Abstract
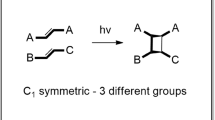
Packing constraints and precise placement of functional groups are the reason that organic molecules in the crystalline state often display unusual physical or chemical properties not observed in solution. Here we report a single-crystal-to-single-crystal dimerization of a bromodiacetylene that involves unusually large atom displacements as well as the cleavage and formation of several bonds. Density functional theory computations support a mechanism in which the dimerization is initiated by a [2 + 1] photocycloaddition favoured by the nature of carbon–carbon short contacts in the crystal structure. The reaction proceeded up to the theoretical degree of conversion without loss of crystallinity, and it was also performed on a preparative scale with good yield. Moreover, it represents the first synthetic pathway to (E)-1,2-dibromo-1,2-diethynylethenes, which could serve as synthetic intermediates for the preparation of molecular carbon scaffolds. Our findings both extend the scope of single-crystal-to-single-crystal reactions and highlight their potential as a synthetic tool for complex transformations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Kobatake, S., Takami, S., Muto, H., Ishikawa, T. & Irie, M. Rapid and reversible shape changes of molecular crystals on photoirradiation. Nature 446, 778–781 (2007).
Pekker, S. et al. Rotor-stator molecular crystals of fullerenes with cubane. Nature Mater. 4, 764–767 (2005).
Kuzmanich, G. et al. Oxyallyl exposed: an open-shell singlet with picosecond lifetimes in solution but persistent in crystals of a cyclobutanedione precursor. J. Am. Chem. Soc. 133, 2342–2345 (2011).
Lemouchi, C. et al. Ultra-fast rotors for molecular machines and functional materials via halogen bonding: crystals of 1,4-bis(iodoethynyl)bicyclo[2.2.2]-octane with distinct gigahertz rotation at two sites. J. Am. Chem. Soc. 133, 6371–6379 (2011).
Das, D., Jacobs, T. & Barbour, L. J. Exceptionally large positive and negative anisotropic thermal expansion of an organic crystalline material. Nature Mater. 9, 36–39 (2010).
Bolton, O., Lee, K., Kim, H-J., Lin, K. Y. & Kim, J. Activating efficient phosphorescence from purely organic materials by crystal design. Nature Chem. 3, 205–210 (2011).
Desiraju, G. R. Crystal engineering: a holistic view. Angew. Chem. Int. Ed. 46, 8342–8356 (2007).
Tanaka, K. & Toda, F. Solvent-free organic synthesis. Chem. Rev. 100, 1025–1074 (2000).
Ramamurthy, V. & Venkatesan, K. Photochemical reactions of organic crystals. Chem. Rev. 87, 433–481 (1987).
Halasz, I. Single-crystal-to-single-crystal reactivity: gray, rather than black or white. Crystal Growth Des. 10, 2817–2823 (2010).
Wegner, G. Topochemical reactions of monomers with conjugated triple bonds. I. Polymerization of derivatives of 2,4-hexadiyne-1,6-diols in the crystalline state. Z. Naturforsch. B 24, 824–832 (1969).
Enkelmann, V. Structural aspects of the topochemical polymerization of diacetylenes. Adv. Polym. Sci. 63, 91–136 (1984).
Zuilhof, H. et al. in Supramolecular Photosensitive and Electroactive Materials (ed. Nalwa, H. S.) 339–437 (Academic Press, 2001).
Xiao, J., Yang, M., Lauher, J. W. & Fowler, F. W. A supramolecular solution to a long-standing problem: the 1,6-polymerization of a triacetylene. Angew. Chem. Int. Ed. 39, 2132–2135 (2000).
Sun, A., Lauher, J. W. & Goroff, N. S. Preparation of poly(diiododiacetylene), an ordered conjugated polymer of carbon and iodine. Science 312, 1030–1034 (2006).
Xu, R., Gramlich, V. & Frauenrath, H. Alternating diacetylene copolymer utilizing perfluorophenyl–phenyl interactions. J. Am. Chem. Soc. 128, 5541–5547 (2006).
Xu, R., Schweizer, W. B. & Frauenrath, H. Perfluorophenyl–phenyl interactions in the crystallization and topochemical polymerization of triacetylene monomers. Chem. Eur. J. 15, 9105–9116 (2009).
Matsumoto, A., Matsumura, T. & Aoki, S. Stereospecific polymerisation of diethyl (Z,Z)-hexa-2,4-dienedioate in the crystalline state. J. Chem. Soc. Chem. Commun. 1389–1390 (1994).
Hoang, T., Lauher, J. W. & Fowler, F. W. The topochemical 1,6-polymerization of a triene. J. Am. Chem. Soc. 124, 10656–10657 (2002).
Cohen, M. D. & Schmidt, G. M. J. Topochemistry. I. A survey. J. Chem. Soc. 1996–2000 (1964).
Coates, G. W. et al. Phenyl–perfluorophenyl stacking interactions: topochemical [2 + 2] photodimerization and photopolymerization of olefinic compounds. J. Am. Chem. Soc. 120, 3641–3649 (1998).
Gao, X., Friscic, T. & MacGillivray, L. R. Supramolecular construction of molecular ladders in the solid state. Angew. Chem. Int. Ed. 43, 232–236 (2004).
Varshney, D. B., Papaefstathiou, G. S. & MacGillivray, L. R. Site-directed regiocontrolled synthesis of a ‘head-to-head’ photodimer via a single-crystal-to-single-crystal transformation involving a linear template. Chem. Commun. 7, 1964–1965 (2002).
Atkinson, M. B. J. et al. A solid-state trimerization of a diene diacid affords a bicyclobutyl: reactant structure from X-ray powder data and product separation and structure determination via co-crystalliztion. Chem. Commun. 47, 236–238 (2011).
Karunatilaka, C. et al. Softening and hardening of macro- and nano-sized organic cocrystals in a single-crystal transformation. Angew. Chem. Int. Ed. 50, 8642–8646 (2011).
MacGillivray, L. R. et al. Supramolecular control of reactivity in the solid state: from templates to ladderanes to metal–organic frameworks. Acc. Chem. Res. 41, 280–291 (2008).
Scheffer, J. R. Crystal lattice control of unimolecular photorearrangements. Acc. Chem. Res. 13, 283–290 (1980).
Siemsen, P., Livingston, R. C. & Diederich, F. Acetylenic coupling: a powerful tool in molecular construction. Angew. Chem. Int. Ed. 39, 2632–2657 (2000).
Trost, B. M., Chan, C. & Ruhter, G. Metal-mediated approach to enynes. J. Am. Chem. Soc. 109, 3486–3487 (1987).
Barluenga, J., de la Rua, R. B., de Saa, D., Ballesteros, A. & Tomas, M. Formal alkyne insertion into alkoxycarbene complexes: simple access to enantiopure group 6 alkynyl(alkoxy)carbene complexes. Angew. Chem. Int. Ed. 44, 4981–4983 (2005).
Barluenga, J., Gonzalez, J. M., Llorente, I. & Campos, P. J. 1-Iodoalkynes can also be dimerized: a new head-to-tail coupling. Angew. Chem. Int. Ed. Engl. 32, 893–894 (1993).
Diederich, F. Carbon scaffolding: building acetylenic all-carbon and carbon-rich compounds. Nature 369, 199–207 (1994).
Pahadi, N. K., Camacho, D. H., Nakamura, I. & Yamamoto, Y. Palladium-catalyzed dimerization of conjugated diynes: synthesis of (E)-1,2-divinyldiethynylethenes having donor and acceptor chromophores at the terminus of alkyne. J. Org. Chem. 71, 1152–1155 (2006).
Barluenga, J. et al. Metal carbene dimerization: versatile approach to polyalkynylethenes. Angew. Chem. Int. Ed. 47, 6225–6228 (2008).
Kim, S., Kim, S., Lee, T., Ko, H. & Kim, D. A new, iterative strategy for the synthesis of unsymmetrical polyynes: application to the total synthesis of 15,16-dihydrominquartynoic acid. Org. Lett. 6, 3601–3604 (2004).
Hoheisel, T. N. & Frauenrath, H. A convenient Negishi protocol for the synthesis of glycosylated oligo(ethynylene)s. Org. Lett. 10, 4525–4528 (2008).
Metrangolo, P. & Resnati, G. Halogen bonding: a paradigm in supramolecular chemistry. Chem. Eur. J. 7, 2511–2519 (2001).
Auffinger, P., Hays, F. A., Westhof, E. & Ho, P. S. Halogen bonds in biological molecules. Proc. Natl Acad. Sci. USA 101, 16789–16794 (2004).
Plourde, G. W. II, Warner, P. M., Parrish, D. A. & Jones, G. B. Halo-enediynes: probing the electronic and stereoelectronic contributions to the Bergman cycloaromatization. J. Org. Chem. 67, 5369–5374 (2002).
Desiraju, G. R. & Kannan, V. What is the maximum yield in the solid state cinnamic acid dimerization? A combinatorial mathematical approach. Proc. Indian Acad. Sci. (Chem. Sci.) 96, 351–362 (1986).
Cohen, M. D., Schmidt, G. M. J. & Sonntag, F. I. Topochemistry. II. The photochemistry of trans-cinnamic acids. J. Chem. Soc. 2000–2013 (1964).
Schmidt, G. M. J. Topochemistry. III. The crystal chemistry of some trans-cinnamic acids. J. Chem. Soc. 2014–2021 (1964).
Schmider, H. L. & Becke, A. D. Chemical content of the kinetic energy density. J. Mol. Struct. (Theochem) 527, 51–61 (2000).
Steinmann, S. N., Mo, Y. & Corminboeuf, C. How do electron localization functions describe π-electron delocalization? Phys. Chem. Chem. Phys. 13, 20584–20592 (2011).
Steinmann, S. N. & Corminboeuf, C. A system-dependent density-based dispersion correction. J. Chem. Theo. Comp. 6, 1990–2001 (2010).
Steinmann, S. N. & Corminboeuf, C. A generalized-gradient approximation exchange hole model for dispersion coefficients. J. Chem. Phys. 134, 044117 (2011).
Seburg, R. A. & McMahon, R. J. Automerizations and isomerization in propynylidene (HCCCH), propadienylidene (H2CCC), and cyclopropenylidene (c-C3H2). Angew. Chem. Int. Ed. Engl. 34, 2009–2012 (1995).
Thomas, P. S., Bowling, N. P. & McMahon, R. J. Spectroscopy and photochemistry of triplet methylpentadiynylidene. J. Am. Chem. Soc. 131, 8649–8659 (2009).
Ito, S. & Morita, N. Product class 10: fulvenes. Sci. Synth. 45a, 429–482 (2009).
Kotting, C., Sander, W. & Senzlober, M. Evidence for the non-concerted addition of difluorovinylidene to acetylenes. Chem. Eur. J. 4, 2360–2365 (1998).
Baird, M. S. Thermally induced cyclopropene–carbene rearrangements: an overview. Chem. Rev. 103, 1271–1294 (2003).
Acknowledgements
Funding from the European Research Council (ERC Grant 239831, ‘OrgElNanoCarbMater’), the Swiss National Science Foundation (SNF Grant 200020-121812) and ETH Zürich (ETH Research Grant ETH-05 08-2) is acknowledged. T.K.T. acknowledges financial support from the Marie Heim-Voegtlin grant PMPDP2_134151/1. C.C. thanks the Sandoz Family Foundation and the Swiss National Science Foundation (Grants 200021-121577 and 200021-137529).
Author information
Authors and Affiliations
Contributions
T.N.H. and S.S. synthesized, characterized and crystallized the monomers, conducted the dimerization experiments, characterized the products and contributed to the preparation of the manuscript. R.M. assisted in the synthesis. T.K.T. performed the DFT computations designed and directed by C.C.; both these authors contributed to the preparation of the manuscript. A.S. performed the ESR measurements. R.S. and W.B.S. performed the X-ray crystal structure analyses. H.F. designed and directed the research and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 10599 kb)
Supplementary information
Crystallographic data for compound 1. (CIF 20 kb)
Supplementary information
Crystallographic data for compound 3. (CIF 20 kb)
Supplementary information
Crystallographic data for a second polymorph of compound 1. (CIF 17 kb)
Rights and permissions
About this article
Cite this article
Hoheisel, T., Schrettl, S., Marty, R. et al. A multistep single-crystal-to-single-crystal bromodiacetylene dimerization. Nature Chem 5, 327–334 (2013). https://doi.org/10.1038/nchem.1575
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1575
This article is cited by
-
X-ray induced dimerization of cinnamic acid: Time-resolved inelastic X-ray scattering study
Scientific Reports (2015)
-
Functional carbon nanosheets prepared from hexayne amphiphile monolayers at room temperature
Nature Chemistry (2014)