Abstract
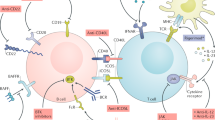
The cytokine TNF signals via two distinct receptors, TNF receptor 1 (TNFR1) and TNFR2, and is a central mediator of various immune-mediated diseases. Indeed, TNF-neutralizing biologic drugs have been in clinical use for the treatment of many inflammatory pathological conditions, including various rheumatic diseases, for decades. TNF has pleiotropic effects and can both promote and inhibit pro-inflammatory processes. The integrated net effect of TNF in vivo is a result of cytotoxic TNFR1 signalling and the stimulation of pro-inflammatory processes mediated by TNFR1 and TNFR2 and also TNFR2-mediated anti-inflammatory and tissue-protective activities. Inhibition of the beneficial activities of TNFR2 might explain why TNF-neutralizing drugs, although highly effective in some diseases, have limited benefit in the treatment of other TNF-associated pathological conditions (such as graft-versus-host disease) or even worsen the pathological condition (such as multiple sclerosis). Receptor-specific biologic drugs have the potential to tip the balance from TNFR1-mediated activities to TNFR2-mediated activities and enable the treatment of diseases that do not respond to current TNF inhibitors. Accordingly, a variety of reagents have been developed that either selectively inhibit TNFR1 or selectively activate TNFR2. Several of these reagents have shown promise in preclinical studies and are now in, or approaching, clinical trials.
Key points
-
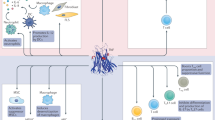
TNF is an important pleotropic cytokine that triggers complex immune-regulatory circuits of crucial relevance in tissue homeostasis as well as in many inflammatory diseases and pathological conditions.
-
The clinically approved TNF blockers inhibit activation of both the pro-inflammatory and cytotoxic TNF receptor 1 (TNFR1) and the largely anti-inflammatory and tissue-protective TNF receptor 2 (TNFR2).
-
TNFR2 promotes the expansion of regulatory T cells and enhances the suppressive activity of these cells, and is thus a promising therapeutic target in the treatment of autoimmunity.
-
Biologic drugs that selectively and/or preferentially interfere with TNFR1 activation are in clinical trials and are effective in various preclinical disease models, including models of autoimmune disease.
-
Various ligand-based and antibody-based TNFR2 agonists are therapeutically effective in preclinical disease models including collagen-induced arthritis.
-
TNF receptor-targeting reagents with conditional and/or local activity are in early preclinical development and have the potential to expand the spectrum of applications of TNF receptor-regulating biologics in the future.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Medler, J., Kucka, K. & Wajant, H. Tumor necrosis factor receptor 2 (TNFR2): an emerging target in cancer therapy. Cancers 14, 2603 (2022).
Medler, J. & Wajant, H. Tumor necrosis factor receptor-2 (TNFR2): an overview of an emerging drug target. Expert Opin. Ther. Targets 23, 295–307 (2019).
Rolski, F. & Błyszczuk, P. Complexity of TNF-α signaling in heart disease. J. Clin. Med. 9, 3267 (2020).
Salomon, B. L. Insights into the biology and therapeutic implications of TNF and regulatory T cells. Nat. Rev. Rheumatol. 17, 487–504 (2021).
Zahid, M. et al. Tumor necrosis factor alpha blockade and multiple sclerosis: exploring new avenues. Cureus 13, e18847 (2021).
Urquhart, L. Top companies and drugs by sales in 2021. Nat. Rev. Drug. Discov. 21, 251 (2022).
Hurrell, B. P. et al. TNFR2 signaling enhances ILC2 survival, function, and induction of airway hyperreactivity. Cell Rep. 29, 4509–4524.e4505 (2019).
Al-Lamki, R. S. et al. TL1A both promotes and protects from renal inflammation and injury. J. Am. Soc. Nephrol. 19, 953–960 (2008).
Suto, T. et al. TNFR2 is critical for TNF-induced rheumatoid arthritis fibroblast-like synoviocyte inflammation. Rheumatology 61, 4535–4546 (2022).
Wang, F. et al. IFN-γ-induced TNFR2 expression is required for TNF-dependent intestinal epithelial barrier dysfunction. Gastroenterology 131, 1153–1163 (2006).
Winzen, R., Wallach, D., Kemper, O., Resch, K. & Holtmann, H. Selective up-regulation of the 75-kDa tumor necrosis factor (TNF) receptor and its mRNA by TNF and IL-1. J. Immunol. 150, 4346–4353 (1993).
Blaser, H., Dostert, C., Mak, T. W. & Brenner, D. TNF and ROS crosstalk in inflammation. Trends Cell Biol. 26, 249–261 (2016).
van Loo, G. & Bertrand, M. J. M. Death by TNF: a road to inflammation. Nat. Rev. Immunol. 23, 289–303 (2023).
Wajant, H., Pfizenmaier, K. & Scheurich, P. Tumor necrosis factor signaling. Cell Death Differ. 10, 45–65 (2003).
Steeland, S., Libert, C. & Vandenbroucke, R. E. A new venue of TNF targeting. Int. J. Mol. Sci. 19, 1442 (2018).
Chandrasekharan, U. M. et al. TNFR2 depletion reduces psoriatic inflammation in mice by downregulating specific dendritic cell populations in lymph nodes and inhibiting IL-23/IL-17 pathways. J. Invest. Dermatol. 142, 2159–2172.e2159 (2022).
Chandrasekharan, U. M. et al. Tumor necrosis factor alpha (TNF-α) receptor-II is required for TNF-α-induced leukocyte-endothelial interaction in vivo. Blood 109, 1938–1944 (2007).
Funk, J. O. et al. Cutting edge: resistance to apoptosis and continuous proliferation of dendritic cells deficient for TNF receptor-1. J. Immunol. 165, 4792–4796 (2000).
Maney, N. J., Reynolds, G., Krippner-Heidenreich, A. & Hilkens, C. M. U. Dendritic cell maturation and survival are differentially regulated by TNFR1 and TNFR2. J. Immunol. 193, 4914–4923 (2014).
Calligaris, M. et al. Strategies to target ADAM17 in disease: from its discovery to the iRhom revolution. Molecules 26, 944 (2021).
Grell, M. et al. The transmembrane form of tumor necrosis factor is the prime activating ligand of the 80 kDa tumor necrosis factor receptor. Cell 83, 793–802 (1995).
Grell, M., Wajant, H., Zimmermann, G. & Scheurich, P. The type 1 receptor (CD120a) is the high-affinity receptor for soluble tumor necrosis factor. Proc. Natl Acad. Sci. USA 95, 570–575 (1998).
Medler, J. et al. TNFRSF receptor-specific antibody fusion proteins with targeting controlled FcγR-independent agonistic activity. Cell Death Dis. 10, 224 (2019).
Browning, J. L. et al. Characterization of surface lymphotoxin forms. Use of specific monoclonal antibodies and soluble receptors. J. Immunol. 154, 33–46 (1995).
Kucka, K. et al. Membrane lymphotoxin-α2β is a novel tumor necrosis factor (TNF) receptor 2 (TNFR2) agonist. Cell Death Dis. 12, 360 (2021).
Young, J. et al. Lymphotoxin-αβ heterotrimers are cleaved by metalloproteinases and contribute to synovitis in rheumatoid arthritis. Cytokine 51, 78–86 (2010).
Mauri, D. N. et al. LIGHT, a new member of the TNF superfamily, and lymphotoxin α are ligands for herpesvirus entry mediator. Immunity 8, 21–30 (1998).
Banner, D. W. et al. Crystal structure of the soluble human 55 kd TNF receptor-human TNF β complex: implications for TNF receptor activation. Cell 73, 431–445 (1993).
Kucka, K. & Wajant, H. Receptor oligomerization and its relevance for signaling by receptors of the tumor necrosis factor receptor superfamily. Front. Cell Dev. Biol. 8, 615141 (2020).
Mukai, Y. et al. Solution of the structure of the TNF-TNFR2 complex. Sci. Signal. 3, ra83 (2010).
Chen, X. et al. Progranulin does not bind tumor necrosis factor (TNF) receptors and is not a direct regulator of TNF-dependent signaling or bioactivity in immune or neuronal cells. J. Neurosci. 33, 9202–9213 (2013).
Etemadi, N., Webb, A., Bankovacki, A., Silke, J. & Nachbur, U. Progranulin does not inhibit TNF and lymphotoxin-α signalling through TNF receptor 1. Immunol. Cell Biol. 91, 661–664 (2013).
Lang, I., Füllsack, S. & Wajant, H. Lack of evidence for a direct interaction of progranulin and tumor necrosis factor receptor-1 and tumor necrosis factor receptor-2 from cellular binding studies. Front. Immunol. 9, 793 (2018).
Tang, W. et al. The growth factor progranulin binds to TNF receptors and is therapeutic against inflammatory arthritis in mice. Science 332, 478–484 (2011).
Rauert, H. et al. Membrane tumor necrosis factor (TNF) induces p100 processing via TNF receptor-2 (TNFR2). J. Biol. Chem. 285, 7394–7404 (2010).
Schneider, P. et al. Conversion of membrane-bound Fas(CD95) ligand to its soluble form is associated with downregulation of its proapoptotic activity and loss of liver toxicity. J. Exp. Med. 187, 1205–1213 (1998).
Sheehan, K. C. et al. Monoclonal antibodies specific for murine p55 and p75 tumor necrosis factor receptors: identification of a novel in vivo role for p75. J. Exp. Med. 181, 607–617 (1995).
Gerspach, J. et al. Restoration of membrane TNF-like activity by cell surface targeting and matrix metalloproteinase-mediated processing of a TNF prodrug. Cell Death Differ. 13, 273–284 (2006).
Karathanasis, C. et al. Single-molecule imaging reveals the oligomeric state of functional TNFα-induced plasma membrane TNFR1 clusters in cells. Sci. Signal 13, eaax5647 (2020).
Krippner-Heidenreich, A. et al. Control of receptor-induced signaling complex formation by the kinetics of ligand/receptor interaction. J. Biol. Chem. 277, 44155–44163 (2002).
Chan, F. K. et al. A domain in TNF receptors that mediates ligand-independent receptor assembly and signaling. Science 288, 2351–2354 (2000).
Weinelt, N. et al. Quantitative single-molecule imaging of TNFR1 reveals zafirlukast as antagonist of TNFR1 clustering and TNFα-induced NF-ĸB signaling. J. Leukoc. Biol. 109, 363–371 (2021).
Prada, J. P. et al. A systems-biology model of the tumor necrosis factor (TNF) interactions with TNF receptor 1 and 2. Bioinformatics 37, 669–676 (2021).
Kupka, S., Reichert, M., Draber, P. & Walczak, H. Formation and removal of poly-ubiquitin chains in the regulation of tumor necrosis factor-induced gene activation and cell death. FEBS J. 283, 2626–2639 (2016).
Lafont, E. et al. TBK1 and IKKε prevent TNF-induced cell death by RIPK1 phosphorylation. Nat. Cell Biol. 20, 1389–1399 (2018).
Zhang, Q., Lenardo, M. J. & Baltimore, D. 30 years of NF-κB: a blossoming of relevance to human pathobiology. Cell 168, 37–57 (2017).
Sabio, G. & Davis, R. J. TNF and MAP kinase signalling pathways. Semin. Immunol. 26, 237–245 (2014).
Vandenabeele, P., Declercq, W., Van Herreweghe, F. & Vanden Berghe, T. The role of the kinases RIP1 and RIP3 in TNF-induced necrosis. Sci. Signal. 3, re4 (2010).
Wajant, H. & Siegmund, D. TNFR1 and TNFR2 in the control of the life and death balance of macrophages. Front. Cell Dev. Biol. 7, 91 (2019).
Yang, X. D. & Sun, S. C. Targeting signaling factors for degradation, an emerging mechanism for TRAF functions. Immunol. Rev. 266, 56–71 (2015).
Borghi, A. et al. The E3 ubiquitin ligases HOIP and cIAP1 are recruited to the TNFR2 signaling complex and mediate TNFR2-induced canonical NF-κB signaling. Biochem. Pharmacol. 153, 292–298 (2018).
Pan, S. et al. Etk/Bmx as a tumor necrosis factor receptor type 2-specific kinase: role in endothelial cell migration and angiogenesis. Mol. Cell Biol. 22, 7512–7523 (2002).
Zhang, R. et al. Etk/Bmx transactivates vascular endothelial growth factor 2 and recruits phosphatidylinositol 3-kinase to mediate the tumor necrosis factor-induced angiogenic pathway. J. Biol. Chem. 278, 51267–51276 (2003).
Pegoretti, V., Baron, W., Laman, J. D. & Eisel, U. L. M. Selective modulation of TNF-TNFRs signaling: insights for multiple sclerosis treatment. Front. Immunol. 9, 925 (2018).
Ji, W. et al. Both internalization and AIP1 association are required for tumor necrosis factor receptor 2-mediated JNK signaling. Arterioscler. Thromb. Vasc. Biol. 32, 2271–2279 (2012).
Calmon-Hamaty, F., Combe, B., Hahne, M. & Morel, J. Lymphotoxin α stimulates proliferation and pro-inflammatory cytokine secretion of rheumatoid arthritis synovial fibroblasts. Cytokine 53, 207–214 (2011).
Robak, T., Gladalska, A. & Stepień, H. The tumour necrosis factor family of receptors/ligands in the serum of patients with rheumatoid arthritis. Eur. Cytokine Netw. 9, 145–154 (1998).
Kennedy, W. P. et al. Efficacy and safety of pateclizumab (anti-lymphotoxin-α) compared to adalimumab in rheumatoid arthritis: a head-to-head phase 2 randomized controlled study (The ALTARA Study). Arthritis Res. Ther. 16, 467 (2014).
Markey, K. A. et al. Soluble lymphotoxin is an important effector molecule in GVHD and GVL. Blood 115, 122–132 (2010).
Bingham, C. O. 3rd et al. Effectiveness and safety of etanercept in subjects with RA who have failed infliximab therapy: 16-week, open-label, observational study. Curr. Med. Res. Opin. 25, 1131–1142 (2009).
Buch, M. H. et al. Therapy of patients with rheumatoid arthritis: outcome of infliximab failures switched to etanercept. Arthritis Rheum. 57, 448–453 (2007).
Buch, M. H. et al. C-reactive protein as a predictor of infliximab treatment outcome in patients with rheumatoid arthritis: defining subtypes of nonresponse and subsequent response to etanercept. Arthritis Rheum. 52, 42–48 (2005).
Di Poi, E. et al. Switching to etanercept in patients with rheumatoid arthritis with no response to infliximab. Clin. Exp. Rheumatol. 25, 85–87 (2007).
Fleischmann, R. et al. Infliximab efficacy in rheumatoid arthritis after an inadequate response to etanercept or adalimumab: results of a target-driven active switch study. Curr. Med. Res. Opin. 30, 2139–2149 (2014).
Arora, T. et al. Differences in binding and effector functions between classes of TNF antagonists. Cytokine 45, 124–131 (2009).
Mitoma, H. et al. Mechanisms for cytotoxic effects of anti-tumor necrosis factor agents on transmembrane tumor necrosis factor alpha-expressing cells: comparison among infliximab, etanercept, and adalimumab. Arthritis Rheum. 58, 1248–1257 (2008).
Ueda, N. et al. The cytotoxic effects of certolizumab pegol and golimumab mediated by transmembrane tumor necrosis factor α. Inflamm. Bowel Dis. 19, 1224–1231 (2013).
Mori, L., Iselin, S., De Libero, G. & Lesslauer, W. Attenuation of collagen-induced arthritis in 55-kDa TNF receptor type 1 (TNFR1)-IgG1-treated and TNFR1-deficient mice. J. Immunol. 157, 3178–3182 (1996).
Eugster, H. P. et al. Severity of symptoms and demyelination in MOG-induced EAE depends on TNFR1. Eur. J. Immunol. 29, 626–632 (1999).
Grine, L., Dejager, L., Libert, C. & Vandenbroucke, R. E. Dual inhibition of TNFR1 and IFNAR1 in imiquimod-induced psoriasiform skin inflammation in mice. J. Immunol. 194, 5094–5102 (2015).
Guinea-Viniegra, J. et al. TNF α αshedding and epidermal inflammation are controlled by Jun proteins. Genes. Dev. 23, 2663–2674 (2009).
He, P. et al. Deletion of tumor necrosis factor death receptor inhibits amyloid beta generation and prevents learning and memory deficits in Alzheimer’s mice. J. Cell Biol. 178, 829–841 (2007).
Fontaine, V. et al. Neurodegenerative and neuroprotective effects of tumor Necrosis factor (TNF) in retinal ischemia: opposite roles of TNF receptor 1 and TNF receptor 2. J. Neurosci. 22, Rc216 (2002).
Duerrschmid, C. et al. TNF receptor 1 signaling is critically involved in mediating angiotensin-II-induced cardiac fibrosis. J. Mol. Cell Cardiol. 57, 59–67 (2013).
Monden, Y. et al. Tumor necrosis factor-α is toxic via receptor 1 and protective via receptor 2 in a murine model of myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 293, H743–H753 (2007).
Acton, R. D. et al. Differential sensitivity to Escherichia coli infection in mice lacking tumor necrosis factor p55 or interleukin-1 p80 receptors. Arch. Surg. 131, 1216–1221 (1996).
Amar, S. et al. Tumor necrosis factor (TNF)-induced cutaneous necrosis is mediated by TNF receptor 1. J. Inflamm. 47, 180–189 (1995).
Nowak, M. et al. LPS-induced liver injury in D-galactosamine-sensitized mice requires secreted TNF-α and the TNF-p55 receptor. Am. J. Physiol. Regul. Integr. Comp. Physiol. 278, R1202–R1209 (2000).
Shimizu, S. et al. Liver injury induced by lipopolysaccharide is mediated by TNFR-1 but not by TNFR-2 or Fas in mice. Hepatol. Res. 31, 136–142 (2005).
Moulin, M. et al. IAPs limit activation of RIP kinases by TNF receptor 1 during development. EMBO J. 31, 1679–1691 (2012).
Newton, K. et al. Cleavage of RIPK1 by caspase-8 is crucial for limiting apoptosis and necroptosis. Nature 574, 428–431 (2019).
Piao, X. et al. c-FLIP maintains tissue homeostasis by preventing apoptosis and programmed necrosis. Sci. Signal. 5, ra93 (2012).
Rosenfeld, M. E., Prichard, L., Shiojiri, N. & Fausto, N. Prevention of hepatic apoptosis and embryonic lethality in RelA/TNFR-1 double knockout mice. Am. J. Pathol. 156, 997–1007 (2000).
Steed, P. M. et al. Inactivation of TNF signaling by rationally designed dominant-negative TNF variants. Science 301, 1895–1898 (2003).
Zalevsky, J. et al. Dominant-negative inhibitors of soluble TNF attenuate experimental arthritis without suppressing innate immunity to infection. J. Immunol. 179, 1872–1883 (2007).
Barnum, C. J. et al. Peripheral administration of the selective inhibitor of soluble tumor necrosis factor (TNF) XPro®1595 attenuates nigral cell loss and glial activation in 6-OHDA hemiparkinsonian rats. J. Parkinsons Dis. 4, 349–360 (2014).
Brambilla, R. et al. Inhibition of soluble tumour necrosis factor is therapeutic in experimental autoimmune encephalomyelitis and promotes axon preservation and remyelination. Brain 134, 2736–2754 (2011).
Cavanagh, C. et al. Inhibiting tumor necrosis factor-α before amyloidosis prevents synaptic deficits in an Alzheimer’s disease model. Neurobiol. Aging 47, 41–49 (2016).
Clausen, B. H. et al. Systemically administered anti-TNF therapy ameliorates functional outcomes after focal cerebral ischemia. J. Neuroinflammation 11, 203 (2014).
Cueva Vargas, J. L. et al. Soluble tumor necrosis factor α promotes retinal ganglion cell death in glaucoma via calcium-permeable AMPA receptor activation. J. Neurosci. 35, 12088–12102 (2015).
Griepke, S. et al. Selective inhibition of soluble tumor necrosis factor signaling reduces abdominal aortic aneurysm progression. Front. Cardiovasc. Med. 9, 942342 (2022).
Hsiao, H. Y. et al. Inhibition of soluble tumor necrosis factor is therapeutic in Huntington’s disease. Hum. Mol. Genet. 23, 4328–4344 (2014).
Huang, S. C., Lee, C. T. & Chung, B. C. Tumor necrosis factor suppresses NR5A2 activity and intestinal glucocorticoid synthesis to sustain chronic colitis. Sci. Signal. 7, ra20 (2014).
Karamita, M. et al. Therapeutic inhibition of soluble brain TNF promotes remyelination by increasing myelin phagocytosis by microglia. JCI Insight 2, e87455 (2017).
Maillet, I. et al. Allergic lung inflammation is mediated by soluble tumor necrosis factor (TNF) and attenuated by dominant-negative TNF biologics. Am. J. Respir. Cell Mol. Biol. 45, 731–739 (2011).
Novrup, H. G. et al. Central but not systemic administration of XPro1595 is therapeutic following moderate spinal cord injury in mice. J. Neuroinflammation 11, 159 (2014).
Olleros, M. L. et al. Dominant-negative tumor necrosis factor protects from Mycobacterium bovis bacillus Calmette Guérin (BCG) and endotoxin-induced liver injury without compromising host immunity to BCG and Mycobacterium tuberculosis. J. Infect. Dis. 199, 1053–1063 (2009).
Randhi, R., Damon, M. & Dixon, K. J. Selective inhibition of soluble TNF using XPro1595 relieves pain and attenuates cerulein-induced pathology in mice. BMC Gastroenterol. 21, 243 (2021).
Sobo-Vujanovic, A. et al. Inhibition of soluble tumor necrosis factor prevents chemically induced carcinogenesis in mice. Cancer Immunol. Res. 4, 441–451 (2016).
Chou, R. C., Kane, M., Ghimire, S., Gautam, S. & Gui, J. Treatment for rheumatoid arthritis and risk of Alzheimer’s disease: a nested case-control analysis. CNS Drugs 30, 1111–1120 (2016).
Zhou, M., Xu, R., Kaelber, D. C. & Gurney, M. E. Tumor Necrosis Factor (TNF) blocking agents are associated with lower risk for Alzheimer’s disease in patients with rheumatoid arthritis and psoriasis. PLoS One 15, e0229819 (2020).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT05522387 (2022).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT05318976 (2023).
Shibata, H. et al. Creation and X-ray structure analysis of the tumor necrosis factor receptor-1-selective mutant of a tumor necrosis factor-α antagonist. J. Biol. Chem. 283, 998–1007 (2008).
Nomura, T. et al. Therapeutic effect of PEGylated TNFR1-selective antagonistic mutant TNF in experimental autoimmune encephalomyelitis mice. J. Control. Rel. 149, 8–14 (2011).
Shibata, H. et al. The treatment of established murine collagen-induced arthritis with a TNFR1-selective antagonistic mutant TNF. Biomaterials 30, 6638–6647 (2009).
Thoma, B., Grell, M., Pfizenmaier, K. & Scheurich, P. Identification of a 60-kD tumor necrosis factor (TNF) receptor as the major signal transducing component in TNF responses. J. Exp. Med. 172, 1019–1023 (1990).
Richter, F., Seifert, O., Herrmann, A., Pfizenmaier, K. & Kontermann, R. E. Improved monovalent TNF receptor 1-selective inhibitor with novel heterodimerizing Fc. MAbs 11, 653–665 (2019).
Zettlitz, K. A. et al. ATROSAB, a humanized antagonistic anti-tumor necrosis factor receptor one-specific antibody. MAbs 2, 639–647 (2010).
Richter, F. et al. The TNFR1 antagonist atrosimab is therapeutic in mouse models of acute and chronic inflammation. Front. Immunol. 12, 705485 (2021).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT04650126 (2022).
US National Library of Medicine. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT01476046 (2017).
Holland, M. C. et al. Autoantibodies to variable heavy (VH) chain Ig sequences in humans impact the safety and clinical pharmacology of a VH domain antibody antagonist of TNF-α receptor 1. J. Clin. Immunol. 33, 1192–1203 (2013).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT01818024 (2017).
Cordy, J. C. et al. Specificity of human anti-variable heavy (VH) chain autoantibodies and impact on the design and clinical testing of a VH domain antibody antagonist of tumour necrosis factor-α receptor 1. Clin. Exp. Immunol. 182, 139–148 (2015).
US National Library of Medicine. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT01587807 (2019).
Proudfoot, A. et al. Novel anti-tumour necrosis factor receptor-1 (TNFR1) domain antibody prevents pulmonary inflammation in experimental acute lung injury. Thorax 73, 723–730 (2018).
Steeland, S. et al. Generation and characterization of small single domain antibodies inhibiting human tumor necrosis factor receptor 1. J. Biol. Chem. 290, 4022–4037 (2015).
Steeland, S. et al. TNFR1 inhibition with a nanobody protects against EAE development in mice. Sci. Rep. 7, 13646 (2017).
Deng, G. M., Liu, L. & Tsokos, G. C. Targeted tumor necrosis factor receptor I preligand assembly domain improves skin lesions in MRL/lpr mice. Arthritis Rheum. 62, 2424–2431 (2010).
Deng, G. M., Zheng, L., Chan, F. K. & Lenardo, M. Amelioration of inflammatory arthritis by targeting the pre-ligand assembly domain of tumor necrosis factor receptors. Nat. Med. 11, 1066–1072 (2005).
Wang, Y. L. et al. Targeting pre-ligand assembly domain of TNFR1 ameliorates autoimmune diseases — an unrevealed role in downregulation of Th17 cells. J. Autoimmun. 37, 160–170 (2011).
Murali, R. et al. Disabling TNF receptor signaling by induced conformational perturbation of tryptophan-107. Proc. Natl Acad. Sci. USA 102, 10970–10975 (2005).
Rowe, R. K. et al. Novel TNF receptor-1 inhibitors identified as potential therapeutic candidates for traumatic brain injury. J. Neuroinflammation 15, 154 (2018).
Chen, S. et al. Discovery of novel ligands for TNF-α and TNF Receptor-1 through structure-based virtual screening and biological assay. J. Chem. Inf. Model. 57, 1101–1111 (2017).
Lo, C. H. et al. Noncompetitive inhibitors of TNFR1 probe conformational activation states. Sci. Signal. 12, eaav5637 (2019).
Lo, C. H. et al. An innovative high-throughput screening approach for discovery of small molecules that inhibit TNF receptors. SLAS Discov. 22, 950–961 (2017).
de Kivit, S. et al. Stable human regulatory T cells switch to glycolysis following TNF receptor 2 costimulation. Nat. Metab. 2, 1046–1061 (2020).
Lubrano di Ricco, M. et al. Tumor necrosis factor receptor family costimulation increases regulatory T-cell activation and function via NF-κB. Eur. J. Immunol. 50, 972–985 (2020).
Santinon, F. et al. Involvement of tumor necrosis factor receptor type II in FoxP3 stability and as a marker of Treg cells specifically expanded by anti-tumor necrosis factor treatments in rheumatoid arthritis. Arthritis Rheumatol. 72, 576–587 (2020).
Tseng, W. Y. et al. TNF receptor 2 signaling prevents DNA methylation at the Foxp3 promoter and prevents pathogenic conversion of regulatory T cells. Proc. Natl Acad. Sci. USA 116, 21666–21672 (2019).
Hövelmeyer, N., Schmidt-Supprian, M. & Ohnmacht, C. NF-κB in control of regulatory T cell development, identity, and function. J. Mol. Med. 100, 985–995 (2022).
Claus, C. et al. CD27 signaling increases the frequency of regulatory T cells and promotes tumor growth. Cancer Res. 72, 3664–3676 (2012).
Mahmud, S. A. et al. Costimulation via the tumor-necrosis factor receptor superfamily couples TCR signal strength to the thymic differentiation of regulatory T cells. Nat. Immunol. 15, 473–481 (2014).
Hu, X. et al. Transmembrane TNF-α promotes suppressive activities of myeloid-derived suppressor cells via TNFR2. J. Immunol. 192, 1320–1331 (2014).
Zhao, X. et al. TNF signaling drives myeloid-derived suppressor cell accumulation. J. Clin. Invest. 122, 4094–4104 (2012).
Beldi, G. et al. TNFR2 is a crucial hub controlling mesenchymal stem cell biological and functional properties. Front. Cell Dev. Biol. 8, 596831 (2020).
Beldi, G. et al. TNFα/TNFR2 signaling pathway: an active immune checkpoint for mesenchymal stem cell immunoregulatory function. Stem Cell Res. Ther. 11, 281 (2020).
Chopra, M. et al. Exogenous TNFR2 activation protects from acute GvHD via host T reg cell expansion. J. Exp. Med. 213, 1881–1900 (2016).
Dong, Y. et al. Essential protective role of tumor necrosis factor receptor 2 in neurodegeneration. Proc. Natl Acad. Sci. USA 113, 12304–12309 (2016).
Fischer, R. et al. Novel strategies to mimic transmembrane tumor necrosis factor-dependent activation of tumor necrosis factor receptor 2. Sci. Rep. 7, 6607 (2017).
Inoue, M. et al. Structural optimization of a TNFR1-selective antagonistic TNFα mutant to create new-modality TNF-regulating biologics. J. Biol. Chem. 295, 9379–9391 (2020).
Vargas, J. G. et al. A TNFR2-specific TNF fusion protein with improved in vivo activity. Front. Immunol. 13, 888274 (2022).
Loetscher, H., Stueber, D., Banner, D., Mackay, F. & Lesslauer, W. Human tumor necrosis factor alpha (TNFα) mutants with exclusive specificity for the 55-kDa or 75-kDa TNF receptors. J. Biol. Chem. 268, 26350–26357 (1993).
Hutt, M. et al. Superior properties of Fc-comprising scTRAIL fusion proteins. Mol. Cancer Ther. 16, 2792–2802 (2017).
Siegemund, M. et al. IgG-single-chain TRAIL fusion proteins for tumour therapy. Sci. Rep. 8, 7808 (2018).
Beilhack, A., Kums, J., Nelke, J. & Wajant, H. Tumor necrosis factor (TNF) receptor superfamily (TNFRSF) receptor-activating antibody fusion proteins with FcγR-independent agonistic activity. (TNFRSF receptor-activating antibody fusion proteins with FcγR-independent agonistic activity; TRAAFFIAA). World patent WO 2019/129644 (2019).
Li, M., Zhang, X., Bai, X. & Liang, T. Targeting TNFR2: a novel breakthrough in the treatment of cancer. Front. Oncol. 12, 862154 (2022).
US National Library of Medicine. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT04752826 (2023).
US National Library of Medicine. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT05569057 (2022).
US National Library of Medicine. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT05238883 (2023).
National Cancer Institute. Anti-TNFR2 Monoclonal Antibody SIM1811-03 (Code C191777). https://ncit.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCI_Thesaurus&ns=ncit&code=C191777 (2023).
Linda Mårtensson, K. C. et al. Proceedings of the Annual Meeting of the American Association for Cancer Research 2020 (AACR, 2020).
Shuo Wei, G. Y. et al. Discovery and characterization of novel TNFR2 antibodies to modulate T cell activities in immunosuppressive environment. Proc. Annu. Meeting Am. Assoc. Cancer Res. 2020. 80, 2282 (AACR, 2020).
Drutskaya, M. S., Efimov, G. A., Kruglov, A. A. & Nedospasov, S. A. Can we design a better anti-cytokine therapy? J. Leukoc. Biol. 102, 783–790 (2017).
Efimov, G. A. et al. Cell-type-restricted anti-cytokine therapy: TNF inhibition from one pathogenic source. Proc. Natl Acad. Sci. USA 113, 3006–3011 (2016).
The Antibody Society. Therapeutic Monoclonal Antibodies Approved or in Review in the EU or US https://www.antibodysociety.org/resources/approved-antibodies (6 July 2023).
Ishiwatari-Ogata, C. et al. Ozoralizumab, a humanized anti-TNFα NANOBODY® compound, exhibits efficacy not only at the onset of arthritis in a human TNF transgenic mouse but also during secondary failure of administration of an anti-TNFα IgG. Front. Immunol. 13, 853008 (2022).
Berns, M. & Hommes, D. W. Anti-TNF-α therapies for the treatment of Crohn’s disease: the past, present and future. Expert. Opin. Investig. Drugs 25, 129–143 (2016).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT00808262 (2011).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT01040715 (2014).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT01291810 (2014).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT01911234 (2019).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT01759056 (2014).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT02221037 (2020).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT03943264 (2023).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT04370236 (2022).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT05321498 (2022).
Inoue, M. et al. Characterization of a TNFR2-selective agonistic TNF-α mutant and its derivatives as an optimal regulatory T cell expander. J. Immunol. 206, 1740–1751 (2021).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT05781386 (2023).
Schmid, T. et al. Chronic inflammation increases the sensitivity of mouse Treg for TNFR2 costimulation. Front. Immunol. 8, 1471 (2017).
Lamontain, V. et al. Stimulation of TNF receptor type 2 expands regulatory T cells and ameliorates established collagen-induced arthritis in mice. Cell Mol. Immunol. 16, 65–74 (2019).
Gouweleeuw, L. et al. Effects of selective TNFR1 inhibition or TNFR2 stimulation, compared to non-selective TNF inhibition, on (neuro)inflammation and behavior after myocardial infarction in male mice. Brain Behav. Immun. 93, 156–171 (2021).
Ronin, E. et al. Tissue-restricted control of established central nervous system autoimmunity by TNF receptor 2-expressing Treg cells. Proc. Natl Acad. Sci. USA 118, e2014043118 (2021).
Ortí-Casañ, N. et al. A TNF receptor 2 agonist ameliorates neuropathology and improves cognition in an Alzheimer’s disease mouse model. Proc. Natl Acad. Sci. USA 119, e2201137119 (2022).
Fischer, R. et al. TNFR2 promotes Treg-mediated recovery from neuropathic pain across sexes. Proc. Natl Acad. Sci. USA 116, 17045–17050 (2019).
Gerald, M. J. et al. Continuous infusion of an agonist of the tumor necrosis factor receptor 2 in the spinal cord improves recovery after traumatic contusive injury. CNS Neurosci. Ther. 25, 884–893 (2019).
Fischer, R. et al. Selective activation of tumor necrosis factor receptor II induces antiinflammatory responses and alleviates experimental arthritis. Arthritis Rheumatol. 70, 722–735 (2018).
Acknowledgements
The research work of the authors is supported by the Deutsche Forschungsgemeinschaft grants DFG Si 1128/6–1 (to D.S.) and DFG WA 1025/33–1 and 324392634–TR221 (to H.W.).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
H.W. declares that he received consultancy fees from Dualyx NV regarding the targeting of Treg cells via TNF receptor 2 (TNFR2). D.S. declares no competing interests. The University of Würzburg has filed patent applications for “Novel TNFR2 binding molecules” and “Tumour necrosis factor (TNF) receptor superfamily (TNFRSF) receptor-activating antibody fusion proteins with FcγR-independent agonistic activity (TRAAFFIA)”, with H.W. as one of the inventors.
Peer review
Peer review information
Nature Reviews Rheumatology thanks D. Wallach and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Antibody-dependent cellular cytotoxicity
-
An antibody effector function by which antibody-opsonized cells are recognized and killed or lysed by immune cells that express appropriate Fcγ receptors (such as natural killers).
- Antibody-dependent cellular phagocytosis
-
An antibody effector function by which antibody-opsonized cells are recognized and phagocytosed by macrophages.
- Association rate constant
-
This constant describes the kinetics by which two components (such as a ligand and receptor) form a complex.
- Auto-affinity
-
Affinity of proteins for self-assembly.
- Complement-dependent cytotoxicity
-
An antibody effector function triggered by cell-bound IgG and IgM antibodies, which results in cell killing or lysis by the so-called membrane attack complex of the complement system.
- Dissociation rate constant
-
This constant describes the kinetics of the dissociation of a two-component complex (such as a ligand–receptor complex) into the individual components.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Siegmund, D., Wajant, H. TNF and TNF receptors as therapeutic targets for rheumatic diseases and beyond. Nat Rev Rheumatol 19, 576–591 (2023). https://doi.org/10.1038/s41584-023-01002-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-023-01002-7
This article is cited by
-
25 years of biologic DMARDs in rheumatology
Nature Reviews Rheumatology (2023)
-
Atsttrin regulates osteoblastogenesis and osteoclastogenesis through the TNFR pathway
Communications Biology (2023)