isakov, N. T cell activation stimulates the association of enzymatically active tyrosine-phosphorylated ZAP-70 with the Crk adapter proteins. J. Biol. Chem. 274, 21519–21527 (1999).
Sasahara, Y. et al. Mechanism of recruitment of WASP to the immunological synapse and of its activation following TCR ligation. Mol. Cell 10, 1269–1281 (2002).
Zeng, R. et al. SLP-76 coordinates Nck-dependent Wiskott-Aldrich syndrome protein recruitment with Vav-1/Cdc42-dependent Wiskott-Aldrich syndrome protein activation at the T cell-APC contact site. J. Immunol. 171, 1360–1368 (2003).
Griffiths, E.K. & Penninger, J.M. Communication between the TCR and integrins: role of the molecular adapter ADAP/Fyb/Slap. Curr. Opin. Immunol. 14, 317–322 (2002).
Krause, M. et al. Fyn-binding protein (Fyb)/SLP-76-associated protein (SLAP), Ena/vasodilator-stimulated phosphoprotein (VASP) proteins and the Arp2/3 complex link T cell receptor (TCR) signaling to the actin cytoskeleton. J. Cell Biol. 149, 181–194 (2000).
Dustin, M.L. Stop and go traffic to tune T cell responses. Immunity 21, 305–314 (2004).
Maldonado, R.A., Irvine, D.J., Schreiber, R. & Glimcher, L.H. A role for the immunological synapse in lineage commitment of CD4 lymphocytes. Nature 431, 527–532 (2004).
Valitutti, S., Muller, S., Cella, M., Padovan, E. & Lanzavecchia, A. Serial triggering of many T-cell receptors by a few peptide-MHC complexes. Nature 375, 148–151 (1995).
Davis, S.J. & van der Merwe, P.A. The immunological synapse: required for T cell receptor signalling or directing T cell effector function? Curr. Biol. 11, R289–R291 (2001).
Delon, J. & Germain, R.N. Information transfer at the immunological synapse. Curr. Biol. 10, R923–R933 (2000).
Wulfing, C. et al. Costimulation and endogenous MHC ligands contribute to T cell recognition. Nat. Immunol. 3, 42–47 (2002).
Campbell, R.E. et al. A monomeric red fluorescent protein. Proc. Natl. Acad. Sci. USA 99, 7877–7882 (2002).
Axelrod, D. Cell-substrate contacts illuminated by total internal reflection fluorescence. J. Cell Biol. 89, 141–145 (1981).
Tokunaga, M., Kitamura, K., Saito, K., Iwane, A.H. & Yanagida, T. Single molecule imaging of fluorophores and enzymatic reactions achieved by objective-type total internal reflection fluorescence microscopy. Biochem. Biophys. Res. Commun. 235, 47–53 (1997).
Acknowledgements
We thank R. Varma, G. Campi and T. Starr for instructions on the preparation of planar bilayers and for sharing data before publication, and H. Yamaguchi for secretarial assistance. Supported by the Ministry of Education, Culture, Sports, Science and Technology of Japan (T.Y., M.T. and T.S.), New Energy Development Organization (M.T.), and the National Institutes of Health (AI043542 and AI044931 to M.L.D.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Statistical analysis of the movement of CD3ζ-MCs at the initiation of T cell-bilayer contact. (PDF 952 kb)
Supplementary Fig. 2
The area and relative fluorescence intensity of CD3ζ-MCs. (PDF 1076 kb)
Supplementary Fig. 3
Generation of CD3ζ-, ZAP-70- and SLP-76-MCs under suboptimal concentrations of the agonistic peptide. (PDF 4662 kb)
Supplementary Fig. 4
CD3ζ-, ZAP-70- and SLP-76-MCs are generated at the initiation of the T cell-APC interaction. (PDF 1926 kb)
Supplementary Fig. 5
Microclusters are formed in the initial interface of the normal T cell – APC interaction. (PDF 708 kb)
Supplementary Fig. 6
ZAP-70 is continuously phosphorylated throughout IS formation. (PDF 1093 kb)
Supplementary Fig. 7
Three-dimensional views of the CD3ζ-expressing cells on bilayers, in the presence or absence of PP2. (PDF 1336 kb)
Supplementary Fig. 8
Quantification of CD3ζ-EGFP proteins within individual MC before and after IS formation. (PDF 841 kb)
Supplementary Fig. 9
Statistical analysis of the movement of CD3ζ-, ZAP-70- and SLP-76-MCs after mature IS formation. (PDF 700 kb)
Supplementary Table 1
AND-Tg T cells expressing ZAP-70-EGFP or SLP-76-EGFP, or both ZAP-70-mRFP and SLP-76-EGFP were plated on a PCC88-104-prepulsed planar bilayer containing I-Ek and ICAM-1. (PDF 276 kb)
Supplementary Video 1
AND-Tg T cells expressing CD3ζ-EGFP were plated on PCC88-104-prepulsed planar bilayers containing non-labeled I-Ek and ICAM-1. Images were collected at video rate (30 frames/sec) by TIRFM, 8-frame averaged, and rebuilt at 1 frame/sec, from time zero to 300 sec. (MOV 2932 kb)
Supplementary Video 2
AND-Tg T cells expressing ZAP-70-EGFP were plated on PCC88-104-prepulsed planar bilayers containing non-labeled I-Ek and ICAM-1. Images were collected at video rate (30 frames/sec) by TIRFM, 8-frame averaged, and rebuilt at 1 frame/sec, from time zero to 300 sec. (MOV 2932 kb)
Supplementary Video 3
AND-Tg T cells expressing SLP-76-EGFP were plated on PCC88-104-prepulsed planar bilayers containing non-labeled I-Ek and ICAM-1. Images were collected at video rate (30 frames/sec) by TIRFM, 8-frame averaged, and rebuilt at 1 frame/sec, from time zero to 300 sec. (MOV 2903 kb)
Supplementary Video 4
AND-Tg T cells expressing CD3ζ-EGFP were plated on PCC88-104-prepulsed planar bilayers containing non-labeled I-Ek and ICAM-1. Images were collected at video rate (30 frames/sec) from 15 min after the cell-bilayer contacts. They were 8-frame averaged and rebuilt at 1 frame/sec for the duration of 90 sec. (MOV 1956 kb)
Supplementary Video 5
AND-Tg T cells expressing ZAP-70-EGFP were plated on PCC88-104-prepulsed planar bilayers containing non-labeled I-Ek and ICAM-1. Images were collected at video rate (30 frames/sec) from 15 min after the cell-bilayer contacts. They were 8-frame averaged and rebuilt at 1 frame/sec for the duration of 90 sec. (MOV 1956 kb)
Supplementary Video 6
AND-Tg T cells expressing SLP-76-EGFP were plated on PCC88-104-prepulsed planar bilayers containing non-labeled I-Ek and ICAM-1. Images were collected at video rate (30 frames/sec) from 15 min after the cell-bilayer contacts. They were 8-frame averaged and rebuilt at 1 frame/sec for the duration of 90 sec. (MOV 1956 kb)
Rights and permissions
About this article
Cite this article
Yokosuka, T., Sakata-Sogawa, K., Kobayashi, W. et al. Newly generated T cell receptor microclusters initiate and sustain T cell activation by recruitment of Zap70 and SLP-76. Nat Immunol 6, 1253–1262 (2005). https://doi.org/10.1038/ni1272
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1272
This article is cited by
-
Membrane-anchored DNA nanojunctions enable closer antigen-presenting cell–T-cell contact in elevated T-cell receptor triggering
Nature Nanotechnology (2023)
-
Evaluation of therapeutic PD-1 antibodies by an advanced single-molecule imaging system detecting human PD-1 microclusters
Nature Communications (2023)
-
Discrete LAT condensates encode antigen information from single pMHC:TCR binding events
Nature Communications (2022)
-
Kinetic proofreading through the multi-step activation of the ZAP70 kinase underlies early T cell ligand discrimination
Nature Immunology (2022)
-
The interplay between membrane topology and mechanical forces in regulating T cell receptor activity
Communications Biology (2022)