Abstract
Environmental enrichment increases adult hippocampal neurogenesis and alters hippocampal-dependent behavior in rodents. To investigate a causal link between these two observations, we analyzed the effect of enrichment on spatial learning and anxiety-like behavior while blocking adult hippocampal neurogenesis. We report that environmental enrichment alters behavior in mice regardless of their hippocampal neurogenic capability, providing evidence that the newborn cells do not mediate these effects of enrichment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gage, F.H. Science 287, 1433–1438 (2000).
van Praag, H., Kempermann, G. & Gage, F.H. Nat. Rev. Neurosci. 1, 191–198 (2000).
Kempermann, G., Kuhn, H.G. & Gage, F.H. Nature 386, 493–495 (1997).
Bruel-Jungerman, E., Laroche, S. & Rampon, C. Eur. J. Neurosci. 21, 513–521 (2005).
Shors, T.J., Townsend, D.A., Zhao, M., Kozorovitskiy, Y. & Gould, E. Hippocampus 12, 578–584 (2002).
Snyder, J.S., Hong, N.S., McDonald, R.J. & Wojtowicz, J.M. Neuroscience 130, 843–852 (2005).
Santarelli, L. et al. Science 301, 805–809 (2003).
Brown, J. et al. Eur. J. Neurosci. 17, 2042–2046 (2003).
Gray, J.A. & McNaughton, N. The Neuropsychology of Anxiety (Oxford Univ. Press, New York, 2000).
Nilsson, M., Perfilieva, E., Johansson, U., Orwar, O. & Eriksson, P.S. J. Neurobiol. 39, 569–578 (1999).
Roy, V., Belzung, C., Delarue, C. & Chapillon, P. Physiol. Behav. 74, 313–320 (2001).
Monje, M.L., Mizumatsu, S., Fike, J.R. & Palmer, T.D. Nat. Med. 8, 955–962 (2002).
Couillard-Despres, S. et al. Eur. J. Neurosci. 21, 1–14 (2005).
Elliott, B.M. & Grunberg, N.E. Behav. Brain Res. 165, 187–196 (2005).
Dupret, D. et al. Eur. J. Neurosci. 22, 778–783 (2005).
Acknowledgements
We thank J.A. Gordon, C.O. Lacefield and A. Sahay for their comments on the manuscript. This work was supported by US National Institute of Mental Health grants 5R01MH068542-03, 5T32MH018264-22 and 5F31MH069089-02.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
CD68 immunoreactivity in the dentate gyrus and hilus of mice. (a) Irradiated mice exhibited significantly more CD68-positive cells than sham mice one month after x-irradiation (T(1,4) = 3.425, p = 0.027). No difference was observed between sham and irradiated groups two months after irradiation (T(1,4) = 0.079, p = 0.941). (b) Representative image showing CD68 positive cells in the dentate gyrus and hilus one month after irradiation. Error bars = 1 S.E.M. (PDF 2715 kb)
Supplementary Fig. 2
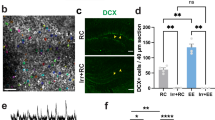
Images of (a) BrdU and (b) doublecortin immunohistochemistry in the dentate gyrus after housing in either standard or enriched environment, and treatment with either sham or hippocampal x-irradiation. Immunostaining of both BrdU and doublecortin is increased in enriched animals that received sham treatment (see Fig. 2). When animals in both standard and enriched housing received irradiation treatment, absence of BrdU and doublecortin immunostaining was observed. (PDF 28309 kb)
Supplementary Fig. 3
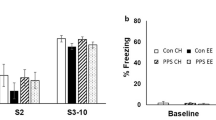
Proportion of time active in the habituation of activity test. On both days of the test enriched animals spent less time active (F's(1,60) > 14, p's < 0.001) and exhibited steeper within-session declines in activity (housing X minute interaction: F’s(2,120) > 13, p's < 0.001). There was no effect of irradiation treatment on either day (F’s(1,60) < 1), and irradiation did not interact significantly with the other variables. Error bars = 1 S.E.M. (PDF 467 kb)
Supplementary Fig. 4
Latency to feed in the novelty-suppressed feeding test. Male mice were transferred to the enriched environment immediately after irradiation and then tested after 6 weeks of enrichment. Enriched mice exhibit a significantly reduced latency to feed (F(1,75) = 36.647, p < 0.001). There was no effect of irradiation (F(1,75) = 0.112), and no interaction between housing and irradiation treatment (F(1,75) = 0.554). n = 20 per group. Error bars = 1 S.E.M. (PDF 465 kb)
Rights and permissions
About this article
Cite this article
Meshi, D., Drew, M., Saxe, M. et al. Hippocampal neurogenesis is not required for behavioral effects of environmental enrichment. Nat Neurosci 9, 729–731 (2006). https://doi.org/10.1038/nn1696
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1696
This article is cited by
-
Adult hippocampal neurogenesis shapes adaptation and improves stress response: a mechanistic and integrative perspective
Molecular Psychiatry (2022)
-
Severe hydroxymethylbilane synthase deficiency causes depression-like behavior and mitochondrial dysfunction in a mouse model of homozygous dominant acute intermittent porphyria
Acta Neuropathologica Communications (2020)
-
Recruitment of parvalbumin and somatostatin interneuron inputs to adult born dentate granule neurons
Scientific Reports (2020)
-
The role of adult hippocampal neurogenesis in brain health and disease
Molecular Psychiatry (2019)
-
The role of the GABAA receptor Alpha 1 subunit in the ventral hippocampus in stress resilience
Scientific Reports (2019)