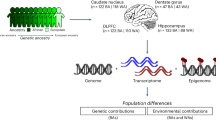
Abstract
Addictions are heritable and unfold dynamically across the lifespan. One prominent neurobiological theory proposes that substance-induced changes in neural circuitry promote the progression of addiction. Genome-wide association studies have begun to characterize the polygenic architecture undergirding addiction liability and revealed that genetic loci associated with risk can be divided into those associated with a general broad-spectrum liability to addiction and those associated with drug-specific addiction risk. In this Perspective, we integrate these genomic findings with our current understanding of the neurobiology of addiction to propose a new Genetically Informed Neurobiology of Addiction (GINA) model.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Peacock, A. et al. Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction 113, 1905–1926 (2018).
Volkow, N. D., Gordon, J. A. & Koob, G. F. Choosing appropriate language to reduce the stigma around mental illness and substance use disorders. Neuropsychopharmacology 46, 2230–2232 (2021).
Agrawal, A. et al. The genetics of addiction-a translational perspective. Transl. Psychiatry 2, e140 (2012).
Kendler, K. S. et al. Recent advances in the genetic epidemiology and molecular genetics of substance use disorders. Nat. Neurosci. 15, 181–189 (2012).
Gelernter, J. & Polimanti, R. Genetics of substance use disorders in the era of big data. Nat. Rev. Genet. 22, 712–729 (2021).
Koob, G. F. & Volkow, N. D. Neurocircuitry of addiction. Neuropsychopharmacology 35, 217–238 (2010).
Volkow, N. D., Koob, G. F. & McLellan, A. T. Neurobiologic advances from the brain disease model of addiction. N. Engl. J. Med. 374, 363–371 (2016).
Koob, G. F. & Volkow, N. D. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry 3, 760–773 (2016).
Volkow, N. D. & Boyle, M. Neuroscience of addiction: relevance to prevention and treatment. Am. J. Psychiatry 175, 729–740 (2018).
Koob, G. F. Drug addiction: hyperkatifeia/negative reinforcement as a framework for medications development. Pharmacol. Rev. 73, 163–201 (2021).
Haber, S. N. Corticostriatal circuitry. Dialogues Clin. Neurosci. 18, 7–21 (2016).
Janak, P. H. & Tye, K. M. From circuits to behaviour in the amygdala. Nature 517, 284–292 (2015).
Volkow, N. D., Fowler, J. S., Wang, G. J., Baler, R. & Telang, F. Imaging dopamine’s role in drug abuse and addiction. Neuropharmacology 56, 3–8 (2009).
Volkow, N. D., Michaelides, M. & Baler, R. The neuroscience of drug reward and addiction. Physiol. Rev. 99, 2115–2140 (2019).
Schultz, W. Dopamine reward prediction error coding. Dialogues Clin. Neurosci. 18, 23–32 (2016).
Keiflin, R. & Janak, P. H. Dopamine prediction errors in reward learning and addiction: from theory to neural circuitry. Neuron 88, 247–263 (2015).
Volkow, N. D. & Morales, M. The brain on drugs: from reward to addiction. Cell 162, 712–725 (2015).
Filbey, F. M. et al. fMRI study of neural sensitization to hedonic stimuli in long-term, daily cannabis users. Hum. Brain Mapp. 37, 3431–3443 (2016).
Perry, J. L. & Carroll, M. E. The role of impulsive behavior in drug abuse. Psychopharmacology 200, 1–26 (2008).
Willuhn, I., Burgeno, L. M., Groblewski, P. A. & Phillips, P. E. Excessive cocaine use results from decreased phasic dopamine signaling in the striatum. Nat. Neurosci. 17, 704–709 (2014).
Koob, G. F. Negative reinforcement in drug addiction: the darkness within. Curr. Opin. Neurobiol. 23, 559–563 (2013).
Koob, G. F. & Le Moal, M. Plasticity of reward neurocircuitry and the ‘dark side’ of drug addiction. Nat. Neurosci. 8, 1442–1444 (2005).
Ahmed, S. H. & Koob, G. F. Transition from moderate to excessive drug intake: change in hedonic set point. Science 282, 298–300 (1998).
Martz, M. E. et al. Association of marijuana use with blunted nucleus accumbens response to reward anticipation. JAMA Psychiatry 73, 838–844 (2016).
Balodis, I. M. & Potenza, M. N. Anticipatory reward processing in addicted populations: a focus on the monetary incentive delay task. Biol. Psychiatry 77, 434–444 (2015).
Zorrilla, E. P., Logrip, M. L. & Koob, G. F. Corticotropin releasing factor: a key role in the neurobiology of addiction. Front. Neuroendocrinol. 35, 234–244 (2014).
Naqvi, N. H., Gaznick, N., Tranel, D. & Bechara, A. The insula: a critical neural substrate for craving and drug seeking under conflict and risk. Ann. N. Y. Acad. Sci. 1316, 53–70 (2014).
McClure, S. M. & Bickel, W. K. A dual-systems perspective on addiction: contributions from neuroimaging and cognitive training. Ann. N. Y. Acad. Sci. 1327, 62–78 (2014).
Heather, N. The concept of akrasia as the foundation for a dual systems theory of addiction. Behav. Brain Res. 390, 112666 (2020).
Cadet, J. L. & Bisagno, V. Neuropsychological consequences of chronic drug use: relevance to treatment approaches. Front. Psychiatry 6, 189 (2015).
Brockett, A. T., Pribut, H. J., Vazquez, D. & Roesch, M. R. The impact of drugs of abuse on executive function: characterizing long-term changes in neural correlates following chronic drug exposure and withdrawal in rats. Learn. Mem. 25, 461–473 (2018).
Gerdeman, G. L., Partridge, J. G., Lupica, C. R. & Lovinger, D. M. It could be habit forming: drugs of abuse and striatal synaptic plasticity. Trends Neurosci. 26, 184–192 (2003).
Grant, B. F. et al. Epidemiology of DSM-5 drug use disorder: results from the national epidemiologic survey on alcohol and related conditions-III. JAMA Psychiatry 73, 39–47 (2016).
Sinha, R. New findings on biological factors predicting addiction relapse vulnerability. Curr. Psychiatry Rep. 13, 398–405 (2011).
Stinson, F. S. et al. Comorbidity between DSM-IV alcohol and specific drug use disorders in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Drug Alcohol Depend. 80, 105–116 (2005).
Mackey, S. et al. Mega-analysis of gray matter volume in substance dependence: general and substance-specific regional effects. Am. J. Psychiatry 176, 119–128 (2019).
Hatoum, A. S. et al. Multivariate genome-wide association meta-analysis of over 1 million subjects identifies loci underlying multiple substance use disorders. medRxiv https://doi.org/10.1101/2022.01.06.22268753v1 (2022).
Schoeler, T. et al. Novel biological insights into the common heritable liability to substance involvement: a multivariate genome-wide association study. Biol. Psychiatry https://doi.org/10.1016/j.biopsych.2022.07.027 (2022).
Zucker, R. A. et al. Assessment of culture and environment in the Adolescent Brain and Cognitive Development Study: Rationale, description of measures, and early data. Dev. Cogn. Neurosci. 32, 107–120 (2018).
Chassin, L., Sher, K. J., Hussong, A. & Curran, P. The developmental psychopathology of alcohol use and alcohol disorders: research achievements and future directions. Dev. Psychopathol. 25, 1567–1584 (2013).
Casey, B. J. & Jones, R. M. Neurobiology of the adolescent brain and behavior: implications for substance use disorders. J. Am. Acad. Child Adolesc. Psychiatry 49, 1189–1201 (2010).
Rapuano, K. M. et al. Behavioral and brain signatures of substance use vulnerability in childhood. Dev. Cogn. Neurosci. 46, 100878 (2020).
Deak, J. D. & Johnson, E. C. Genetics of substance use disorders: a review. Psychol. Med. 51, 2189–2200 (2021).
Johnson, E. C. et al. No evidence that schizophrenia candidate genes are more associated with schizophrenia than noncandidate genes. Biol. Psychiatry 82, 702–708 (2017).
Border, R. et al. No support for historical candidate gene or candidate gene-by-interaction hypotheses for major depression across multiple large samples. Am. J. Psychiatry 176, 376–387 (2019).
Johnson, E. C. et al. A large-scale genome-wide association study meta-analysis of cannabis use disorder. Lancet Psychiatry 7, 1032–1045 (2020).
Liu, M. et al. Association studies of up to 1.2 million individuals yield new insights into the genetic etiology of tobacco and alcohol use. Nat. Genet. 51, 237–244 (2019).
Polimanti, R. et al. Leveraging genome-wide data to investigate differences between opioid use vs. opioid dependence in 41,176 individuals from the Psychiatric Genomics Consortium. Mol. Psychiatry 25, 1673–1687 (2020).
Walters, R. K. et al. Transancestral GWAS of alcohol dependence reveals common genetic underpinnings with psychiatric disorders. Nat. Neurosci. 21, 1656–1669 (2018).
Zhou, H. et al. Genome-wide meta-analysis of problematic alcohol use in 435,563 individuals yields insights into biology and relationships with other traits. Nat. Neurosci. 23, 809–818 (2020).
Pasman, J. A. et al. GWAS of lifetime cannabis use reveals new risk loci, genetic overlap with psychiatric traits, and a causal influence of schizophrenia. Nat. Neurosci. 21, 1161–1170 (2018).
Xu, K. et al. Genome-wide association study of smoking trajectory and meta-analysis of smoking status in 842,000 individuals. Nat. Commun. 11, 5302 (2020).
Zhou, H. et al. Association of OPRM1 functional coding variant with opioid use disorder: a genome-wide association study. JAMA Psychiatry 77, 1072–1080 (2020).
Kranzler, H. R. et al. Genome-wide association study of alcohol consumption and use disorder in 274,424 individuals from multiple populations. Nat. Commun. 10, 1499 (2019).
Gelernter, J. et al. Genome-wide association study of maximum habitual alcohol intake in>140,000 U.S. European and African American veterans yields novel risk loci. Biol. Psychiatry 86, 365–376 (2019).
Brazel, D. M. et al. Exome Chip meta-analysis fine maps causal variants and elucidates the genetic architecture of rare coding variants in smoking and alcohol use. Biol. Psychiatry 85, 946–955 (2019).
Jang, S. K. et al. Rare genetic variants explain missing heritability in smoking. Nat. Hum. Behav. https://doi.org/10.1038/s41562-022-01408-5 (2022).
Sullivan, P. F. & Geschwind, D. H. Defining the genetic, genomic, cellular, and diagnostic architectures of psychiatric disorders. Cell 177, 162–183 (2019).
Yang, J. et al. Genetic variance estimation with imputed variants finds negligible missing heritability for human height and body mass index. Nat. Genet. 47, 1114–1120 (2015).
Mallard, T. T. et al. Item-level genome-wide association study of the alcohol use disorders identification test in three population-based cohorts. Am. J. Psychiatry 179, 58–70 (2022).
Sanchez-Roige, S., Palmer, A. A. & Clarke, T. K. Recent efforts to dissect the genetic basis of alcohol use and abuse. Biol. Psychiatry 87, 609–618 (2020).
Marees, A. T. et al. Genetic correlates of socio-economic status influence the pattern of shared heritability across mental health traits. Nat. Hum. Behav. 5, 1065–1073 (2021).
Hatoum, A. S. et al. The addiction risk factor: a unitary genetic vulnerability characterizes substance use disorders and their associations with common correlates. Neuropsychopharmacology 47, 1739–1745 (2022).
Karlsson Linner, R. et al. Multivariate analysis of 1.5 million people identifies genetic associations with traits related to self-regulation and addiction. Nat. Neurosci. 24, 1367–1376 (2021).
Liu, X., Zhong, P., Vickstrom, C., Li, Y. & Liu, Q. S. PDE4 inhibition restores the balance between excitation and inhibition in VTA dopamine neurons disrupted by repeated in vivo cocaine exposure. Neuropsychopharmacology 42, 1991–1999 (2017).
Tibbo, A. J. & Baillie, G. S. Phosphodiesterase 4B: master regulator of brain signaling. Cells 9, 1254 (2020).
Kuroiwa, M. et al. Phosphodiesterase 4 inhibition enhances the dopamine D1 receptor/PKA/DARPP-32 signaling cascade in frontal cortex. Psychopharmacology 219, 1065–1079 (2012).
Arends, R. M. et al. Associations between the CADM2 gene, substance use, risky sexual behavior, and self-control: A phenome-wide association study. Addict. Biol. 26, e13015 (2021).
Morris, J. et al. Genetic variation in CADM2 as a link between psychological traits and obesity. Sci. Rep. 9, 7339 (2019).
Sanchez-Roige, S. et al. CADM2 is implicated in impulsive personality traits by genome- and phenome-wide association studies in humans, with further support from studies of Cadm2 mutant mice. medRxiv https://doi.org/10.1101/2022.01.29.22270095v3 (2022).
Gerring, Z. F., Vargas, A. M., Gamazon, E. R. & Derks, E. M. An integrative systems-based analysis of substance use: eQTL-informed gene-based tests, gene networks, and biological mechanisms. Am. J. Med. Genet. B Neuropsychiatr. Genet. 186, 162–172 (2021).
Kapoor, M. et al. Multi-omics integration analysis identifies novel genes for alcoholism with potential overlap with neurodegenerative diseases. Nat. Commun. 12, 5071 (2021).
Baranger, D. A. A. et al. Convergent evidence for predispositional effects of brain gray matter volume on alcohol consumption. Biol. Psychiatry 87, 645–655 (2020).
Marees, A. T. et al. Post-GWAS analysis of six substance use traits improves the identification and functional interpretation of genetic risk loci. Drug Alcohol Depend. 206, 107703 (2020).
Lin, H. et al. Prefrontal cortex eQTLs/mQTLs enriched in genetic variants associated with alcohol use disorder and other diseases. Epigenomics 12, 789–800 (2020).
Sey, N. Y. A. et al. Chromatin architecture in addiction circuitry identifies risk genes and potential biological mechanisms underlying cigarette smoking and alcohol use traits. Mol. Psychiatry 27, 3085–3094 (2022).
Mignogna, K. M., Bacanu, S. A., Riley, B. P., Wolen, A. R. & Miles, M. F. Cross-species alcohol dependence-associated gene networks: Co-analysis of mouse brain gene expression and human genome-wide association data. PLoS One 14, e0202063 (2019).
Huggett, S. B., Bubier, J. A., Chesler, E. J. & Palmer, R. H. C. Do gene expression findings from mouse models of cocaine use recapitulate human cocaine use disorder in reward circuitry? Genes Brain Behav. 20, e12689 (2021).
Palmer, R. H. C. et al. Multi-omic and multi-species meta-analyses of nicotine consumption. Transl. Psychiatry 11, 98 (2021).
Kapoor, M. et al. Analysis of whole genome-transcriptomic organization in brain to identify genes associated with alcoholism. Transl. Psychiatry 9, 89 (2019).
Huggett, S. B. & Stallings, M. C. Genetic architecture and molecular neuropathology of human cocaine addiction. J. Neurosci. 40, 5300–5313 (2020).
Gottesman, I. I. & Gould, T. D. The endophenotype concept in psychiatry: etymology and strategic intentions. Am. J. Psychiatry 160, 636–645 (2003).
Jansen, A. G., Mous, S. E., White, T., Posthuma, D. & Polderman, T. J. What twin studies tell us about the heritability of brain development, morphology, and function: a review. Neuropsychol. Rev. 25, 27–46 (2015).
Grasby, K. L. et al. The genetic architecture of the human cerebral cortex. Science 367, eaay6690 (2020).
Hofer, E. et al. Genetic correlations and genome-wide associations of cortical structure in general population samples of 22,824 adults. Nat. Commun. 11, 4796 (2020).
Elliott, L. T. et al. Genome-wide association studies of brain imaging phenotypes in UK Biobank. Nature 562, 210–216 (2018).
Smith, S. M. et al. An expanded set of genome-wide association studies of brain imaging phenotypes in UK Biobank. Nat. Neurosci. 24, 737–745 (2021).
Bulik-Sullivan, B. et al. An atlas of genetic correlations across human diseases and traits. Nat. Genet. 47, 1236–1241 (2015).
Hatoum, A. S., Johnson, E. C., Agrawal, A. & Bogdan, R. Brain structure and problematic alcohol use: a test of plausible causation using latent causal variable analysis. Brain Imaging Behav. 15, 2741–2745 (2021).
Rabinowitz, J. A. et al. Shared genetic etiology between cortical brain morphology and tobacco, alcohol, and cannabis use. Cereb. Cortex 32, 796–807 (2022).
Marek, S. et al. Reproducible brain-wide association studies require thousands of individuals. Nature 603, 654–660 (2022).
Guimaraes, J. P. O. F. T., Sprooten, E., Beckmann, C. F., Franke, B. & Bralten, J. Shared genetic influences on resting-state functional networks of the brain. Hum. Brain Mapp. 43, 1787–1803 (2022).
Elliott, M. L. et al. What is the test-retest reliability of common task-functional MRI measures? New empirical evidence and a meta-analysis. Psychol. Sci. 31, 792–806 (2020).
Gordon, E. M. et al. Precision functional mapping of individual human brains. Neuron 95, 791–807.e7 (2017).
Hayes, A., Herlinger, K., Paterson, L. & Lingford-Hughes, A. The neurobiology of substance use and addiction: evidence from neuroimaging and relevance to treatment. BJPsych Adv. 26, 367–378 (2020).
Brown, S. A. et al. The National Consortium on Alcohol and NeuroDevelopment in Adolescence (NCANDA): A multisite study of adolescent development and substance use. J. Stud. Alcohol Drugs 76, 895–908 (2015).
Pfefferbaum, A. et al. Altered brain developmental trajectories in adolescents after initiating drinking. Am. J. Psychiatry 175, 370–380 (2018).
Zhao, Q. et al. Association of heavy drinking with deviant fiber tract development in frontal brain systems in adolescents. JAMA Psychiatry 78, 407–415 (2021).
Mascarell Maricic, L. et al. The IMAGEN study: a decade of imaging genetics in adolescents. Mol. Psychiatry 25, 2648–2671 (2020).
Albaugh, M. D. et al. Association of cannabis use during adolescence with neurodevelopment. JAMA Psychiatry 78, 1–11 (2021).
Luby, J. L. et al. Developmental trajectories of the orbitofrontal cortex and anhedonia in middle childhood and risk for substance use in adolescence in a longitudinal sample of depressed and healthy preschoolers. Am. J. Psychiatry 175, 1010–1021 (2018).
Cheetham, A. et al. Orbitofrontal cortex volume and effortful control as prospective risk factors for substance use disorder in adolescence. Eur. Addict. Res. 23, 37–44 (2017).
Wade, N. E. et al. Orbitofrontal cortex volume prospectively predicts cannabis and other substance use onset in adolescents. J. Psychopharmacol. 33, 1124–1131 (2019).
Taffe, M. A. et al. Long-lasting reduction in hippocampal neurogenesis by alcohol consumption in adolescent nonhuman primates. Proc. Natl Acad. Sci. USA 107, 11104–11109 (2010).
Shnitko, T. A., Liu, Z., Wang, X., Grant, K. A. & Kroenke, C. D. Chronic alcohol drinking slows brain development in adolescent and young adult nonhuman primates. eNeuro 6, ENEURO.0044-19.2019 (2019).
Kibaly, C., Xu, C., Cahill, C. M., Evans, C. J. & Law, P. Y. Non-nociceptive roles of opioids in the CNS: opioids’ effects on neurogenesis, learning, memory and affect. Nat. Rev. Neurosci. 20, 5–18 (2019).
Coleman, L. G. Jr., He, J., Lee, J., Styner, M. & Crews, F. T. Adolescent binge drinking alters adult brain neurotransmitter gene expression, behavior, brain regional volumes, and neurochemistry in mice. Alcohol. Clin. Exp. Res. 35, 671–688 (2011).
Douet, V., Chang, L., Cloak, C. & Ernst, T. Genetic influences on brain developmental trajectories on neuroimaging studies: from infancy to young adulthood. Brain Imaging Behav. 8, 234–250 (2014).
Brouwer, R. M. et al. The speed of development of adolescent brain age depends on sex and is genetically determined. Cereb. Cortex 31, 1296–1306 (2021).
Dager, A. D. et al. Shared genetic factors influence amygdala volumes and risk for alcoholism. Neuropsychopharmacology 40, 412–420 (2015).
Henderson, K. E. et al. Cortical thickness in adolescents with a family history of alcohol use disorder. Alcohol. Clin. Exp. Res. 42, 89–99 (2018).
Baranger, D. A. A. & Bogdan, R. Editorial: causal, predispositional, or correlate? Group differences in cognitive control-related brain function in cannabis-using youth raise new questions. J. Am. Acad. Child Adolesc. Psychiatry 58, 665–667 (2019).
Gage, S. H., Munafo, M. R., MacLeod, J., Hickman, M. & Smith, G. D. Cannabis and psychosis. Lancet Psychiatry 2, 380 (2015).
Hatoum, A. S. et al. Polygenic risk scores for alcohol involvement relate to brain structure in substance-naive children: Results from the ABCD study. Genes Brain Behav. 20, e12756 (2021).
Munafo, M. R., Higgins, J. P. T. & Smith, G. D. Triangulating evidence through the inclusion of genetically informed designs. Cold Spring Harb. Perspect. Med. 11, a040659 (2021).
O’Connor, L. J. & Price, A. L. Distinguishing genetic correlation from causation across 52 diseases and complex traits. Nat. Genet. 50, 1728–1734 (2018).
Gillespie, N. A. & Kendler, K. S. Use of genetically informed methods to clarify the nature of the association between cannabis use and risk for schizophrenia. JAMA Psychiatry 78, 467–468 (2021).
Elam, J. S. et al. The human connectome project: a retrospective. Neuroimage 244, 118543 (2021).
Pagliaccio, D. et al. Shared predisposition in the association between cannabis use and subcortical brain structure. JAMA Psychiatry 72, 994–1001 (2015).
Iacono, W. G. & McGue, M. Minnesota twin family study. Twin Res. 5, 482–487 (2002).
Harper, J. et al. Orbitofrontal cortex thickness and substance use disorders in emerging adulthood: causal inferences from a co-twin control/discordant twin study. Addiction 116, 2548–2558 (2021).
Harper, J. et al. The effects of alcohol and cannabis use on the cortical thickness of cognitive control and salience brain networks in emerging adulthood: a co-twin control study. Biol. Psychiatry 89, 1012–1022 (2021).
Hill, S. Y. Trajectories of alcohol use and electrophysiological and morphological indices of brain development: distinguishing causes from consequences. Ann. N. Y. Acad. Sci. 1021, 245–259 (2004).
Rangaswamy, M. & Porjesz, B. Uncovering genes for cognitive (dys)function and predisposition for alcoholism spectrum disorders: a review of human brain oscillations as effective endophenotypes. Brain Res. 1235, 153–171 (2008).
Weiland, B. J. et al. Substance abuse risk in emerging adults associated with smaller frontal gray matter volumes and higher externalizing behaviors. Drug Alcohol Depend. 137, 68–75 (2014).
Hill, S. Y., Lichenstein, S. D., Wang, S. & O’Brien, J. Volumetric differences in cerebellar lobes in individuals from multiplex alcohol dependence families and controls: their relationship to externalizing and internalizing disorders and working memory. Cerebellum 15, 744–754 (2016).
Cservenka, A., Gillespie, A. J., Michael, P. G. & Nagel, B. J. Family history density of alcoholism relates to left nucleus accumbens volume in adolescent girls. J. Stud. Alcohol Drugs 76, 47–56 (2015).
Hill, S. Y. et al. Right amygdala volume in adolescent and young adult offspring from families at high risk for developing alcoholism. Biol. Psychiatry 49, 894–905 (2001).
Acheson, A. et al. Increased forebrain activations in youths with family histories of alcohol and other substance use disorders performing a Go/NoGo task. Alcohol. Clin. Exp. Res. 38, 2944–2951 (2014).
Cservenka, A., Herting, M. M. & Nagel, B. J. Atypical frontal lobe activity during verbal working memory in youth with a family history of alcoholism. Drug Alcohol Depend. 123, 98–104 (2012).
Cservenka, A. & Nagel, B. J. Risky decision-making: an FMRI study of youth at high risk for alcoholism. Alcohol. Clin. Exp. Res. 36, 604–615 (2012).
Yau, W. Y. et al. Nucleus accumbens response to incentive stimuli anticipation in children of alcoholics: relationships with precursive behavioral risk and lifetime alcohol use. J. Neurosci. 32, 2544–2551 (2012).
Heitzeg, M. M., Nigg, J. T., Yau, W. Y., Zucker, R. A. & Zubieta, J. K. Striatal dysfunction marks preexisting risk and medial prefrontal dysfunction is related to problem drinking in children of alcoholics. Biol. Psychiatry 68, 287–295 (2010).
Zucker, R. A., Heitzeg, M. M. & Nigg, J. T. Parsing the undercontrol/disinhibition pathway to substance use disorders: a multilevel developmental problem. Child Dev. Perspect. 5, 248–255 (2011).
Kong, A. et al. The nature of nurture: Effects of parental genotypes. Science 359, 424–428 (2018).
Molina, B. S., Donovan, J. E. & Belendiuk, K. A. Familial loading for alcoholism and offspring behavior: mediating and moderating influences. Alcohol. Clin. Exp. Res. 34, 1972–1984 (2010).
Balbona, J. V., Kim, Y. & Keller, M. C. Estimation of parental effects using polygenic scores. Behav. Genet. 51, 264–278 (2021).
Maes, H. H. et al. Cross-cultural comparison of genetic and cultural transmission of smoking initiation using an extended twin kinship model. Twin Res. Hum. Genet. 21, 179–190 (2018).
Volkow, N. D. et al. The conception of the ABCD study: From substance use to a broad NIH collaboration. Dev. Cogn. Neurosci. 32, 4–7 (2018).
Wray, N. R. et al. From basic science to clinical application of polygenic risk scores: a primer. JAMA Psychiatry 78, 101–109 (2021).
Lisdahl, K. M. et al. Substance use patterns in 9-10 year olds: Baseline findings from the adolescent brain cognitive development (ABCD) study. Drug Alcohol Depend. 227, 108946 (2021).
Paul, S. E. et al. Associations between prenatal cannabis exposure and childhood outcomes: results from the ABCD study. JAMA Psychiatry 78, 64–76 (2021).
Baranger, D. A. A. et al. Association of mental health burden with prenatal cannabis exposure from childhood to early adolescence: longitudinal findings from the Adolescent Brain Cognitive Development (ABCD) study. JAMA Pediatrics https://doi.org/10.1001/jamapediatrics.2022.3191 (2022).
Roffman, J. L. et al. Association of adverse prenatal exposure burden with child psychopathology in the Adolescent Brain Cognitive Development (ABCD) study. PLoS One 16, e0250235 (2021).
El Marroun, H. et al. Prenatal tobacco exposure and brain morphology: a prospective study in young children. Neuropsychopharmacology 39, 792–800 (2014).
Ross, E. J., Graham, D. L., Money, K. M. & Stanwood, G. D. Developmental consequences of fetal exposure to drugs: what we know and what we still must learn. Neuropsychopharmacology 40, 61–87 (2015).
D’Onofrio, B. M., Sjolander, A., Lahey, B. B., Lichtenstein, P. & Oberg, A. S. Accounting for confounding in observational studies. Annu. Rev. Clin. Psychol. 16, 25–48 (2020).
Quinn, P. D. et al. Association between maternal smoking during pregnancy and severe mental illness in offspring. JAMA Psychiatry 74, 589–596 (2017).
Micalizzi, L. et al. A sibling-comparison study of smoking during pregnancy and risk for reading-related problems. Neurotoxicol Teratol. 84, 106961 (2021).
Donald, K. A. et al. Neuroimaging effects of prenatal alcohol exposure on the developing human brain: a magnetic resonance imaging review. Acta Neuropsychiatr. 27, 251–269 (2015).
Easey, K. E. et al. Characterization of alcohol polygenic risk scores in the context of mental health outcomes: Within-individual and intergenerational analyses in the Avon Longitudinal Study of Parents and Children. Drug Alcohol Depend. 221, 108654 (2021).
Vrieze, S. I., McGue, M., Miller, M. B., Hicks, B. M. & Iacono, W. G. Three mutually informative ways to understand the genetic relationships among behavioral disinhibition, alcohol use, drug use, nicotine use/dependence, and their co-occurrence: twin biometry, GCTA, and genome-wide scoring. Behav. Genet. 43, 97–107 (2013).
Dick, D. M., Adkins, A. E. & Kuo, S. I. Genetic influences on adolescent behavior. Neurosci. Biobehav. Rev. 70, 198–205 (2016).
Kendler, K. S., Prescott, C. A., Myers, J. & Neale, M. C. The structure of genetic and environmental risk factors for common psychiatric and substance use disorders in men and women. Arch. Gen. Psychiatry 60, 929–937 (2003).
Iacono, W. G., Malone, S. M. & McGue, M. Behavioral disinhibition and the development of early-onset addiction: common and specific influences. Annu. Rev. Clin. Psychol. 4, 325–348 (2008).
McGue, M., Irons, D. & Iacono, W. G. The adolescent origins of substance use disorders: a behavioral genetic perspective. Nebr Symp. Motiv. 61, 31–50 (2014).
Hamilton, P. J. & Nestler, E. J. Epigenetics and addiction. Curr. Opin. Neurobiol. 59, 128–136 (2019).
Pang, R. D., Farrahi, L., Glazier, S., Sussman, S. & Leventhal, A. M. Depressive symptoms, negative urgency and substance use initiation in adolescents. Drug Alcohol Depend. 144, 225–230 (2014).
Levey, D. F. et al. Bi-ancestral depression GWAS in the Million Veteran Program and meta-analysis in >1.2 million individuals highlight new therapeutic directions. Nat. Neurosci. 24, 954–963 (2021).
Nagel, M. et al. Meta-analysis of genome-wide association studies for neuroticism in 449,484 individuals identifies novel genetic loci and pathways. Nat. Genet. 50, 920–927 (2018).
Nagel, M., Watanabe, K., Stringer, S., Posthuma, D. & van der Sluis, S. Item-level analyses reveal genetic heterogeneity in neuroticism. Nat. Commun. 9, 905 (2018).
Sanchez-Roige, S. et al. Genome-wide association studies of impulsive personality traits (BIS-11 and UPPS-P) and drug experimentation in up to 22,861 adult research participants identify loci in the CACNA1I and CADM2 genes. J. Neurosci. 39, 2562–2572 (2019).
Zorrilla, E. P. & Koob, G. F. Impulsivity derived from the dark side: neurocircuits that contribute to negative urgency. Front. Behav. Neurosci. 13, 136 (2019).
Koob, G. F. Anhedonia, hyperkatifeia, and negative reinforcement in substance use disorders. Curr. Top. Behav. Neurosci. 58, 147–165 (2022).
Grotzinger, A. D. et al. Genomic structural equation modelling provides insights into the multivariate genetic architecture of complex traits. Nat. Hum. Behav. 3, 513–525 (2019).
Edenberg, H. J. & McClintick, J. N. Alcohol dehydrogenases, aldehyde dehydrogenases, and alcohol use disorders: a critical review. Alcohol. Clin. Exp. Res. 42, 2281–2297 (2018).
Andrews, S. J., Fulton-Howard, B. & Goate, A. Interpretation of risk loci from genome-wide association studies of Alzheimer’s disease. Lancet Neurol. 19, 326–335 (2020).
Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature 511, 421–427 (2014).
Okbay, A. et al. Genome-wide association study identifies 74 loci associated with educational attainment. Nature 533, 539–542 (2016).
Janes, A. C. et al. Association between CHRNA5 genetic variation at rs16969968 and brain reactivity to smoking images in nicotine dependent women. Drug Alcohol Depend. 120, 7–13 (2012).
Hong, L. E. et al. A genetically modulated, intrinsic cingulate circuit supports human nicotine addiction. Proc. Natl Acad. Sci. USA 107, 13509–13514 (2010).
Chaarani, B. et al. Low smoking exposure, the adolescent brain, and the modulating role of CHRNA5 polymorphisms. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 4, 672–679 (2019).
Hartwell, E. E. et al. Systematic review and meta-analysis of the moderating effect of rs1799971 in OPRM1, the mu-opioid receptor gene, on response to naltrexone treatment of alcohol use disorder. Addiction 115, 1426–1437 (2020).
Biernacka, J. M. et al. Genetic contributions to alcohol use disorder treatment outcomes: a genome-wide pharmacogenomics study. Neuropsychopharmacology 46, 2132–2139 (2021).
Deak, J. D. et al. Million Veteran Program. Genome-wide investigation of maximum habitual alcohol intake in US veterans in relation to alcohol consumption traits and alcohol use disorder. JAMA Netw. Open 5, e2238880 (2022).
Sherva, R. et al. Association of a single nucleotide polymorphism in neuronal acetylcholine receptor subunit alpha 5 (CHRNA5) with smoking status and with ‘pleasurable buzz’ during early experimentation with smoking. Addiction 103, 1544–1552 (2008).
McCarthy, D. M., Pedersen, S. L., Lobos, E. A., Todd, R. D. & Wall, T. L. ADH1B*3 and response to alcohol in African-Americans. Alcohol. Clin. Exp. Res. 34, 1274–1281 (2010).
Duranceaux, N. C. et al. Associations of variations in alcohol dehydrogenase genes with the level of response to alcohol in non-Asians. Alcohol. Clin. Exp. Res. 30, 1470–1478 (2006).
Jensen, K. P. et al. A CHRNA5 smoking risk variant decreases the aversive effects of nicotine in humans. Neuropsychopharmacology 40, 2813–2821 (2015).
Kreek, M. J., Nielsen, D. A., Butelman, E. R. & LaForge, K. S. Genetic influences on impulsivity, risk taking, stress responsivity and vulnerability to drug abuse and addiction. Nat. Neurosci. 8, 1450–1457 (2005).
Wright, S. N. & Little, A. R. NIDA vision for big data science to understand the biological underpinnings of substance use disorders. Neuropsychopharmacology 46, 262 (2021).
Veligati, S. et al. Changes in alcohol and cigarette consumption in response to medical and recreational cannabis legalization: Evidence from U.S. state tax receipt data. Int. J. Drug Policy 75, 102585 (2020).
Kendler, K. S. et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women: an epidemiological and cotwin control analysis. Arch. Gen. Psychiatry 57, 953–959 (2000).
Sartor, C. E., Agrawal, A., McCutcheon, V. V., Duncan, A. E. & Lynskey, M. T. Disentangling the complex association between childhood sexual abuse and alcohol-related problems: a review of methodological issues and approaches. J. Stud. Alcohol Drugs 69, 718–727 (2008).
Tomasi, D. & Volkow, N. D. Associations of family income with cognition and brain structure in USA children: prevention implications. Mol. Psychiatry 26, 6619–6629 (2021).
Yehuda, R. & Lehrner, A. Intergenerational transmission of trauma effects: putative role of epigenetic mechanisms. World Psychiatry 17, 243–257 (2018).
Meyers, J. et al. Early sexual trauma exposure and neural response inhibition in adolescence and young adults: trajectories of frontal theta oscillations during a Go/No-Go Task. J. Am. Acad. Child Adolesc. Psychiatry 58, 242–255.e2 (2019).
Meyers, J. L. et al. Psychosocial moderation of polygenic risk for cannabis involvement: the role of trauma exposure and frequency of religious service attendance. Transl. Psychiatry 9, 269 (2019).
Szutorisz, H. & Hurd, Y. L. Feeding the developing brain: the persistent epigenetic effects of early life malnutrition. Biol. Psychiatry 80, 730–732 (2016).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (American Psychiatric Association, 2013).
McLellan, A. T., Koob, G. F. & Volkow, N. D. Preaddiction-a missing concept for treating substance use disorders. JAMA Psychiatry 79, 749–751 (2022).
Lane, S. P., Steinley, D. & Sher, K. J. Meta-analysis of DSM alcohol use disorder criteria severities: structural consistency is only ‘skin deep’. Psychol. Med. 46, 1769–1784 (2016).
Hines, L. A. et al. Overlap of heritable influences between cannabis use disorder, frequency of use and opportunity to use cannabis: trivariate twin modelling and implications for genetic design. Psychol. Med. 48, 2786–2793 (2018).
Gillespie, N. A. et al. Longitudinal modeling of genetic and environmental influences on self-reported availability of psychoactive substances: alcohol, cigarettes, marijuana, cocaine and stimulants. Psychol. Med. 37, 947–959 (2007).
Agrawal, A., Madden, P. A., Bucholz, K. K., Heath, A. C. & Lynskey, M. T. Initial reactions to tobacco and cannabis smoking: a twin study. Addiction 109, 663–671 (2014).
Edwards, A. C. et al. Meta-analysis of genetic influences on initial alcohol sensitivity. Alcohol. Clin. Exp. Res. 42, 2349–2359 (2018).
Grant, J. D. et al. Subjective reactions to cocaine and marijuana are associated with abuse and dependence. Addict. Behav. 30, 1574–1586 (2005).
Lyons, M. J. et al. How do genes influence marijuana use? The role of subjective effects. Addiction 92, 409–417 (1997).
Windle, M. A multilevel developmental contextual approach to substance use and addiction. Biosocieties 5, 124–136 (2010).
Lee, M. R. & Sher, K. J. “Maturing out” of binge and problem drinking. Alcohol Res. 39, 31–42 (2018).
Verges, A. et al. Refining the notion of maturing out: results from the national epidemiologic survey on alcohol and related conditions. Am. J. Public. Health 103, e67–e73 (2013).
Iacono, W. G. et al. The utility of twins in developmental cognitive neuroscience research: How twins strengthen the ABCD research design. Dev. Cogn. Neurosci. 32, 30–42 (2018).
Porjesz, B., Jones, K. & Begleiter, H. The genetics of oscillations in the human brain. Suppl. Clin. Neurophysiol. 57, 441–449 (2004).
Anokhin, A. P. Genetic psychophysiology: advances, problems, and future directions. Int. J. Psychophysiol. 93, 173–197 (2014).
Mackey, S. et al. Genetic imaging consortium for addiction medicine: from neuroimaging to genes. Prog. Brain Res. 224, 203–223 (2016).
Van Essen, D. C. et al. The WU-Minn human connectome project: an overview. Neuroimage 80, 62–79 (2013).
Kwako, L. E., Momenan, R., Litten, R. Z., Koob, G. F. & Goldman, D. Addictions neuroclinical assessment: a neuroscience-based framework for addictive disorders. Biol. Psychiatry 80, 179–189 (2016).
Vanyukov, M. M. et al. Liability to substance use disorders: 1. Common mechanisms and manifestations. Neurosci. Biobehav. Rev. 27, 507–515 (2003).
Vanyukov, M. M. et al. Liability to substance use disorders: 2. A measurement approach. Neurosci. Biobehav. Rev. 27, 517–526 (2003).
Latzman, R. D. & DeYoung, C. G., Hitop Neurobiological Foundations Workgroup. Using empirically-derived dimensional phenotypes to accelerate clinical neuroscience: the Hierarchical Taxonomy of Psychopathology (HiTOP) framework. Neuropsychopharmacology 45, 1083–1085 (2020).
Boness, C. L., Watts, A. L., Moeller, K. N. & Sher, K. J. The etiologic, theory-based, ontogenetic hierarchical framework of alcohol use disorder: a translational systematic review of reviews. Psychol. Bull. 147, 1075–1123 (2021).
Watanabe, K. et al. A global overview of pleiotropy and genetic architecture in complex traits. Nat. Genet. 51, 1339–1348 (2019).
Barr, P. B. et al. Using polygenic scores for identifying individuals at increased risk of substance use disorders in clinical and population samples. Transl. Psychiatry 10, 196 (2020).
Dick, A. S. et al. Meaningful associations in the adolescent brain cognitive development study. Neuroimage 239, 118262 (2021).
Baranger, D. A. A. et al. Borderline personality traits are not correlated with brain structure in two large samples. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 5, 669–677 (2020).
Bogdan, R., Baranger, D. A. A. & Agrawal, A. Polygenic risk scores in clinical psychology: bridging genomic risk to individual differences. Annu. Rev. Clin. Psychol. 14, 119–157 (2018).
Frangou, S. et al. Cortical thickness across the lifespan: Data from 17,075 healthy individuals aged 3-90 years. Hum. Brain Mapp. 43, 431–451 (2022).
Martin, A. R. et al. Clinical use of current polygenic risk scores may exacerbate health disparities. Nat. Genet. 51, 584–591 (2019).
Li, D. et al. Genome-wide association study of copy number variations (CNVs) with opioid dependence. Neuropsychopharmacology 40, 1016–1026 (2015).
Sulovari, A., Liu, Z., Zhu, Z. & Li, D. Genome-wide meta-analysis of copy number variations with alcohol dependence. Pharmacogenomics J. 18, 398–405 (2018).
Mukamel, R. E. et al. Protein-coding repeat polymorphisms strongly shape diverse human phenotypes. Science 373, 1499–1505 (2021).
Skene, N. G. et al. Genetic identification of brain cell types underlying schizophrenia. Nat. Genet. 50, 825–833 (2018).
Ruzicka, W. B. et al. Single-cell dissection of schizophrenia reveals neurodevelopmental-synaptic axis and transcriptional resilience. medRxiv https://doi.org/10.1101/2020.11.06.20225342v1 (2020).
Nasser, J. et al. Genome-wide enhancer maps link risk variants to disease genes. Nature 593, 238–243 (2021).
Baranger, D. A. A. et al. Multi-omics analyses cannot identify true-positive novel associations from underpowered genome-wide association studies of four brain-related traits. bioRxiv https://doi.org/10.1101/2022.04.13.487655 (2022).
Liang, Y. et al. Polygenic transcriptome risk scores (PTRS) can improve portability of polygenic risk scores across ancestries. Genome Biol. 23, 23 (2022).
Marquez-Luna, C. et al. Incorporating functional priors improves polygenic prediction accuracy in UK Biobank and 23andMe data sets. Nat. Commun. 12, 6052 (2021).
Hu, Y. et al. Leveraging functional annotations in genetic risk prediction for human complex diseases. PLoS Comput. Biol. 13, e1005589 (2017).
Amariuta, T. et al. Improving the trans-ancestry portability of polygenic risk scores by prioritizing variants in predicted cell-type-specific regulatory elements. Nat. Genet. 52, 1346–1354 (2020).
Hall, W., Carter, A. & Forlini, C. Brain disease model of addiction: misplaced priorities? Lancet Psychiatry 2, 867 (2015).
Risch, N. & Merikangas, K. The future of genetic studies of complex human diseases. Science 273, 1516–1517 (1996).
Potenza, M. N. et al. Gambling disorder. Nat. Rev. Dis. Prim. 5, 51 (2019).
Slutske, W. S. et al. Common genetic vulnerability for pathological gambling and alcohol dependence in men. Arch. Gen. Psychiatry 57, 666–673 (2000).
Kaye, W. H. et al. Does a shared neurobiology for foods and drugs of abuse contribute to extremes of food ingestion in anorexia and bulimia nervosa? Biol. Psychiatry 73, 836–842 (2013).
Hasin, D. S. et al. DSM-5 criteria for substance use disorders: recommendations and rationale. Am. J. Psychiatry 170, 834–851 (2013).
Saunders, J. B., Degenhardt, L., Reed, G. M. & Poznyak, V. Alcohol use disorders in ICD-11: past, present, and future. Alcohol. Clin. Exp. Res. 43, 1617–1631 (2019).
Saunders, J. B., Aasland, O. G., Babor, T. F., de la Fuente, J. R. & Grant, M. Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption–II. Addiction 88, 791–804 (1993).
Quach, B. C. et al. Expanding the genetic architecture of nicotine dependence and its shared genetics with multiple traits. Nat. Commun. 11, 5562 (2020).
Gaddis, N. et al. Multi-trait genome-wide association study of opioid addiction: OPRM1 and beyond. Sci. Rep. 12, 16873 (2022).
Kember, R. L. et al. Cross-ancestry meta-analysis of opioid use disorder uncovers novel loci with predominant effects in brain regions associated with addiction. Nat. Neurosci. https://doi.org/10.1038/s41593-022-01160-z (2022).
Deak, J. D. et al. Genome-wide association study in individuals of European and African ancestry and multi-trait analysis of opioid use disorder identifies 19 independent genome-wide significant risk loci. Mol. Psychiatry https://doi.org/10.1038/s41380-022-01709-1 (2022).
Cabana-Dominguez, J., Shivalikanjli, A., Fernandez-Castillo, N. & Cormand, B. Genome-wide association meta-analysis of cocaine dependence: Shared genetics with comorbid conditions. Prog. Neuropsychopharmacol. Biol. Psychiatry 94, 109667 (2019).
Acknowledgements
The authors acknowledge the following funding from the United States National Institutes of Health: R.B. (R01DA54750; R21AA27827, U01DA055367), A.S.H. (T32DA007261, K01AA030083), E.C.J. (K01DA51759), A.A. (K02DA32573, R01DA54750). Funders were not involved in the preparation of this manuscript in any way.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article and wrote the article. A.A., A.S.H. and R.B. contributed substantially to discussion of the content. A.A., E.C.J. and R.B. reviewed and/or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neuroscience thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
HEALthy Brain and Cognitive Development (HBCD) study: https://heal.nih.gov/research/infants-and-children/healthy-brain
Glossary
- Anhedonia
-
The loss of pleasure or lack of reactivity to pleasurable stimuli.
- Binging
-
Consuming a large amount of a substance (typically alcohol) in a short period of time.
- Candidate gene
-
A gene posited to be associated with a phenotype based on prior knowledge.
- Compulsive use
-
Drug consumption that is not under control and typically functions to achieve drug-present homeostasis and alleviation of negative affect/withdrawal as opposed to drug-induced euphoric reward.
- Craving
-
A persistent desire to use a substance.
- Developmental vulnerability
-
Vulnerability to a given outcome that arises in the context of typical development.
- Executive function
-
Complex mental processes and cognition (for example, working memory) that control skills (for example, organizing, solving) and regulate emotion and behaviour.
- Expression quantitative trait loci
-
(eQTL). Genetic variants that modify the expression of a gene by acting upon the regulatory elements of the gene.
- Fractional anisotropy
-
A measure of the degree of anisotropy of a diffusion process ranging from 0 to 1. In the context of diffusion tensor imaging, it reflects the uniform directionality of white-matter fibres in the brain and is often conceptualized as an index of white matter integrity and structural connectivity.
- Gene variants
-
Sections of DNA sequence that differ across groups of individuals.
- Genetic architectures
-
Distinct genetic factors that influence one or more traits.
- Genetic liability
-
The contribution of genetic factors to the likelihood of observing a phenotype.
- Genetic nurture
-
The effect of genetically influenced parent behaviour on offspring behaviour.
- Genome-wide association studies
-
(GWAS). A hypothesis-free analysis of the association between common genetic variation across the genome and a phenotype.
- Genomic structural equation modelling
-
A statistical genetics method for identifying genetic variants that influence multiple phenotypes as well as each individual phenotype.
- Heritability
-
The proportion of total variation in a phenotype that is due to genetic factors.
- Incentive salience
-
A cognitive process that motivates behaviour towards reward.
- Machine learning
-
A data-driven approach that iteratively examines a training data set for patterns across large numbers and diverse types of variables associated with an outcome and, upon ‘learning’ these data patterns, can be used to test whether these patterns accurately predict the outcome in independent data sets.
- Negative reinforcement
-
The removal of something unpleasant or uncomfortable by a stimulus and/or behaviour.
- Negative urgency
-
A personality facet related to impulsive behaviour in the context of negative mood or experiences.
- Pleiotropic effects
-
The influences of a variant, gene or groups of variants on multiple phenotypes.
- Polygenic
-
The genetic characteristic of traits that is due to the aggregated small effects of many genetic variants.
- Positive reinforcement
-
Reward obtained after a stimulus and/or behaviour.
- Positive urgency
-
A facet of personality related to impulsive behaviour in the context of anticipated reward.
- Predictive reward signals
-
Neural signals that demarcate the expected delivery of reward following extrinsic and/or intrinsic cues.
- Predispositional liability
-
The aspect of an outcome that is attributable to predispositional (that is, genetic variation, prior experiences) factors.
- Regulatory elements
-
Components of a gene, such as the promoter and introns, that regulate its expression.
- Resting-state functional connectivity
-
Correlated signal between brain regions in the absence of any stimulus or task.
- Single nucleotide polymorphism
-
(SNP). A single base pair in the genome that varies across individuals.
- Trait-like vulnerability
-
Vulnerability to a given trait.
- Twin studies
-
Comparisons of phenotype correlations in identical and fraternal twins to parse the role of genetic and environmental effects on a given phenotype or set of phenotypes.
- Withdrawal
-
Physical (for example, headaches and insomnia) and psychological (for example, depressed mood) aversive experiences that occur when use of a substance is discontinued.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bogdan, R., Hatoum, A.S., Johnson, E.C. et al. The Genetically Informed Neurobiology of Addiction (GINA) model. Nat Rev Neurosci 24, 40–57 (2023). https://doi.org/10.1038/s41583-022-00656-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41583-022-00656-8
This article is cited by
-
Addiction biotypes: a paradigm shift for future treatment strategies?
Molecular Psychiatry (2024)