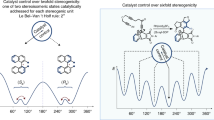
Abstract
Stereoselective chemical synthesis requires the two faces of a π bond to be differentiated. Theoretically sound qualitative models for understanding stereoinduction seem to break down in sterically unbiased cyclic systems. Presented here as the distortional asymmetry model is new insight that identifies circumstances where distortional ground state contributions are highly asymmetric and thereby contribute significantly to face selectivity. Out-of-plane distortional potential calculations, transition state calculations and molecular orbital analysis agree with experimental data that cannot otherwise be attributed to steric, torsion, polar or emergent transition state stabilizing effects. The model is readily understood in terms of reaction theory. The explanatory power of the model is also discussed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chem. Rev. 99(5), 1067–1480 (1999).
Dannenberg, J. J. Using perturbation and frontier molecular orbital theory to predict diastereofacial selectivity. Chem. Rev. 99, 1225–1242 (1999).
Tomoda, S. The exterior frontier orbital extension model. Chem. Rev. 99, 1243–1264 (1999).
Cieplak, A. S. Inductive and resonance effects of substituents on π-face selection. Chem. Rev. 99, 1265–1336 (1999).
Ohwada, T. Orbital-controlled stereoselection in sterically unbiased cyclic systems. Chem. Rev. 99, 1337–1376 (1999).
Gung, B. W. Structure distortions in heteroatom-substituted cyclohexanones, adamantanones, and adamantanes: origin of diastereofacial selectivity. Chem. Rev. 99, 1377–1386 (1999).
Kaselji, M., Chung, W. S. & le Noble, W. J. Face selection in addition and elimination in sterically unbiased systems. Chem. Rev. 99, 1387–1414 (1999).
Adcock, W. & Trout, N. A. Nature of the electronic factor governing diastereofacial selectivity in some reactions of rigid saturated model substrates. Chem. Rev. 99, 1415–1436 (1999).
Mehta, G. & Chandrasekar, J. Electronic control of facial selection in additions to sterically unbiased ketones and olefins. Chem. Rev. 99, 1437–1468 (1999).
Wipf, P. & Jung, J.-K. Nucleophilic additions to 4,4-disubstituted 2,5-cyclohexadienones: can dipole effects control facial selectivity? Chem. Rev. 99, 1469–1480 (1999).
Polanyi, J. C. Concepts in reaction dynamics. Acc. Chem. Res. 5, 161–168 (1972).
Zare, R. N. Laser control of chemical reactions. Science 279, 1875–1879 (1998).
Xidos, J. D., Poirier, R. A., Pye, C. C. & Burnell, D. J. An ab initio study of facial selectivity in the Diels-Alder reaction. J. Org. Chem. 63, 105–112 (1998).
Barnes, G. L. & Hase, W. L. Transition state analysis: Bent out of shape. Nature Chem. 1, 103–104 10.1038/nchem.193(2009).
Xu, L., Doubleday, C. E. & Houk, K. N. Dynamics of 1,3-dipolar cycloaddition reactions of diazonium betaines to acetylene and ethylene: bending vibrations facilitate reaction. Angew. Chem. Int. Ed. 48, 2746–2748 (2009).
Ess, D. H. & Houk, K. N. Distortion/interaction energy control of 1,3-dipolar cycloaddition reactivity. J. Am. Chem. Soc. 129, 10646–10647 (2007).
Wipff, G. & Morokuma, K. Nonplanarity of π systems. An ab initio study of norbornene and norbornadiene. Tetrahedron Lett. 21, 4445–4448 (1980).
Laube, T. & Hollenstein, S. Crystal structures of two activated cylcohexanones with opposite pyramidalizations of the carbonyl groups. J. Am. Chem. Soc. 114, 8812–8817 (1992).
Holthausen, M. C. & Koch, W. Double-bond geometry in norbornene, sesquinorbornenes, and related compounds. a high-level quantum chemical investigation. J. Phys. Chem. 97, 10021–10027 (1993).
Tsuji, M., Ohwada, T. & Shudo, K. A cyclopropyl group shows reverse facial selectivity depending on the bicyclic ring system. Tetrahedron Lett. 38, 6693–6696 (1997).
Winstein, S., Shatavsky, M., Norton, C. & Woodward, R. B. 7-Norbornenyl and 7-norbornyl cations. J. Am. Chem. Soc. 77, 4183–4184 (1955).
Frisch, M. J. et al. Gaussian 03, Revision C. 02 (Gaussian, 2004).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Houk, K. N. et al. Theory and modeling of stereoselective organic reactions. Science 231, 1108–1117 (1986).
Zheng, J., Zhoa, Y. & Truhlar, D. G. Representative benchmarck suites for barrier heights of diverse reaction types and assessment of electronic structure methods for thermochemical kinetics. J. Chem. Theory Comput. 3, 569–582 (2007).
Singleton, D. A., Merrigan, S. R., Liu, J. & Houk, K. N. Experimental geometry of the epoxidation transition state. J. Am. Chem. Soc. 119, 3385–3386 (1997).
Grote, R. F. & Hynes, J. T. The stable states picture of chemical reactions. II. Rate constants for condensed and gas phase reaction models. J. Chem. Phys. 73, 2715–2732 (1980).
Marcus, R. Transfer reactions in chemistry. Theory and experiment. Pure Appl. Chem. 69, 13–29 (1997).
Vázquez, S. & Camps, P. Chemistry of pyramidalized alkenes. Tetrahedron 61, 5147–5208 (2005).
Seebach, D., Zimmerman, J., Gysel, U., Ziegler, R. & Ha, T. K. Totally stereoselective additions to 2,6-disubstituted 1,3-dioxin-4-ones (chiral acetoacetic acid derivatives). Synthetic and mechanistic aspects of remote stereoselectivity. J. Am. Chem. Soc. 110, 4763–4772 (1988).
Cee, V. J., Cramer, C. J. & Evans, D. A. Theoretical investigation of enolborane addition to α-heteroatom-substituted aldehydes. Relevance of the Cornforth and polar Felkin-Anh models for asymmetric induction. J. Am. Chem. Soc. 128, 2920–2930 (2006).
Acknowledgements
Support from Merck is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
L.J.W. and R.V.K. designed experiments and analysed data, R.V.K. performed experiments, and L.J.W. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1692 kb)
Rights and permissions
About this article
Cite this article
Kolakowski, R., Williams, L. Stereoinduction by distortional asymmetry. Nature Chem 2, 303–307 (2010). https://doi.org/10.1038/nchem.577
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.577