Abstract
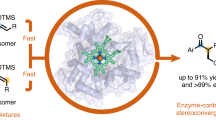
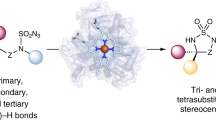
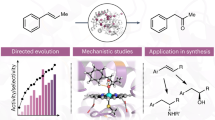
Whole-cell catalysts for non-natural chemical reactions will open new routes to sustainable production of chemicals. We designed a cytochrome 'P411' with unique serine-heme ligation that catalyzes efficient and selective olefin cyclopropanation in intact Escherichia coli cells. The mutation C400S in cytochrome P450BM3 gives a signature ferrous CO Soret peak at 411 nm, abolishes monooxygenation activity, raises the resting-state FeIII-to-FeII reduction potential and substantially improves NAD(P)H-driven activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
19 December 2013
In the version of this article initially published, the Protein Data Bank codes for the P450 and P411 constructs were inadvertently switched. Accession code 4H23 actually corresponds to the P411 structure, and 4H24 corresponds to the P450 structure. The error has been corrected in the HTML and PDF versions of the article.
References
Ajikumar, P.K. et al. Science 330, 70–74 (2010).
Westfall, P.J. et al. Proc. Natl. Acad. Sci. USA 109, E111–E118 (2012).
Kataoka, M. et al. Appl. Microbiol. Biotechnol. 62, 437–445 (2003).
Boyce, M. & Bertozzi, C.R. Nat. Methods 8, 638–642 (2011).
Coelho, P.S., Brustad, E.M., Kannan, A. & Arnold, F.H. Science 339, 307–310 (2013).
Lebel, H., Marcoux, J.-F., Molinaro, C. & Charette, A.B. Chem. Rev. 103, 977–1050 (2003).
Evans, D.A., Woerpel, K.A., Hinman, M.M. & Faul, M.M. J. Am. Chem. Soc. 113, 726–728 (1991).
Davies, H.M.L. & Venkataramani, C. Org. Lett. 5, 1403–1406 (2003).
Maas, G. Chem. Soc. Rev. 33, 183–190 (2004).
Ost, T.W.B. et al. Biochemistry 40, 13421–13429 (2001).
Wuttke, D.S. & Gray, H.B. Curr. Opin. Struct. Biol. 3, 555–563 (1993).
Reedy, C.J., Elvekrog, M.M. & Gibney, B.R. Nucleic Acids Res. 36, D307–D313 (2008).
Dawson, J.H. Science 240, 433–439 (1988).
Vatsis, K.P., Peng, H.-M. & Coon, M.J. J. Inorg. Biochem. 91, 542–553 (2002).
Wolf, J.R., Hamaker, C.G., Djukic, J.-P., Kodadek, T. & Woo, L.K. J. Am. Chem. Soc. 117, 9194–9199 (1995).
Perera, R., Sono, M., Voegtle, H.L. & Dawson, J.H. Arch. Biochem. Biophys. 507, 119–125 (2011).
Dunford, A.J., Girvan, H.M., Scrutton, N.S. & Munro, A.W. Biochim. Biophys. Acta 1794, 1181–1189 (2009).
Wessjohann, L.A., Brandt, W. & Thiemann, T. Chem. Rev. 103, 1625–1648 (2003).
Penoni, A. et al. Eur. J. Inorg. Chem. 1452–1460 (2003).
Watanabe, N., Matsuda, H., Kuribayashi, H. & Hashimoto, S.-i. Heterocycles 42, 537–542 (1996).
Sambrook, J., Frisch, E. & Maniatis, T. Molecular Cloning: a Laboratory Manual, Vol. 2 (Cold Spring Harbor Laboratory Press, New York, 1989).
Omura, T. & Sato, R. J. Biol. Chem. 239, 2370–2378 (1964).
Kabsch, W. Acta Crystallogr. D Biol. Crystallogr. 66, 133–144 (2010).
Evans, P. Acta Crystallogr. D Biol. Crystallogr. 62, 72–82 (2006).
Haines, D.C., Tomchick, D.R., Machius, M. & Peterson, J.A. Biochemistry 40, 13456–13465 (2001).
Vagin, A. & Teplyakov, A. Acta Crystallogr. D Biol Crystallogr. 30, 1022–1025 (1997).
Bailey, S. Acta Crystallogr. D Biol Crystallogr. 50, 760–763 (1994).
Emsley, P. & Cowtan, K. Acta Crystallogr. D Biol Crystallogr. 60, 2126–2132 (2004).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Adams, P.D. et al. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Acknowledgements
This research is supported by the Gordon and Betty Moore Foundation through the Caltech Programmable Molecular Technology Initiative. E.M.B. was supported by US National Institutes of Health (NIH) postdoctoral award F32GM087102 and a generous startup fund from University of North Carolina–Chapel Hill (UNC). Z.J.W. was supported by NIH 1F32EB015846-01. M.E.E. was supported by NIH grant RO1-DK019038. We thank N. Peck for help with preparative-scale experiments. We thank the Redinbo laboratory at UNC for assistance with X-ray data collection. M.E.E. thanks J.D. Blakemore and J.R. Winkler for electrodes and helpful discussions.
Author information
Authors and Affiliations
Contributions
P.S.C., F.H.A. and E.M.B. conceived the project and wrote the paper; P.S.C., E.M.B. and Z.J.W. designed the experiments; E.M.B. and S.A.B. performed the crystallography; M.E.E. performed the redox titrations; P.S.C., Z.J.W. and A.K. performed the catalysis experiments; all authors discussed the results.
Corresponding authors
Ethics declarations
Competing interests
P.S.C., E.M.B., Z.J.W. and F.H.A. have filed through Caltech a provisional patent application that is based on results presented here.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–17, Supplementary Tables 1–12 (PDF 3560 kb)
Rights and permissions
About this article
Cite this article
Coelho, P., Wang, Z., Ener, M. et al. A serine-substituted P450 catalyzes highly efficient carbene transfer to olefins in vivo. Nat Chem Biol 9, 485–487 (2013). https://doi.org/10.1038/nchembio.1278
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1278
This article is cited by
-
Mechanistic manifold in a hemoprotein-catalyzed cyclopropanation reaction with diazoketone
Nature Communications (2023)
-
Chemodivergent C(sp3)–H and C(sp2)–H cyanomethylation using engineered carbene transferases
Nature Catalysis (2023)
-
Multifaceted personality and roles of heme enzymes in industrial biotechnology
3 Biotech (2023)
-
Dual-function enzyme catalysis for enantioselective carbon–nitrogen bond formation
Nature Chemistry (2021)
-
Engineering new catalytic activities in enzymes
Nature Catalysis (2020)