Abstract
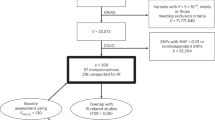
UK Biobank is among the world's largest repositories for phenotypic and genotypic information in individuals of European ancestry1. We performed a genome-wide association study in UK Biobank testing ∼9 million DNA sequence variants for association with coronary artery disease (4,831 cases and 115,455 controls) and carried out meta-analysis with previously published results. We identified 15 new loci, bringing the total number of loci associated with coronary artery disease to 95 at the time of analysis. Phenome-wide association scanning showed that CCDC92 likely affects coronary artery disease through insulin resistance pathways, whereas experimental analysis suggests that ARHGEF26 influences the transendothelial migration of leukocytes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Collins, R. What makes UK Biobank special? Lancet 379, 1173–1174 (2012).
GBD 2015 Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388, 1459–1544 (2016).
Schunkert, H. et al. Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat. Genet. 43, 333–338 (2011).
Deloukas, P. et al. Large-scale association analysis identifies new risk loci for coronary artery disease. Nat. Genet. 45, 25–33 (2013).
CARDIoGRAMplusC4D Consortium. A comprehensive 1000 Genomes–based genome-wide association meta-analysis of coronary artery disease. Nat. Genet. 47, 1121–1130 (2015).
Myocardial Infarction Genetics and CARDIoGRAM Exome Consortia Investigators. Coding variation in ANGPTL4, LPL, and SVEP1 and the risk of coronary disease. N. Engl. J. Med. 374, 1134–1144 (2016).
Nioi, P. et al. Variant ASGR1 associated with a reduced risk of coronary artery disease. N. Engl. J. Med. 374, 2131–2141 (2016).
Webb, T.R. et al. Systematic evaluation of pleiotropy identifies 6 further loci associated with coronary artery disease. J. Am. Coll. Cardiol. 69, 823–836 (2017).
Howson, J.M.M. et al. Fifteen new risk loci for coronary artery disease highlight arterial-wall-specific mechanisms. Nat. Genet. http://dx.doi.org/10.1038/ng.3874 (2017).
Musunuru, K. et al. From noncoding variant to phenotype via SORT1 at the 1p13 cholesterol locus. Nature 466, 714–719 (2010).
Myocardial Infarction Genetics Consortium. Genome-wide association of early-onset myocardial infarction with single nucleotide polymorphisms and copy number variants. Nat. Genet. 41, 334–341 (2009).
Trégouët, D.A. et al. Genome-wide haplotype association study identifies the SLC22A3–LPAL2–LPA gene cluster as a risk locus for coronary artery disease. Nat. Genet. 41, 283–285 (2009).
Samani, N.J. et al. Genomewide association analysis of coronary artery disease. N. Engl. J. Med. 357, 443–453 (2007).
Denny, J.C. et al. Systematic comparison of phenome-wide association study of electronic medical record data and genome-wide association study data. Nat. Biotechnol. 31, 1102–1110 (2013).
Aguet, F. et al. Local genetic effects on gene expression across 44 human tissues. Prepinrt at bioRxiv http://dx.doi.org/10.1101/074450 (2016).
Global Lipids Genetics Consortium. Discovery and refinement of loci associated with lipid levels. Nat. Genet. 45, 1274–1283 (2013).
Manning, A.K. et al. A genome-wide approach accounting for body mass index identifies genetic variants influencing fasting glycemic traits and insulin resistance. Nat. Genet. 44, 659–669 (2012).
Prokopenko, I. et al. A central role for GRB10 in regulation of islet function in man. PLoS Genet. 10, e1004235 (2014).
Wood, A.R. et al. Defining the role of common variation in the genomic and biological architecture of adult human height. Nat. Genet. 46, 1173–1186 (2014).
Berndt, S.I. et al. Genome-wide meta-analysis identifies 11 new loci for anthropometric traits and provides insights into genetic architecture. Nat. Genet. 45, 501–512 (2013).
Pattaro, C. et al. Genetic associations at 53 loci highlight cell types and biological pathways relevant for kidney function. Nat. Commun. 7, 10023 (2016).
Liu, J.Z. et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat. Genet. 47, 979–986 (2015).
Dastani, Z. et al. Novel loci for adiponectin levels and their influence on type 2 diabetes and metabolic traits: a multi-ethnic meta-analysis of 45,891 individuals. PLoS Genet. 8, e1002607 (2012).
Morris, A.P. et al. Large-scale association analysis provides insights into the genetic architecture and pathophysiology of type 2 diabetes. Nat. Genet. 44, 981–990 (2012).
Maass, P.G. et al. PDE3A mutations cause autosomal dominant hypertension with brachydactyly. Nat. Genet. 47, 647–653 (2015).
Kato, N. et al. Trans-ancestry genome-wide association study identifies 12 genetic loci influencing blood pressure and implicates a role for DNA methylation. Nat. Genet. 47, 1282–1293 (2015).
Chung, C.M. et al. A genome-wide association study reveals a quantitative trait locus of adiponectin on CDH13 that predicts cardiometabolic outcomes. Diabetes 60, 2417–2423 (2011).
Shungin, D. et al. New genetic loci link adipose and insulin biology to body fat distribution. Nature 518, 187–196 (2015).
Lotta, L.A. et al. Integrative genomic analysis implicates limited peripheral adipose storage capacity in the pathogenesis of human insulin resistance. Nat. Genet. 49, 17–26 (2017).
Sakai, T., Larsen, M. & Yamada, K.M. Fibronectin requirement in branching morphogenesis. Nature 423, 876–881 (2003).
Erler, J.T. et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature 440, 1222–1226 (2006).
Lee, V.S. et al. Loss of function mutation in LOX causes thoracic aortic aneurysm and dissection in humans. Proc. Natl. Acad. Sci. USA 113, 8759–8764 (2016).
Guo, D.C. et al. LOX mutations predispose to thoracic aortic aneurysms and dissections. Circ. Res. 118, 928–934 (2016).
Hood, J.D. & Cheresh, D.A. Role of integrins in cell invasion and migration. Nat. Rev. Cancer 2, 91–100 (2002).
Samson, T. et al. The guanine-nucleotide exchange factor SGEF plays a crucial role in the formation of atherosclerosis. PLoS One 8, e55202 (2013).
van Rijssel, J. et al. The Rho-guanine nucleotide exchange factor Trio controls leukocyte transendothelial migration by promoting docking structure formation. Mol. Biol. Cell 23, 2831–2844 (2012).
van Buul, J.D. et al. RhoG regulates endothelial apical cup assembly downstream from ICAM1 engagement and is involved in leukocyte trans-endothelial migration. J. Cell Biol. 178, 1279–1293 (2007).
Zahedi, F. et al. Dicer generates a regulatory microRNA network in smooth muscle cells that limits neointima formation during vascular repair. Cell. Mol. Life Sci. 74, 359–372 (2017).
Ellerbroek, S.M. et al. SGEF, a RhoG guanine nucleotide exchange factor that stimulates macropinocytosis. Mol. Biol. Cell 15, 3309–3319 (2004).
Gerhardt, T. & Ley, K. Monocyte trafficking across the vessel wall. Cardiovasc. Res. 107, 321–330 (2015).
Khera, A.V. & Kathiresan, S. Genetics of coronary artery disease: discovery, biology and clinical translation. Nat. Rev. Genet. 18, 331–344 (2017).
Wain, L.V. et al. Novel insights into the genetics of smoking behaviour, lung function, and chronic obstructive pulmonary disease (UK BiLEVE): a genetic association study in UK Biobank. Lancet Respir. Med. 3, 769–781 (2015).
Loh, P.R. et al. Efficient Bayesian mixed-model analysis increases association power in large cohorts. Nat. Genet. 47, 284–290 (2015).
Willer, C.J., Li, Y. & Abecasis, G.R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010).
Castel, S.E., Levy-Moonshine, A., Mohammadi, P., Banks, E. & Lappalainen, T. Tools and best practices for data processing in allelic expression analysis. Genome Biol. 16, 195 (2015).
Acknowledgements
This research has been conducted using the UK Biobank resource, application 7089. The PheWAS carried out by Genomics plc was also conducted using the UK Biobank resource under a separate agreement. The authors thank K. Burridge and E. Wittchen for technical advice on the leukocyte transendothelial migration assay. This analysis was supported by the National Heart, Lung, and Blood Institute of the US National Institutes of Health under award number T32 HL007734 (D.K.), the John S. LaDue Memorial Fellowship in Cardiology at Harvard Medical School (P.N.), the KL2/Catalyst Medical Research Investigator Training award from Harvard Catalyst funded by the US National Institutes of Health (TR001100) (A.V.K.), the Ofer and Shelly Nemirovsky Research Scholar award from Massachusetts General Hospital (MGH), the Donovan Family Foundation, and US National Institutes of Health grant R01 HL127564 (S.K.). The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Consortia
Contributions
Concept and design: D.K., Q.M.Z., M.E.W., A.V.K., P.N., S.K. Acquisition, analysis, or interpretation of data: D.K., Q.M.Z., C.A.E., M.C., S.H., B.J.M., A.L., M.E.W., C.C.A.S., F.A., A.V.S., K.G.A., A.V.K., V.K.K., P.N., S.K. Drafting of the manuscript: D.K., Q.M.Z., C.A.E., M.E.W., A.V.K., P.N., S.K. Critical revision of the manuscript for important intellectual content: D.K., Q.M.Z., C.A.E., M.C., S.H., B.J.M., A.L., M.E.W, C.C.A.S., F.A., A.V.S., K.G.A., A.V.K., V.K.K., P.N., S.K. Administrative, technical, or material support: D.K., S.K.
Ethics declarations
Competing interests
S.K. reports grant support from Regeneron and Bayer, grant support and personal fees from Aegerion, personal fees from Regeneron Genetics Center, Merck, Celera, Novartis, Bristol-Myers Squibb, Sanofi, AstraZeneca, Alnylam, Eli Lilly, and Leerink Partners, personal fees and other support from Catabasis, and other support from San Therapeutics outside the submitted work. He is also the chair of the scientific advisory board at Genomics plc.
Additional information
A list of members and affiliations appears in the Supplementary Note.
Integrated supplementary information
Supplementary Figure 1 Quantile–quantile plot for the stage 1 CAD GWAS.
The expected association P values versus the observed distribution of P values for CAD association are displayed. Significant systemic inflation is not observed (λGC = 1.05).
Supplementary Figure 2 Manhattan plot for the stage 1 CAD GWAS.
Plot of –log10 (P) for association of imputed variants by chromosomal position for all autosomal polymorphisms analyzed in the UK Biobank, stage 1 CAD GWAS. The genes nearest to the top associated variants are displayed. CAD, coronary artery disease; GWAS, genome-wide association study.
Supplementary Figure 3 Risk allele effect estimates in the literature and in UK Biobank for a set of previously reported CAD variants.
Plot of the effect estimates for 56 CAD-associated DNA sequence variants as reported in the 1000G-imputed CARDIoGRAMplusC4D analysis1 and in our UK Biobank GWAS analysis. β = 0.92, 95% CI = 0.77–1.06; P = 1.8 × 10−17.
Supplementary Figure 4 Stage 2 regional association plots for novel CAD loci.
These regional association plots demonstrate the strength of association, by –log10 (P), for four of the novel CAD loci in stage 2, within a window of ±400 kb.
Supplementary Figure 5 Stage 3 regional association plots for novel CAD loci.
These regional association plots demonstrate the strength of association, by –log10 (P), for six novel CAD loci in stage 3, within a window of ±400 kb.
Supplementary Figure 6 Stage 3 regional association plots for novel CAD loci.
These regional association plots demonstrate the strength of association, by –log10 (P), for five novel CAD loci in stage 3, within a window of ±400 kb.
Supplementary Figure 7 Analyses of gene expression associated with the rs12493885 alleles.
(a) eQTL analysis. In 133 coronary artery samples obtained by GTEx, eQTL analysis does not demonstrate evidence of altered expression associated with the ARHGEF26 p.Val29Leu (rs12493885) variant. β = 0.22, P = 0.16. No other variants in the region demonstrate significant eQTL effects at a threshold of FDR < 0.05 in coronary artery. The β estimate is aligned to the C (Leu29) allele. (b) Allele-specific expression analysis. In 20 coronary artery samples obtained from the GTEx Consortium heterozygous for the ARHGEF26 p.Val29Leu (rs12493885) variant, no individual demonstrated significant evidence of allele imbalance in coronary artery at a threshold of FDR < 0.05 (n.s., two-sided binomial test non-significant). REF refers to the reference (G, Val29) allele and ALT to the alternative (C, Leu29) allele.
Supplementary Figure 8 ARHGEF26 promoter activity luciferase assay.
The −2,516 to +2 region 5′ of the ARHGEF26 gene was cloned for the haplotypes with the G (reference) and C (alternative) alleles at rs12493885. The reference and alternative haplotypes were coupled with a firefly luciferase reporter gene and cotransfected with a Renilla luciferase co-reporter in HEK293 cells, HAECs, and HUVECs. Promoterless firefly luciferase reporter was included as negative control. Firefly luciferase activity relative to Renilla luciferase activity was measured 48 h after transfection and is expressed as fold changes over promoterless vectors (HEK293 cells, n = 4; HAECs, n = 6; HUVECs, n = 6; mean ± s.d.; separate one-way ANOVA with Tukey’s multiple-comparisons tests and multiplicity-adjusted P values for each cell type; F = 23.88, DF = 2 for HEK293 cells; F = 0.8038, DF = 2 in HAECs; F = 0.02397, DF = 2 in HUVECs).
Supplementary Figure 9 Western blots of transfected vascular cells.
HAECs or HCASMCs were transfected with non-targeting siRNA plus empty vector (Control), siRNA against ARHGEF26 3′ UTR and empty vector (siRNA + empty vector), siRNA and a wild-type FLAG-ARHGEF26 vector (siRNA + WT), or siRNA and a mutant vector (siRNA + Leu29). Transfected HAECs or HCASMCs were harvested 72 h after transfection. Normalized cell lysates (20 μg/lane) were resolved by SDS–PAGE and probed for ARHGEF26, FLAG, and actin by respective antibodies and imaged by enhanced chemiluminescence.
Supplementary Figure 10 Effects of p.Val29Leu mutant on ARHGEF26 protein nucleotide-exchange activity.
Evaluation of wild-type and Leu29 ARHGEF26 nucleotide-exchange activity. Full-length, N-terminally His-SUMO-tagged wild-type and Leu29 ARHGEF26 and full-length RhoG were expressed in E. coli. The nucleotide-exchange assay was prepared with equal amounts of recombinant ARHGEF26 WT (blue) and ARHGEF26 Leu29 (red) in reaction buffer containing MANT-GTP. Just prior to reading, recombinant RhoG protein, preloaded with GDP, was added to the reaction buffer at a final concentration of 0.4 μM. MANT-GTP fluorescence was monitored for 60 min on a SpectraMax M2 at 37 ºC using an excitation wavelength of 280 nm and an emission wavelength of 440 nm with a cutoff at 435 nm. No significant difference in nucleotide-exchange activity was observed between ARHGEF26 WT (blue) and ARHGEF26 Leu29 (red) in the presence of RhoG.
Supplementary Figure 11 Evaluation of ARHGEF26 protein stability in cells.
Wild-type (WT) or Leu29 Flag-ARHGEF26 were overexpressed in HEK293 cells for 48 h followed by treatment with 50 μg/mL and 100 μg/mL cycloheximide. Cells were harvested at indicated time points after treatment, and normalized lysates (20 μg/lane) were probed for FLAG by western blot. For each cycloheximide dose, two blot sections (WT and Leu29) from the same membrane simultaneously imaged are shown in juxtaposition for contrast.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11, Supplementary Tables 1, 2, 7 and 8, and Supplementary Note. (PDF 2413 kb)
Supplementary Table 3
Descriptions of novel loci and supportive evidence suggesting causal genes. (XLSX 32 kb)
Supplementary Table 4
Genome-wide significant variant–gene cis-eQTL pairs for 15 novel CAD risk variants queried in GTEx Consortium Project data, aligned to the CAD risk allele. (XLSX 45 kb)
Supplementary Table 5
Phenome-wide association results for the 15 novel CAD variants. (XLSX 74 kb)
Supplementary Table 6
Sources of cases and controls in the CARDIoGRAM Exome Consortia Study for stage 2. (XLSX 39 kb)
Rights and permissions
About this article
Cite this article
Klarin, D., Zhu, Q., Emdin, C. et al. Genetic analysis in UK Biobank links insulin resistance and transendothelial migration pathways to coronary artery disease. Nat Genet 49, 1392–1397 (2017). https://doi.org/10.1038/ng.3914
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3914
This article is cited by
-
Exome sequence analysis identifies rare coding variants associated with a machine learning-based marker for coronary artery disease
Nature Genetics (2024)
-
MSGene: a multistate model using genetic risk and the electronic health record applied to lifetime risk of coronary artery disease
Nature Communications (2024)
-
Protein interaction networks in the vasculature prioritize genes and pathways underlying coronary artery disease
Communications Biology (2024)
-
CCDC92 promotes podocyte injury by regulating PA28α/ABCA1/cholesterol efflux axis in type 2 diabetic mice
Acta Pharmacologica Sinica (2024)
-
Genomic approaches to identify and investigate genes associated with atrial fibrillation and heart failure susceptibility
Human Genomics (2023)