Abstract
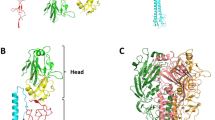
The three-dimensional structures of influenza virus haemagglutinins complexed with cell receptor analogues show sialic acids bound to a pocket of conserved amino acids surrounded by antibody-binding sites. Sialic acid fills the conserved pocket, demonstrating that it is the influenza virus receptor. The proximity of the antibody-binding sites suggests that antibodies neutralize virus infectivity by preventing virus-to-cell binding. The structures suggest approaches to the design of anti-viral drugs that could block attachment of viruses to cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gottschalk, A. in The Viruses Vol. 3 (eds Burnet, F. M. & Stanley, W. M.) 51–61 (Academic, New York, 1959).
Paulson, J. C. in The Receptors Vol. 2 (ed. Conn, P. M.) 131–219 (Academic, Orlando, 1985).
Paulson, J. C., Sadler, J. E. & Hill, R. L. J. biol. Chem. 254, 2120–2124 (1979).
Bergelson, L. D. et al. Eur. J. Biochem. 128, 467–474 (1982).
Suzuki, Y., Matsunaga, M. & Masumoto, M. J. biol. Chem. 260, 1362–1365 (1985).
Rogers, G. N. et al. Nature 304, 76–79 (1983).
Burley, S. K. & Petsko, G. A. Science 229, 23–28 (1985).
Quiocho, F. A. A. Rev. Biochem. 55, 287–315 (1986).
O'Connell, A. M. Acta crystallogr. B29, 2320–2328 (1973).
Richards, F. M. A. Rev. Biophys. Bioengng 6, 151–176 (1977).
Quiocho, F. A. in Current Topics in Microbiology and Immunology (in the press).
Quiocho, F. A., Sack, J. S. & Vyas, N. K. Nature 329, 561–564 (1987).
Wright, C. S. J. molec. Biol. 178, 91–104 (1984).
Kronis, K. A. & Carver, J. P. Biochemistry 24, 826–833 (1985).
Kelly, J. A., Sielecki, A. R., Sykes, B. D., James, M. N. G. & Phillips, D. C. Nature 282, 875–878 (1979).
Rogers, G. N. & Paulson, J. C. Virology 127, 361–373 (1983).
Pritchett, T. J., Brossmer, R., Rose, U. & Paulson, J. C. Virology, 160, 502–506 (1987).
Yewdell, J. W., Caton, A. J. & Gerhard, W. J. Virol. 57, 623–628 (1986).
Underwood, P. A. Arch. Virol. 84, 53–61 (1985).
Underwood, P. A., Skehel, J. J. & Wiley, D. C. J. Virol. 61, 206–208 (1987).
Daniels, R. S. et al. EMBO J. 6, 1459–1465 (1987).
Higa, H. H., Rogers, G. N. & Paulson, J. C. Virology 144, 279–282 (1985).
Anders, E. M., Scalzo, A. A., Rogers, G. N. & White, D. O. J. Virol. 60, 476–482 (1986).
Pritchett, T. J. thesis, Univ. of Calif., Los Angeles (1987).
Amit, A. G., Mariuzza, R. A., Phillips, S. E. V. & Poljak, R. J. Science 233, 747–753 (1986).
Colman, P. M. et al. Nature 326, 358–363 (1987).
Sheriff, S. et al. Proc. natn. Acad. Sci. U.S.A. 84, 8075–8079 (1987).
Mandel, B. Comprehensive Virology 15, 37–121 (eds Fraenkel-Conrat, H. & Wagner, R. R.) (Plenum, New York, 1979).
Dimmock, N. J. J. gen. Virol. 65, 1015–1022 (1984).
Dimmock, N. J. Trends biochem. Sci. 12, 70–75 (1987).
Naeve, C. W., Hinshaw, V. S. & Webster, R. G. J. Virol. 51, 567–569 (1984).
Laver, W. G. & Webster, R. G. Virology 51, 383–391 (1973).
Fang, R., Minjou, W., Huylebroeck, D., Devos, R. & Fiers, W. Cell 25, 315–323 (1981).
Ward, C. W. & Dopheide, T. A. Biochem. J. 195, 337–340 (1981).
Daniels, R. S., Skehel, J. J. & Wiley, D. C. J. gen. Virol. 66, 457–464 (1985).
Rogers, G. N. et al. J. biol. Chem. 260, 7362–7367 (1985).
Wilson, I. A., Skehel, J. J. & Wiley, D. C. Nature 289, 366–373 (1981).
Warren, L. J. biol. Chem. 234, 1971–1975 (1959).
Crawford, J. L. Ph. D. thesis, Harvard University (1977).
Kahn, R. et al. J. appl. Cryst. 15, 330–337 (1982).
Winkler, F. K., Schutt, C. E. & Harrison, S. C. Acta crystallogr. A35, 901–911 (1979).
Hendrikson, W. A. & Konnert, J. H. in Biomolecular Structure, Function, Conformation and Evolution (ed. Srinivasan, R.), Vol. 1, 43–57 (Pergamon, Oxford, 1981).
Skehel, J. J. et al. Proc. natn. Acad. Sci. U.S.A. 81, 1779–1783 (1984).
Colman, P. M., Varghese, J. N. & Laver, W. G. Nature 303, 41–44 (1983).
Wiley, D. C., Wilson, I. A. & Skehel, J. J. Nature 289, 373–378 (1981).
Rossmann, M. G. et al. Nature 317, 145–153 (1985).
Flippen, J. L. Acta crystallogr. B29, 1881–1886 (1973).
Bricogne, G. Acta crystallogr. A32, 832–847 (1976).
Weis, W. thesis, Harvard Univ. (1987).
Knossow, M. et al. Acta crystallogr. B42, 627–632 (1986).
Lesk, A. M. & Hardman, K. D. Science 216, 539–540 (1982).
Knossow, M., Daniels, R. S., Douglas, A. R., Skehel, J. J. & Wiley, D. C. Nature 311, 678–680 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weis, W., Brown, J., Cusack, S. et al. Structure of the influenza virus haemagglutinin complexed with its receptor, sialic acid. Nature 333, 426–431 (1988). https://doi.org/10.1038/333426a0
Issue Date:
DOI: https://doi.org/10.1038/333426a0
This article is cited by
-
Rapid and sensitive detection of SARS-CoV-2 virus in human saliva samples using glycan based nanozyme: a clinical study
Microchimica Acta (2024)
-
Structural characterisation of hemagglutinin from seven Influenza A H1N1 strains reveal diversity in the C05 antibody recognition site
Scientific Reports (2023)
-
The green tea catechin EGCG provides proof-of-concept for a pan-coronavirus attachment inhibitor
Scientific Reports (2022)
-
New approach in SARS-CoV-2 surveillance using biosensor technology: a review
Environmental Science and Pollution Research (2022)
-
Combination therapy of zinc and trimethoprim inhibits infection of influenza A virus in chick embryo
Virology Journal (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.