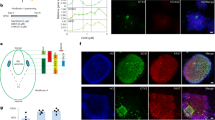
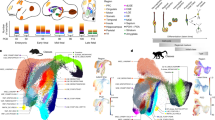
Abstract
Central norepinephrine-producing neurons comprise a diverse population of cells differing in anatomical location, connectivity, function and response to disease and environmental insult. The mechanisms that generate this diversity are unknown. Here we elucidate the lineal relationship between molecularly distinct progenitor populations in the developing mouse hindbrain and mature norepinephrine neuron subtype identity. We have identified four genetically separable subpopulations of mature norepinephrine neurons differing in their anatomical location, axon morphology and efferent projection pattern. One of the subpopulations showed an unexpected projection to the prefrontal cortex, challenging the long-held belief that the locus coeruleus is the sole source of norepinephrine projections to the cortex. These findings reveal the embryonic origins of central norepinephrine neurons and provide multiple molecular points of entry for future study of individual norepinephrine circuits in complex behavioral and physiological processes including arousal, attention, mood, memory, appetite and homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Berridge, C.W. & Waterhouse, B.D. The locus coeruleus-noradrenergic system: modulation of behavioral state and state-dependent cognitive processes. Brain Res. Brain Res. Rev. 42, 33–84 (2003).
Rinaman, L. Hindbrain noradrenergic A2 neurons: diverse roles in autonomic, endocrine, cognitive, and behavioral functions. Am. J. Physiol. Regul. Integr. Comp. Physiol. 300, R222–R235 (2011).
Sara, S.J. & Bouret, S. Orienting and reorienting: the locus coeruleus mediates cognition through arousal. Neuron 76, 130–141 (2012).
Dahlström, A. & Fuxe, K. Evidence for the existence of monoamine containing neurons in the central nervous system, I. Demonstration of monoamines in the cell bodies of brain stem neurons. Acta Physiol. Scand. 62 (suppl. 232): 1–55 (1964).
Grzanna, R. & Fritschy, J.M. Efferent projections of different subpopulations of central noradrenaline neurons. Prog. Brain Res. 88, 89–101 (1991).
German, D.C. et al. Disease-specific patterns of locus coeruleus cell loss. Ann. Neurol. 32, 667–676 (1992).
Brunnström, H., Friberg, N., Lindberg, E. & Englund, E. Differential degeneration of the locus coeruleus in dementia subtypes. Clin. Neuropathol. 30, 104–110 (2011).
Benarroch, E.E., Schmeichel, A.M., Low, P.A., Sandroni, P. & Parisi, J.E. Loss of A5 noradrenergic neurons in multiple system atrophy. Acta Neuropathol. 115, 629–634 (2008).
Bertrand, E., Lechowicz, W., Szpak, G.M. & Dymecki, J. Qualitative and quantitative analysis of locus coeruleus neurons in Parkinson's disease. Folia Neuropathol. 35, 80–86 (1997).
Miller, M.A., Kolb, P.E., Leverenz, J.B., Peskind, E.R. & Raskind, M.A. Preservation of noradrenergic neurons in the locus ceruleus that coexpress galanin mRNA in Alzheimer's disease. J. Neurochem. 73, 2028–2036 (1999).
Busch, C., Bohl, J. & Ohm, T.G. Spatial, temporal and numeric analysis of Alzheimer changes in the nucleus coeruleus. Neurobiol. Aging 18, 401–406 (1997).
Mohideen, S.S., Ichihara, G., Ichihara, S. & Nakamura, S. Exposure to 1-bromopropane causes degeneration of noradrenergic axons in the rat brain. Toxicology 285, 67–71 (2011).
Soulage, C. et al. Central and peripheral changes in catecholamine biosynthesis and turnover in rats after a short period of ozone exposure. Neurochem. Int. 45, 979–986 (2004).
Del Pino, J. et al. Effects of prenatal and postnatal exposure to amitraz on norepinephrine, serotonin and dopamine levels in brain regions of male and female rats. Toxicology 287, 145–152 (2011).
Slotkin, T.A. & Seidler, F.J. Mimicking maternal smoking and pharmacotherapy of preterm labor: fetal nicotine exposure enhances the effect of late gestational dexamethasone treatment on noradrenergic circuits. Brain Res. Bull. 86, 435–440 (2011).
Swanson, L.W. The locus coeruleus: a cytoarchitectonic, Golgi and immunohistochemical study in the albino rat. Brain Res. 110, 39–56 (1976).
Dymecki, S.M. & Kim, J.C. Molecular neuroanatomy's “three Gs”: a primer. Neuron 54, 17–34 (2007).
Champagnat, J., Morin-Surun, M.P., Fortin, G. & Thoby-Brisson, M. Developmental basis of the rostro-caudal organization of the brainstem respiratory rhythm generator. Phil. Trans. R. Soc. Lond. B 364, 2469–2476 (2009).
Gray, P.A. et al. Developmental origin of preBotzinger complex respiratory neurons. J. Neurosci. 30, 14883–14895 (2010).
Grossmann, K.S., Giraudin, A., Britz, O., Zhang, J. & Goulding, M. Genetic dissection of rhythmic motor networks in mice. Prog. Brain Res. 187, 19–37 (2010).
Dasen, J.S. & Jessell, T.M. Hox networks and the origins of motor neuron diversity. Curr. Top. Dev. Biol. 88, 169–200 (2009).
Lumsden, A. & Krumlauf, R. Patterning the vertebrate neuraxis. Science 274, 1109–1115 (1996).
Krumlauf, R. et al. Hox homeobox genes and regionalisation of the nervous system. J. Neurobiol. 24, 1328–1340 (1993).
Chambers, D. et al. Rhombomere-specific analysis reveals the repertoire of genetic cues expressed across the developing hindbrain. Neural Dev. 4, 6 (2009).
Aroca, P., Lorente-Canovas, B., Mateos, F.R. & Puelles, L. Locus coeruleus neurons originate in alar rhombomere 1 and migrate into the basal plate: Studies in chick and mouse embryos. J. Comp. Neurol. 496, 802–818 (2006).
Gaufo, G.O., Wu, S. & Capecchi, M.R. Contribution of Hox genes to the diversity of the hindbrain sensory system. Development 131, 1259–1266 (2004).
Simon, H.H., Scholz, C. & O'Leary, D.D. Engrailed genes control developmental fate of serotonergic and noradrenergic neurons in mid- and hindbrain in a gene dose-dependent manner. Mol. Cell Neurosci. 28, 96–105 (2005).
Awatramani, R., Soriano, P., Rodriguez, C., Mai, J.J. & Dymecki, S.M. Cryptic boundaries in roof plate and choroid plexus identified by intersectional gene activation. Nat. Genet. 35, 70–75 (2003).
Farago, A.F., Awatramani, R.B. & Dymecki, S.M. Assembly of the brainstem cochlear nuclear complex is revealed by intersectional and subtractive genetic fate maps. Neuron 50, 205–218 (2006).
Jensen, P. et al. Redefining the serotonergic system by genetic lineage. Nat. Neurosci. 11, 417–419 (2008).
Bang, S.J., Jensen, P., Dymecki, S.M. & Commons, K.G. Projections and interconnections of genetically defined serotonin neurons in mice. Eur. J. Neurosci. 35, 85–96 (2012).
Engleka, K.A. et al. Islet1 derivatives in the heart are of both neural crest and second heart field origin. Circ. Res. 110, 922–926 (2012).
Li, J.Y., Lao, Z. & Joyner, A.L. Changing requirements for Gbx2 in development of the cerebellum and maintenance of the mid/hindbrain organizer. Neuron 36, 31–43 (2002).
Voiculescu, O., Charnay, P. & Schneider-Maunoury, S. Expression pattern of a Krox-20/Cre knock-in allele in the developing hindbrain, bones, and peripheral nervous system. Genesis 26, 123–126 (2000).
O'Gorman, S. Second branchial arch lineages of the middle ear of wild-type and Hoxa2 mutant mice. Dev. Dyn. 234, 124–131 (2005).
Goridis, C. & Rohrer, H. Specification of catecholaminergic and serotonergic neurons. Nat. Rev. Neurosci. 3, 531–541 (2002).
Paxinos, G. & Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates (Elsevier Academic, 2004).
Hökfelt, T., Fuxe, K., Goldstein, M. & Johansson, O. Evidence for adrenaline neurons in the rat brain. Acta Physiol. Scand. 89, 286–288 (1973).
Garel, S., Garcia-Dominguez, M. & Charnay, P. Control of the migratory pathway of facial branchiomotor neurones. Development 127, 5297–5307 (2000).
Fritschy, J.M. & Grzanna, R. Distribution of locus coeruleus axons within the rat brainstem demonstrated by Phaseolus vulgaris leucoagglutinin anterograde tracing in combination with dopamine-beta-hydroxylase immunofluorescence. J. Comp. Neurol. 293, 616–631 (1990).
Aston-Jones, G. & Cohen, J.D. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu. Rev. Neurosci. 28, 403–450 (2005).
Lorang, D., Amara, S.G. & Simerly, R.B. Cell-type-specific expression of catecholamine transporters in the rat brain. J. Neurosci. 14, 4903–4914 (1994).
Sawchenko, P.E. & Swanson, L.W. Central noradrenergic pathways for the integration of hypothalamic neuroendocrine and autonomic responses. Science 214, 685–687 (1981).
Mejías-Aponte, C.A., Drouin, C. & Aston-Jones, G. Adrenergic and noradrenergic innervation of the midbrain ventral tegmental area and retrorubral field: prominent inputs from medullary homeostatic centers. J. Neurosci. 29, 3613–3626 (2009).
Chandler, D. & Waterhouse, B.D. Evidence for broad versus segregated projections from cholinergic and noradrenergic nuclei to functionally and anatomically discrete subregions of prefrontal cortex. Front. Behav. Neurosci. 6, 20 (2012).
Forray, M.I., Gysling, K., Andres, M.E., Bustos, G. & Araneda, S. Medullary noradrenergic neurons projecting to the bed nucleus of the stria terminalis express mRNA for the NMDA-NR1 receptor. Brain Res. Bull. 52, 163–169 (2000).
Myers, E.A., Banihashemi, L. & Rinaman, L. The anxiogenic drug yohimbine activates central viscerosensory circuits in rats. J. Comp. Neurol. 492, 426–441 (2005).
Davenport, P.W. & Vovk, A. Cortical and subcortical central neural pathways in respiratory sensations. Respir. Physiol. Neurobiol. 167, 72–86 (2009).
Adams, D.J. et al. A genome-wide, end-sequenced 129Sv BAC library resource for targeting vector construction. Genomics 86, 753–758 (2005).
Warming, S., Costantino, N., Court, D.L., Jenkins, N.A. & Copeland, N.G. Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res. 33, e36 (2005).
Raymond, C.S. & Soriano, P. High-efficiency FLP and PhiC31 site-specific recombination in mammalian cells. PLoS ONE 2, e162 (2007).
Liu, P., Jenkins, N.A. & Copeland, N.G. A highly efficient recombineering-based method for generating conditional knockout mutations. Genome Res. 13, 476–484 (2003).
Thomas, S.A., Marck, B.T., Palmiter, R.D. & Matsumoto, A.M. Restoration of norepinephrine and reversal of phenotypes in mice lacking dopamine beta-hydroxylase. J. Neurochem. 70, 2468–2476 (1998).
Thomas, S.A., Matsumoto, A.M. & Palmiter, R.D. Noradrenaline is essential for mouse fetal development. Nature 374, 643–646 (1995).
Lewandoski, M., Meyers, E.N. & Martin, G.R. Analysis of Fgf8 gene function in vertebrate development. Cold Spring Harb. Symp. Quant. Biol. 62, 159–168 (1997).
Kimmel, R.A. et al. Two lineage boundaries coordinate vertebrate apical ectodermal ridge formation. Genes Dev. 14, 1377–1389 (2000).
Jin, S.H., Kim, H.J., Harris, D.C. & Thomas, S.A. Postnatal development of the cerebellum and the CNS adrenergic system is independent of norepinephrine and epinephrine. J. Comp. Neurol. 477, 300–309 (2004).
Schofield, B.R. Retrograde axonal tracing with fluorescent markers. Curr. Protoc. Neurosci. Ch. 1, Unit 1 17 (2008).
Acknowledgements
We thank S. Dymecki (Harvard Medical School) for Hoxa2-cre and RC::FrePe mice and P. Charnay (INSERM) for Krox20cre mice. We thank T. Wolfgang, G. Keeley and the National Institute of Environmental Health Sciences Fluorescence Microscopy, Vivarium, Knockout Mice and Statistics services for assistance. This research was supported by the Intramural Research Program of the US National Institutes of Health, National Institute of Environmental Health Sciences (ZIA-ES-102805).
Author information
Authors and Affiliations
Contributions
S.D.R. and P.J. conceived the project and designed the experiments. S.D.R. contributed to the execution and analysis of all of the experiments and prepared the figures. N.W.P. designed, generated and characterized the DbhFlpo mouse allele and prepared Supplementary Figure 1. J.d.M. designed and conducted the retrograde labeling study. S.D.R., N.W.P. and P.J. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures and Text
Supplementary Figures 1–3 and Supplementary Tables 1 and 2 (PDF 2214 kb)
Rights and permissions
About this article
Cite this article
Robertson, S., Plummer, N., de Marchena, J. et al. Developmental origins of central norepinephrine neuron diversity. Nat Neurosci 16, 1016–1023 (2013). https://doi.org/10.1038/nn.3458
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3458