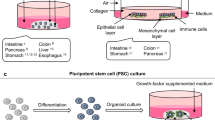
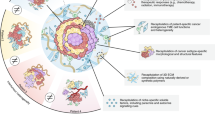
Abstract
Cancer is a major public health problem worldwide. Gastrointestinal cancers account for approximately one-third of the total global cancer incidence and mortality. Historically, the mechanisms of tumour initiation and progression in the gastrointestinal tract have been studied using cancer cell lines in vitro and animal models. Traditional cell culture methods are associated with a strong selection of aberrant genomic variants that no longer reflect the original tumours in terms of their (metastatic) behaviour or response to therapy. Organoid technology has emerged as a powerful alternative method for culturing gastrointestinal tumours and the corresponding normal tissues in a manner that preserves their genetic, phenotypic and behavioural traits. Importantly, accumulating evidence suggests that organoid cultures have great value in predicting the outcome of therapy in individual patients. Herein, we review the current literature on organoid models of the most common gastrointestinal cancers, including colorectal cancer, gastric cancer, oesophageal cancer, liver cancer and pancreatic cancer, and their value in modelling tumour initiation, metastatic progression and therapy response. We also explore the limitations of current organoid models and discuss how they could be improved to maximally benefit basic and translational research in the future, especially in the fields of drug discovery and personalized medicine.
Key points
-
Gastrointestinal cancers account for one-third of the total global cancer incidence and mortality; therefore, it is essential to translate knowledge from basic research into health benefits by advancing therapeutics.
-
Preclinical cancer research has been heavily reliant on cell lines and animal models, but both fail to recapitulate the original human tumours.
-
Organoid technology has emerged as a powerful alternative method for culturing gastrointestinal tumours and their corresponding normal tissues in a manner that preserves their genetic, phenotypic and behavioural traits.
-
Organoid models have been used to model tumour initiation, metastatic progression and therapy response of the most common gastrointestinal cancers, including colorectal cancer and liver cancer.
-
Clinical applications of patient-derived organoids are promising, as accumulating evidence has revealed the potential of organoid models in drug discovery, modelling therapy response and personalized medicine.
-
Current organoid models have multiple limitations; therefore, more work is necessary to enable them to maximally benefit basic and translational research in gastrointestinal cancers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 69, 7–34 (2019).
Griffin-Sobel, J. P. Gastrointestinal cancers: screening and early detection. Semin. Oncol. Nurs. 33, 165–171 (2017).
Shams, A. Z. & Haug, U. Strategies for prevention of gastrointestinal cancers in developing countries: a systematic review. J. Glob. Health. 7, 020405 (2017).
McMillin, D. W., Negri, J. M. & Mitsiades, C. S. The role of tumour-stromal interactions in modifying drug response: challenges and opportunities. Nat. Rev. Drug Discov. 12, 217–228 (2013).
Hay, M., Thomas, D. W., Craighead, J. L., Economides, C. & Rosenthal, J. Clinical development success rates for investigational drugs. Nat. Biotechnol. 32, 40–51 (2014).
Ben-David, U. et al. Genetic and transcriptional evolution alters cancer cell line drug response. Nature 560, 325–330 (2018). This study describes the extent, origins and consequences of genetic variation within cell lines, and provides suggestions for measuring such variation to maximally support reproducible cancer research.
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009). This study describes a novel in vitro system to generate long-term intestinal organoid cultures; this method is fundamental to current organoid technology.
Eiraku, M. & Sasai, Y. Self-formation of layered neural structures in three-dimensional culture of ES cells. Curr. Opin. Neurobiol. 22, 768–777 (2012).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
Rock, J. R. et al. Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc. Natl Acad. Sci. USA 106, 12771–12775 (2009).
Wong, A. P. et al. Directed differentiation of human pluripotent stem cells into mature airway epithelia expressing functional CFTR protein. Nat. Biotechnol. 30, 876–882 (2012).
Karthaus, W. R. et al. Identification of multipotent luminal progenitor cells in human prostate organoid cultures. Cell 159, 163–175 (2014).
Gao, D. et al. Organoid cultures derived from patients with advanced prostate cancer. Cell 159, 176–187 (2014).
Eiraku, M. et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472, 51–56 (2011).
Nakano, T. et al. Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell 10, 771–785 (2012).
Nanduri, L. S. et al. Purification and ex vivo expansion of fully functional salivary gland stem cells. Stem Cell Reports 3, 957–964 (2014).
Maimets, M. et al. Long-term in vitro expansion of salivary gland stem cells driven by Wnt signals. Stem Cell Reports 6, 150–162 (2016).
Sachs, N. et al. A living biobank of breast cancer organoids captures disease heterogeneity. Cell 172, 373–386 (2018).
Ren, W. et al. Single Lgr5- or Lgr6-expressing taste stem/progenitor cells generate taste bud cells ex vivo. Proc. Natl Acad. Sci. USA 111, 16401–16406 (2014).
Clevers, H. Modeling development and disease with organoids. Cell 165, 1586–1597 (2016).
Spence, J. R. et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470, 105–109 (2011). This study establishes a methodology to direct differentiation of hPSCs into intestinal tissue by manipulating the niche factors to mimic embryonic intestinal development.
McCracken, K. W. et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature 516, 400–404 (2014). This study provides evidence that gastric organoids can be generated de novo from hPSCs by manipulating multiple signalling pathways including WNT, EGF and BMP.
Takebe, T. et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499, 481–484 (2013). This study is the first to generate vascularized and functional human liver by transplanting liver buds created from hPSCs in vitro into immunodeficient mice.
Broda, T. R., McCracken, K. W. & Wells, J. M. Generation of human antral and fundic gastric organoids from pluripotent stem cells. Nat. Protoc. 14, 28–50 (2019).
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007). This study provides evidence that Lgr5 acts as a stem cell marker in the small intestine and colon; notably, the identification of Lgr5 facilitated the establishment of a novel intestinal organoid system in a subsequent study by Sato et al. (2009).
Sato, T. et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 141, 1762–1772 (2011). This study establishes methodologies for generating long-term human-derived and mouse-derived epithelial organoids from normal colon and oesophagus as well as from Barrett’s epithelium, colon adenoma and adenocarcinoma.
Jung, P. et al. Isolation and in vitro expansion of human colonic stem cells. Nat. Med. 17, 1225–1227 (2011).
Jass, J. R. Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology 50, 113–130 (2007).
IJspeert, J. E., Vermeulen, L., Meijer, G. A. & Dekker, E. Serrated neoplasia — role in colorectal carcinogenesis and clinical implications. Nat. Rev. Gastroenterol. Hepatol. 12, 401–409 (2015).
Bae, J. M., Kim, J. H. & Kang, G. H. Molecular subtypes of colorectal cancer and their clinicopathologic features, with an emphasis on the serrated neoplasia pathway. Arch. Pathol. Lab. Med. 140, 406–412 (2016).
Inamura, K. Colorectal cancers: an update on their molecular pathology. Cancers 10, 26 (2018).
The Cancer Genome Atlas Network Comprehensive molecular characterization of human colon and rectal cancer. Nature 487, 330–337 (2012).
Guinney, J. et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 21, 1350–1356 (2015).
van de Wetering, M. et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 161, 933–945 (2015). This study performs high-throughput screening on a biobank of PDOs to identify potential target drugs and biomarkers of CRC.
Fujii, M. et al. A colorectal tumor organoid library demonstrates progressive loss of niche factor requirements during tumorigenesis. Cell Stem Cell 18, 827–838 (2016).
Weeber, F. et al. Preserved genetic diversity in organoids cultured from biopsies of human colorectal cancer metastases. Proc. Natl Acad. Sci. USA 112, 13308–13311 (2015). Using whole-genome sequencing, this study provides evidence that organoids derived from patients can preserve the genetic diversity of the original tumours.
Drost, J. et al. Sequential cancer mutations in cultured human intestinal stem cells. Nature 521, 43–47 (2015). Using CRISPR–Cas9 genome editing, the most commonly mutated genes associated with CRC, including APC, SMAD4, TP53 and KRAS, are introduced into human colonic organoids to model the adenoma–carcinoma sequence.
Matano, M. et al. Modeling colorectal cancer using CRISPR–Cas9-mediated engineering of human intestinal organoids. Nat. Med. 21, 256–262 (2015). A study similar to Drost et al. (2015) but with the introduction of an additional mutation in PIK3CA into human colonic organoids; both studies provide evidence that organoids alone or upon transplantation into immunodeficient mice can recapitulate the adenoma–carcinoma sequence and metastasis in humans.
Fumagalli, A. et al. Genetic dissection of colorectal cancer progression by orthotopic transplantation of engineered cancer organoids. Proc. Natl Acad. Sci. USA 114, E2357–E2364 (2017).
Fearon, E. R. & Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 61, 759–767 (1990).
Vogelstein, B. et al. Cancer genome landscapes. Science 339, 1546–1558 (2013).
Bolhaqueiro, A. C. F. et al. Ongoing chromosomal instability and karyotype evolution in human colorectal cancer organoids. Nat. Genet. 51, 824–834 (2019).
Sakai, E. et al. Combined mutation of Apc, Kras, and Tgfbr2 effectively drives metastasis of intestinal cancer. Cancer Res. 78, 1334–1346 (2018).
Roper, J. et al. In vivo genome editing and organoid transplantation models of colorectal cancer and metastasis. Nat. Biotechnol. 35, 569–576 (2017).
O’Rourke, K. P. et al. Transplantation of engineered organoids enables rapid generation of metastatic mouse models of colorectal cancer. Nat. Biotechnol. 35, 577–582 (2017).
Murcia, O. et al. Serrated colorectal cancer: molecular classification, prognosis, and response to chemotherapy. World J. Gastroenterol. 22, 3516–3530 (2016).
Snover, D. C. Update on the serrated pathway to colorectal carcinoma. Hum. Pathol. 42, 1–10 (2011).
Fessler, E. et al. TGFβ signaling directs serrated adenomas to the mesenchymal colorectal cancer subtype. EMBO Mol. Med. 8, 745–760 (2016).
Lannagan, T. R. M. et al. Genetic editing of colonic organoids provides a molecularly distinct and orthotopic preclinical model of serrated carcinogenesis. Gut 68, 684–692 (2019). Using CRISPR–Cas9 genome editing, mutations on serrated CRC-associated genes are introduced into mouse colonic organoids to model serrated carcinogenesis.
Kawasaki, K. et al. Chromosome engineering of human colon-derived organoids to develop a model of traditional serrated adenoma. Gastroenterology 158, 638–651.e8 (2019).
Richard, G. F., Kerrest, A. & Dujon, B. Comparative genomics and molecular dynamics of DNA repeats in eukaryotes. Microbiol. Mol. Biol. Rev. 72, 686–727 (2008).
Boland, C. R. & Goel, A. Microsatellite instability in colorectal cancer. Gastroenterology 138, 2073–2087 (2010).
Thibodeau, S. N., Bren, G. & Schaid, D. Microsatellite instability in cancer of the proximal colon. Science 260, 816–819 (1993).
Drost, J. et al. Use of CRISPR-modified human stem cell organoids to study the origin of mutational signatures in cancer. Science 358, 234–238 (2017). Using CRISPR–Cas9 genome editing, MLH1, a regulatory gene in the DNA mismatch repair system, is deleted in human colonic organoids to model microsatellite instability-driven CRC.
Barker, N. et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457, 608–611 (2009). This study depicts the role of LGR5 + crypt stem cells in carcinogenesis by deleting a CRC-driver gene, APC, and concludes that these cells can be the cell of origin of intestinal cancer.
Vermeulen, L. et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat. Cell Biol. 12, 468–476 (2010).
Hirsch, D. et al. LGR5 positivity defines stem-like cells in colorectal cancer. Carcinogenesis 35, 849–858 (2014).
Shimokawa, M. et al. Visualization and targeting of LGR5+ human colon cancer stem cells. Nature 545, 187–192 (2017).
de Sousa e Melo, F. et al. A distinct role for Lgr5+ stem cells in primary and metastatic colon cancer. Nature 543, 676–680 (2017).
Dame, M. K. et al. Identification, isolation and characterization of human LGR5-positive colon adenoma cells. Development 145, dev153049 (2018).
Tilg, H., Adolph, T. E., Gerner, R. R. & Moschen, A. R. The intestinal microbiota in colorectal cancer. Cancer Cell 33, 954–964 (2018).
Wong, S. H. & Yu, J. Gut microbiota in colorectal cancer: mechanisms of action and clinical applications. Nat. Rev. Gastroenterol. Hepatol. 16, 690–704 (2019).
Nakatsu, G. et al. Gut mucosal microbiome across stages of colorectal carcinogenesis. Nat. Commun. 6, 8727 (2015).
Wong, S. H. et al. Gavage of fecal samples from patients with colorectal cancer promotes intestinal carcinogenesis in germ-free and conventional mice. Gastroenterology 153, 1621–1633 (2017).
Liang, Q. et al. Fecal bacteria act as novel biomarkers for noninvasive diagnosis of colorectal cancer. Clin. Cancer Res. 23, 2061–2070 (2017).
Yu, J. et al. Metagenomic analysis of faecal microbiome as a tool towards targeted non-invasive biomarkers for colorectal cancer. Gut 66, 70–78 (2018).
Hill, D. R. & Spence, J. R. Gastrointestinal organoids: understanding the molecular basis of the host-microbe interface. Cell. Mol. Gastroenterol. Hepatol. 3, 138–149 (2016).
Dutta, D. & Clevers, H. Organoid culture systems to study host-pathogen interactions. Curr. Opin. Immunol. 48, 15–22 (2017).
Li, Y. et al. Constitutive TLR4 signalling in intestinal epithelium reduces tumor load by increasing apoptosis in APCMin/+ mice. Oncogene 33, 369–377 (2014).
Peuker, K. et al. Epithelial calcineurin controls microbiota-dependent intestinal tumor development. Nat. Med. 22, 506–515 (2016).
Barker, N. et al. Lgr5+ve stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell 6, 25–36 (2010).
Stange, D. E. et al. Differentiated Troy+ chief cells act as reserve stem cells to generate all lineages of the stomach epithelium. Cell 155, 357–368 (2013).
Bartfeld, S. et al. In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection. Gastroenterology. 148, 126–136 (2015). This study establishes a methodology to develop long-term gastric organoids from human gastric corpus tissues, and assesses the ability of this model to study H. pylori infection or other gastric pathologies.
Schumacher, M. A. et al. The use of murine-derived fundic organoids in studies of gastric physiology. J. Physiol. 593, 1809–1827 (2015). This study describes a novel in vitro system with features of physiological functions by coculturing mouse gastric organoids and immortalized stomach mesenchymal cells.
Chia, N. Y. & Tan, P. Molecular classification of gastric cancer. Ann. Oncol. 27, 763–769 (2016).
Röcken, C. Molecular classification of gastric cancer. Expert Rev. Mol. Diagn. 17, 293–301 (2017).
Hooi, J. K. Y. et al. Global prevalence of Helicobacter pylori infection: systematic review and meta-analysis. Gastroenterology 153, 420–429 (2017).
Eslick, G. D., Lim, L. L., Byles, J. E., Xia, H. H. & Talley, N. J. Association of Helicobacter pylori infection with gastric carcinoma: a meta-analysis. Am. J. Gastroenterol. 94, 2373–2379 (1999).
Uemura, N. et al. Helicobacter pylori infection and the development of gastric cancer. N. Engl. J. Med. 345, 784–789 (2001).
Waskito, L. A., Salama, N. R. & Yamaoka, Y. Pathogenesis of Helicobacter pylori infection. Helicobacter 23, e12516 (2018).
Burkitt, M. D., Duckworth, C. A., Williams, J. M. & Pritchard, D. M. Helicobacter pylori-induced gastric pathology: insights from in vivo and ex vivo models. Dis. Model. Mech. 10, 89–104 (2017).
Schumacher, M. A. et al. Helicobacter pylori-induced sonic hedgehog expression is regulated by NFκB pathway activation: the use of a novel in vitro model to study epithelial response to infection. Helicobacter 20, 19–28 (2015).
Bertaux-Skeirik, N. et al. CD44 plays a functional role in Helicobacter pylori-induced epithelial cell proliferation. PLoS Pathog. 11, e1004663 (2015).
Wroblewski, L. E. et al. Helicobacter pylori targets cancer-associated apical-junctional constituents in gastroids and gastric epithelial cells. Gut 64, 720–730 (2015).
Holokai, L. et al. Increased programmed death-ligand 1 is an early epithelial cell response to Helicobacter pylori infection. PLoS Pathog. 15, e1007468 (2019).
Schlaermann, P. et al. A novel human gastric primary cell culture system for modelling Helicobacter pylori infection in vitro. Gut 65, 202–213 (2016). This study describes a novel in vitro model of H. pylori infection that can recapitulate most of the key hallmarks of infection by utilizing human gastric organoid-derived primary cells.
The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 513, 202–209 (2014).
Nanki, K. et al. Divergent routes toward Wnt and R-spondin niche independency during human gastric carcinogenesis. Cell 174, 856–869 (2018).
Seidlitz, T. et al. Human gastric cancer modelling using organoids. Gut 68, 207–217 (2019). This study establishes human and mouse gastric cancer organoid models that represent the typical characteristics and altered pathways of each of the four subtypes of gastric cancer as published by TCGA Research Network (2014).
DeWard, A. D., Cramer, J. & Lagasse, E. Cellular heterogeneity in the mouse esophagus implicates the presence of a nonquiescent epithelial stem cell population. Cell Rep. 9, 701–711 (2014). This study establishes a methodology to develop long-term oesophageal organoids featuring the stratified squamous epithelium from mouse oesophageal mucosa.
Trisno, S. L. et al. Esophageal organoids from human pluripotent stem cells delineate Sox2 functions during esophageal specification. Cell Stem Cell 23, 501–515 (2018). This study provides evidence that hPSCs can first be induced into definitive endoderm and then into oesophageal organoids by manipulating multiple signalling pathways such as WNT and EGF.
Zhang, Y. et al. 3D modeling of esophageal development using human PSC-derived basal progenitors reveals a critical role for Notch signaling. Cell Stem Cell 23, 516–529 (2018).
Kijima, T. et al. Three-dimensional organoids reveal therapy resistance of esophageal and oropharyngeal squamous cell carcinoma cells. Cell. Mol. Gastroenterol. Hepatol. 7, 73–91 (2019). This study establishes a methodology to develop long-term organoid cultures derived from patients with oesophageal squamous cell carcinoma, and provides evidence that these organoids enable exploration of mechanisms of resistance to therapy.
Li, X. et al. Organoid cultures recapitulate esophageal adenocarcinoma heterogeneity providing a model for clonality studies and precision therapeutics. Nat. Commun. 9, 2983 (2018). This study establishes a methodology to develop long-term organoid cultures derived from patients with oesophageal adenocarcinoma, and provides evidence that these organoids can be used as preclinical tools for precision therapeutics.
Falk, G. W. et al. Barrett’s esophagus: prevalence-incidence and etiology-origins. Ann. N. Y. Acad. Sci. 1232, 1–17 (2011).
Liu, X. et al. Modeling Wnt signaling by CRISPR-Cas9 genome editing recapitulates neoplasia in human Barrett epithelial organoids. Cancer Lett. 436, 109–118 (2018). This study establishes a methodology to develop long-term organoid cultures derived from patients with Barrett oesophagus with histopathological features.
Kasagi, Y., Chandramouleeswaran, P. M. & Whelan, K. A. The esophageal organoid system reveals functional interplay between Notch and cytokines in reactive epithelial changes. Cell. Mol. Gastroenterol. Hepatol. 5, 333–352 (2018).
von Furstenberg, R. J. et al. Porcine esophageal submucosal gland culture model shows capacity for proliferation and differentiation. Cell. Mol. Gastroenterol. Hepatol. 4, 385–404 (2017).
Jiang, M. et al. Transitional basal cells at the squamous-columnar junction generate Barrett’s oesophagus. Nature 550, 529–533 (2017).
Miyajima, A., Tanaka, M. & Itoh, T. Stem/progenitor cells in liver development, homeostasis, regeneration, and reprogramming. Cell Stem Cell 14, 561–574 (2014).
Itoh, T. Stem/progenitor cells in liver regeneration. Hepatology 64, 663–668 (2016).
Yanger, K. et al. Robust cellular reprogramming occurs spontaneously during liver regeneration. Genes Dev. 27, 719–724 (2013).
Yimlamai, D. et al. Hippo pathway activity influences liver cell fate. Cell 157, 1324–1338 (2014).
Tarlow, B. D. et al. Bipotential adult liver progenitors are derived from chronically injured mature hepatocytes. Cell Stem Cell 15, 605–618 (2014).
Katsuda, T., Kawamata, M. & Hagiwara, K. Conversion of terminally committed hepatocytes to culturable bipotent progenitor cells with regenerative capacity. Cell Stem Cell 20, 41–55 (2017).
Huch, M. et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature 494, 247–250 (2013). This study provides evidence that Lgr5 + liver stem cells from mice can give rise to long-term liver organoids with expression of markers of hepatocytes and bile ducts; notably, these organoids differentiate into mature hepatocytes upon transplantation into immunodeficient mice.
Huch, M. et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 160, 299–312 (2015). This study describes a novel in vitro system to generate long-term organoid cultures derived from human bipotent progenitor liver cells that are able to express bile duct-like phenotypes and to differentiate into functional hepatocytes.
Lugli, N. et al. R-spondin 1 and Noggin facilitate expansion of resident stem cells from non-damaged gallbladders. EMBO Rep. 17, 769–779 (2016).
Sampaziotis, F. et al. Reconstruction of the mouse extrahepatic biliary tree using primary human extrahepatic cholangiocyte organoids. Nat. Med. 23, 954–963 (2017).
Vyas, D. et al. Self-assembled liver organoids recapitulate hepatobiliary organogenesis in vitro. Hepatology 67, 750–761 (2017). This study provides evidence that organoids derived from fetal liver progenitor cells are able to recapitulate simultaneous hepatobiliary organogenesis using a single culture medium combination.
Koike, H. et al. Modelling human hepato-biliary-pancreatic organogenesis from the foregut-midgut boundary. Nature 574, 112–116 (2019).
Cazals-Hatem, D. et al. Clinical and molecular analysis of combined hepatocellular-cholangiocarcinomas. J. Hepatol. 41, 292–298 (2004).
Woo, H. G. et al. Identification of a cholangiocarcinoma-like gene expression trait in hepatocellular carcinoma. Cancer Res. 70, 3034–3041 (2010).
Wege, H., Li, J. & Ittrich, H. Treatment lines in hepatocellular carcinoma. Visc. Med. 35, 266–272 (2019).
Broutier Lm et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nat. Med. 23, 1424–1435 (2017). This study establishes a methodology to develop long-term organoid cultures derived from patients with primary liver cancer, and provides evidence that these organoids can recapitulate the histological characteristics, genetic alterations and metastatic features of their originating tumours.
Nuciforo, S. et al. Organoid models of human liver cancers derived from tumor needle biopsies. Cell Rep. 24, 1363–1376 (2018).
Cao, W. et al. Modeling liver cancer and therapy responsiveness using organoids derived from primary mouse liver tumors. Carcinogenesis 40, 145–1540 (2019).
Li, L. et al. Acquisition of cholangiocarcinoma traits during advanced hepatocellular carcinoma development in mice. Am. J. Pathol. 188, 656–671 (2018). This study provides evidence that features of CCA can be acquired during advanced HCC progression by transplanting organoids derived from a transgenic liver cancer mouse model into immunodeficient mice.
Chan, L. H. et al. PRMT6 regulates RAS/RAF binding and MEK/ERK-mediated cancer stemness activities in hepatocellular carcinoma through CRAF methylation. Cell Rep. 25, 690–701 (2018).
Cohen, J. C., Horton, J. D. & Hobbs, H. H. Human fatty liver disease: old questions and new insights. Science 332, 1519–1523 (2011).
Leite, S. B. et al. Novel human hepatic organoid model enables testing of drug-induced liver fibrosis in vitro. Biomaterials 78, 1–10 (2016).
Kruitwagen, H. S. et al. Long-term adult feline liver organoid cultures for disease modeling of hepatic steatosis. Stem Cell Reports 8, 822–830 (2017).
Sendi, H. et al. miR-122 inhibition in a human liver organoid model leads to liver inflammation, necrosis, steatofibrosis and dysregulated insulin signaling. PLoS One 13, e0200847 (2018).
Nie, Y. Z. et al. Recapitulation of hepatitis B virus-host interactions in liver organoids from human induced pluripotent stem cells. EBioMedicine 35, 114–123 (2018). This study describes a novel method to model HBV infection in liver organoids derived from hPSCs that display characteristics of early acute liver failure and fibrosis.
Huch, M. et al. Unlimited in vitro expansion of adult bi-potent pancreas progenitors through the Lgr5/R-spondin axis. EMBO J. 32, 2708–2721 (2013).This study provides evidence that isolated mouse pancreatic ductal cells can generate long-term organoid cultures that can differentiate along ductal and endocrine lineages upon transplantation into immunodeficient mice.
Boj, S. F. et al. Organoid models of human and mouse ductal pancreatic cancer. Cell 160, 324–338 (2015). This study establishes a methodology to develop long-term organoid cultures derived from pancreatic tissues of healthy individuals and patients with PDAC; notably, the tumour-derived organoids can recapitulate features of pancreatic intraepithelial neoplasia, whereas organoids derived from normal tissues give rise to ductal and endocrine structures upon transplantation into immunodeficient mice.
Huang, L. et al. Ductal pancreatic cancer modeling and drug screening using human pluripotent stem cell- and patient-derived tumor organoids. Nat. Med. 21, 1364–1371 (2015). This study describes a novel method to generate pancreatic organoids from hPSCs that have features of early stages of transformation upon introducing mutations into KRAS and TP53 in these organoids.
Greggio, C. et al. Artificial three-dimensional niches deconstruct pancreas development in vitro. Development 140, 4452–4462 (2013).
Dorrell, C. et al. The organoid-initiating cells in mouse pancreas and liver are phenotypically and functionally similar. Stem Cell Res. 13, 275–283 (2014).
Ryan, D. P., Hong, T. S. & Bardeesy, N. Pancreatic adenocarcinoma. N. Engl. J. Med. 371, 1039–1049 (2014).
Kanda, M. et al. Presence of somatic mutations in most early-stage pancreatic intraepithelial neoplasia. Gastroenterology 142, 730–733 (2012).
Morris, J. P. 4th, Wang, S. C. & Hebrok, M. KRAS, hedgehog, Wnt and the twisted developmental biology of pancreatic ductal adenocarcinoma. Nat. Rev. Cancer 10, 683–695 (2010).
Yachida, S. et al. Clinical significance of the genetic landscape of pancreatic cancer and implications for identification of potential long-term survivors. Clin. Cancer Res. 18, 6339–6347 (2012).
Li, X. et al. Oncogenic transformation of diverse gastrointestinal tissues in primary organoid culture. Nat. Med. 20, 769–777 (2014).
Lee, J. et al. Reconstituting development of pancreatic intraepithelial neoplasia from primary human pancreas duct cells. Nat. Commun. 8, 14686 (2017). Using CRISPR–Cas9 genome editing, overexpression of four driver genes of PDAC — KRAS, CDKN2A, TP53 and SMAD4 — in human pancreatic organoids is used to model carcinogenesis.
Seino, T. et al. Human pancreatic tumor organoids reveal loss of stem cell niche factor dependence during disease progression. Cell Stem Cell 22, 454–467 (2018). A study similar to Lee et al. (2017) except mutations are knocked into organoids rather than being overexpressed; both studies provide evidence that progression from pancreatic intraepithelial neoplasia to adenocarcinoma can be recapitulated accurately in organoids using CRISPR–Cas9 technology.
Chio, I. I. C. et al. NRF2 promotes tumor maintenance by modulating mRNA translation in pancreatic cancer. Cell 166, 963–976 (2016).
Roe, J. S. et al. Enhancer reprogramming promotes pancreatic cancer metastasis. Cell 170, 875–888 (2017).
Sailaja, B. S., He, X. C. & Li, L. The regulatory niche of intestinal stem cells. J. Physiol. 594, 4827–4836 (2016).
Yuan, Y., Jiang, Y. C., Sun, C. K. & Chen, Q. M. Role of the tumor microenvironment in tumor progression and the clinical applications (Review). Oncol. Rep. 35, 2499–2515 (2016).
Bu, L. et al. Biological heterogeneity and versatility of cancer-associated fibroblasts in the tumor microenvironment. Oncogene 38, 4887–4901 (2019).
Takahashi, Y. et al. Reciprocal inflammatory signaling between intestinal epithelial cells and adipocytes in the absence of immune cells. EBioMedicine 23, 34–45 (2017).
Nozaki, K. et al. Co-culture with intestinal epithelial organoids allows efficient expansion and motility analysis of intraepithelial lymphocytes. J. Gastroenterol. 51, 206–213 (2016).
Jung, K. B. et al. Interleukin-2 induces the in vitro maturation of human pluripotent stem cell-derived intestinal organoids. Nat. Commun. 9, 3039 (2018).
Hahn, S. et al. Organoid-based epithelial to mesenchymal transition (OEMT) model: from an intestinal fibrosis perspective. Sci. Rep. 7, 2435 (2017).
Trumpi, K. et al. Macrophages induce “budding” in aggressive human colon cancer subtypes by protease-mediated disruption of tight junctions. Oncotarget 9, 19490–19507 (2018).
Hirokawa, Y., Yip, K. H., Tan, C. W. & Burgess, A. W. Colonic myofibroblast cell line stimulates colonoid formation. Am. J. Physiol. Gastrointest. Liver Physiol. 306, G547–G556 (2014).
Pastuła, A. et al. Three-dimensional gastrointestinal organoid culture in combination with nerves or fibroblasts: a method to characterize the gastrointestinal stem cell niche. Stem Cell Int. 2016, 3710836 (2016).
Neal, J. T. et al. Organoid modeling of the tumor immune microenvironment. Cell 175, 1972–1988 (2018). This study describes a novel method for culturing PDOs with preservation of the stromal architecture, T cell receptor spectrum, immune checkpoint system and functional lymphocytes.
Chakrabarti, J. et al. Hedgehog signaling induces PD-L1 expression and tumor cell proliferation in gastric cancer. Oncotarget 9, 37439–37457 (2018).
Wen, Y. A. et al. Adipocytes activate mitochondrial fatty acid oxidation and autophagy to promote tumor growth in colon cancer. Cell Death Dis. 8, e2593 (2017).
Wang, Y. et al. Microenvironment of a tumor-organoid system enhances hepatocellular carcinoma malignancy-related hallmarks. Organogenesis 13, 83–94 (2017). This study provides evidence that the TME can enhance the malignancy of HCC by coculturing HCC organoids with endothelial cells and fibroblasts.
Öhlund, D. et al. Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J. Exp. Med. 214, 579–596 (2017). This study provides evidence of the presence of heterogeneity in CAFs by coculturing pancreatic stellate cells and organoids derived from a PDAC mouse model.
Biffi, G. et al. IL1-induced JAK/STAT signaling is antagonized by TGFβ to shape CAF heterogeneity in pancreatic ductal adenocarcinoma. Cancer Discov. 9, 282–301 (2019).
Genovese, L. et al. Cellular localization, invasion, and turnover are differently influenced by healthy and tumor-derived extracellular matrix. Tissue Eng. Part. A 20, 2005–2018 (2014).
Romero-López, M. et al. Recapitulating the human tumor microenvironment: colon tumor-derived extracellular matrix promotes angiogenesis and tumor cell growth. Biomaterials 116, 118–129 (2017).
Cukierman, E., Pankov, R., Stevens, D. R. & Yamada, K. M. Taking cell-matrix adhesions to the third dimension. Science 294, 1708–1712 (2001).
Hidalgo, M. et al. Patient-derived xenograft models: an emerging platform for translational cancer research. Cancer Discov. 4, 998–1013 (2014).
Weeber, F., Ooft, S. N., Dijkstra, K. K. & Voest, E. E. Tumor organoids as a pre-clinical cancer model for drug discovery. Cell Chem. Biol. 24, 1092–1100 (2017).
Tiriac, H. et al. Successful creation of pancreatic cancer organoids by means of EUS-guided fine-needle biopsy sampling for personalized cancer treatment. Gastrointest. Endosc. 87, 1474–1480 (2018).
Gao, M. et al. Development of patient-derived gastric cancer organoids from endoscopic biopsies and surgical tissues. Ann. Surg. Oncol. 25, 2767–2775 (2018).
Skardal, A., Devarasetty, M., Rodman, C., Atala, A. & Soker, S. Liver-tumor hybrid organoids for modeling tumor growth and drug response in vitro. Ann. Biomed. Eng. 43, 2361–2373 (2015).
Roy, P. et al. Organoids as preclinical models to improve intraperitoneal chemotherapy effectiveness for colorectal cancer patients with peritoneal metastases: preclinical models to improve HIPEC. Int. J. Pharm. 531, 143–152 (2017).
Sgodda, M. et al. A scalable approach for the generation of human pluripotent stem cell-derived hepatic organoids with sensitive hepatotoxicity features. Stem Cell Dev. 26, 1490–1504 (2017).
Crespo, M. et al. Colonic organoids derived from human induced pluripotent stem cells for modeling colorectal cancer and drug testing. Nat. Med. 23, 878–884 (2017).
Sharifnia, T., Hong, A. L., Painter, C. A. & Boehm, J. S. Emerging opportunities for target discovery in rare cancers. Cell Chem. Biol. 24, 1075–1091 (2017).
Haugabook, S. J., Ferrer, M. & Ottinger, E. A. In vitro and in vivo translational models for rare liver diseases. Biochim. Biophys. Acta Mol. Basis Dis. 1865, 1003–1018 (2018).
Dekkers, J. F. et al. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat. Med. 19, 939–945 (2013).
Dekkers, J. F. et al. Characterizing responses to CFTR-modulating drugs using rectal organoids derived from subjects with cystic fibrosis. Sci. Transl. Med. 8, 344ra84 (2016).
Pauli, C. et al. Personalized in vitro and in vivo cancer models to guide precision medicine. Cancer Discov. 7, 462–477 (2017). This study provides evidence that histopathological features of the native tumours can be conserved in PDOs and PDXs.
Tiriac, H. et al. Organoid profiling identifies common responders to chemotherapy in pancreatic cancer. Cancer Discov. 8, 1112–1129 (2018). This study performs therapeutic profiling to identify chemotherapy responsiveness and potential treatments against chemorefractory organoids derived from patients with PDAC.
Kondo, J. et al. High-throughput screening in colorectal cancer tissue-originated spheroids. Cancer Sci. 110, 345–355 (2019). This study performs high-throughput screening of a panel of 2,427 drugs on PDOs with subsequent validation of the responsiveness of ‘hit’ drugs using a biobank of PDOs.
Yan, H. H. N. et al. A comprehensive human gastric cancer organoid biobank captures tumor subtype heterogeneity and enables therapeutic screening. Cell Stem Cell 23, 882–897 (2018).
Li, L. et al. Human primary liver cancer organoids reveal intratumor and interpatient drug response heterogeneity. JCI Insight 4, 121490 (2019).
Aberle, M. R. et al. Patient-derived organoid models help define personalized management of gastrointestinal cancer. Br. J. Surg. 105, e48–e60 (2018).
Vlachogiannis, G. et al. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science 359, 920–926 (2018). This study is the first to demonstrate the predictive value of PDOs in personalized medicine using a biobank of organoids derived from patients with metastatic CRC and gastro-oesophageal cancer.
Steele, N. G. et al. An organoid-based preclinical model of human gastric cancer. Cell. Mol. Gastroenterol. Hepatol. 7, 161–184 (2019).
Ganesh, K. et al. A rectal cancer organoid platform to study individual responses to chemoradiation. Nat. Med. 25, 1607–1614 (2019).
Ooft, S. N. et al. Patient-derived organoids can predict response to chemotherapy in metastatic colorectal cancer patients. Sci. Transl. Med. 11, pii: eaay2574 (2019).
Yao, Y. et al. Patient-derived organoids predict chemoradiation responses of locally advanced rectal cancer. Cell Stem Cell 26, 17–26.e6 (2019). This study is the first to demonstrate that PDOs could enable tailoring of the treatment approach of chemoradiation or chemotherapy to improve clinical outcomes and reduce toxicities in patients with CRC.
Schumacher, D. et al. Heterogeneous pathway activation and drug response modelled in colorectal-tumor-derived 3D cultures. PLoS Genet. 15, e1008076 (2019). This study is an example of how basic research in PDOs can link to personalized medicine by evaluating the influence of intratumoural heterogeneity on drug response.
Gjorevski, N. et al. Designer matrices for intestinal stem cell and organoid culture. Nature 539, 560–564 (2016). This study establishes a minimal environment for organoid cultures and identifies key parameters in the ECM that govern intestinal organoid formation.
Candiello, J. et al. 3D heterogeneous islet organoid generation from human embryonic stem cells using a novel engineered hydrogel platform. Biomaterials 177, 27–39 (2018).
Ootani, A. et al. Sustained in vitro intestinal epithelial culture within a Wnt-dependent stem cell niche. Nat. Med. 15, 701–706 (2009).
Wang, Y. et al. Formation of human colonic crypt array by application of chemical gradients across a shaped epithelial monolayer. Cell. Mol. Gastroenterol. Hepatol. 5, 113–130 (2017).
Wallaschek, N. et al. Establishing pure cancer organoid cultures: identification, selection and verification of cancer phenotypes and genotypes. J. Mol. Biol. 431, 2884–2893 (2019). This study establishes multiple step-by-step protocols for obtaining pure populations of human gastric cancer organoids with subsequent validation of cancer identity.
Jackstadt, R. & Sansom, O. J. Mouse models of intestinal cancer. J. Pathol. 238, 141–151 (2016).
Oh, B. Y., Hong, H. K., Lee, W. Y. & Cho, Y. B. Animal models of colorectal cancer with liver metastasis. Cancer Lett. 387, 114–120 (2017).
Bhatia, S. N. & Ingber, D. E. Microfluidic organs-on-chips. Nat. Biotechnol. 32, 760–772 (2014).
Mittal, R. et al. Organ-on-chip models: implications in drug discovery and clinical applications. J. Cell Physiol. 234, 8352–8380 (2019).
Mattei, F. et al. A multidisciplinary study using in vivo tumor models and microfluidic cell-on-chip approach to explore the cross-talk between cancer and immune cells. J. Immunotoxicol. 11, 337–346 (2014).
Agliari, E. et al. Cancer-driven dynamics of immune cells in a microfluidic environment. Sci. Rep. 4, 6639 (2014).
Liu, P. F. et al. A bladder cancer microenvironment simulation system based on a microfluidic co-culture model. Oncotarget 6, 37695–37705 (2015).
de Souza, N. Organoid culture. Nat. Methods 14, 35 (2017).
Acknowledgements
The authors thank C. H. Szeto for comments on the structure and content of the manuscript.
Author information
Authors and Affiliations
Contributions
H.C.H.L. researched data for the article, made a substantial contribution to discussion of content, designed the figures, and wrote the article. O.K. and H.X. made a substantial contribution to discussion of content, and reviewed/edited the manuscript before submission. J.Y. researched data for the article, made a substantial contribution to discussion of content, wrote the article, and reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks Y. Zavros, H. Tiriac, D. Stange and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Lau, H.C.H., Kranenburg, O., Xiao, H. et al. Organoid models of gastrointestinal cancers in basic and translational research. Nat Rev Gastroenterol Hepatol 17, 203–222 (2020). https://doi.org/10.1038/s41575-019-0255-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-019-0255-2
This article is cited by
-
Drug repurposing for cancer therapy
Signal Transduction and Targeted Therapy (2024)
-
Advances and Applications of Cancer Organoids in Drug Screening and Personalized Medicine
Stem Cell Reviews and Reports (2024)
-
Progress, application and challenges of liver organoids
Clinical Cancer Bulletin (2024)
-
Metal nanoparticles as a potential technique for the diagnosis and treatment of gastrointestinal cancer: a comprehensive review
Cancer Cell International (2023)
-
Metal-enriched HSP90 nanoinhibitor overcomes heat resistance in hyperthermic intraperitoneal chemotherapy used for peritoneal metastases
Molecular Cancer (2023)