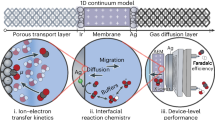
Abstract
New technologies are required to electrocatalytically convert carbon dioxide (CO2) into fuels and chemicals at near-ambient temperatures and pressures more effectively. One particular challenge is mediating the electrochemical CO2 reduction reaction (CO2RR) at low cell voltages while maintaining high conversion efficiencies. Anion exchange membranes (AEMs) in zero-gap reactors offer promise in this direction; however, there remain substantial obstacles to be overcome in tailoring the membranes and other cell components to the requirements of CO2RR systems. Here we review recent advances, and remaining challenges, in AEM materials and devices for CO2RR. We discuss the principles underpinning AEM operation and the properties desired for CO2RR, in addition to reviewing state-of-the-art AEMs in CO2 electrolysers. We close with future design strategies to minimize product crossover, improve mechanical and chemical stability, and overcome the energy losses associated with the use of AEMs for CO2RR systems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Weekes, D. M., Salvatore, D. A., Reyes, A., Huang, A. & Berlinguette, C. P. Electrolytic CO2 reduction in a flow cell. Acc. Chem. Res. 51, 910–918 (2018).
Jouny, M., Luc, W. & Jiao, F. General techno-economic analysis of CO2 electrolysis systems. Ind. Eng. Chem. Res. 57, 2165–2177 (2018).
Mehta, V. & Cooper, J. S. Review and analysis of PEM fuel cell design and manufacturing. J. Power Sources 114, 32–53 (2003).
Millet, P. et al. PEM water electrolyzers: from electrocatalysis to stack development. Int. J. Hydrogen Energy 35, 5043–5052 (2010).
Kutz, R. B. et al. Sustainion imidazolium-functionalized polymers for carbon dioxide electrolysis. Energy Technol. 5, 929–936 (2017). Research paper that describes a Sustainion AEM CO2 electrolyser for CO production and boasts 1,000 h reaction stability.
Delacourt, C., Ridgway, P. L., Kerr, J. B. & Newman, J. Design of an electrochemical cell making syngas (CO + H2) from CO2 and H2O reduction at room temperature. J. Electrochem. Soc. 155, B42–B49 (2008).
Salvatore, D. A. et al. Electrolysis of gaseous CO2 to CO in a flow cell with a bipolar membrane. ACS Energy Lett. 3, 149–154 (2018).
Herranz, J., Patru, A., Fabbri, E. & Schmidt, T. J. Co-electrolysis of CO2 and H2O: from electrode reactions to cell level development. Curr. Opin. Electrochem. https://doi.org/10.1016/j.coelec.2020.05.004 (2020).
Ainscough, C., Peterson, D. & Miller, E. DOE Hydrogen And Fuel Cells Program Record 14004 Technical Report 7 (US DOE, 2014).
Pătru, A., Binninger, T., Pribyl, B. & Schmidt, T. J. Design principles of bipolar electrochemical co-electrolysis cells for efficient reduction of carbon dioxide from gas phase at low temperature. J. Electrochem. Soc. 166, F34–F43 (2019).
Ren, S. et al. Molecular electrocatalysts can mediate fast, selective CO2 reduction in a flow cell. Science 365, 367–369 (2019).
Wang, M. et al. CO2 electrochemical catalytic reduction with a highly active cobalt phthalocyanine. Nat. Commun. 10, 3602 (2019).
Gabardo, C. M. et al. Continuous carbon dioxide electroreduction to concentrated multi-carbon products using a membrane electrode assembly. Joule 3, 2777–2791 (2019). Research paper that describes an AEM CO2 electrolyser for producing C2+ products, ethylene and concentrated ethanol, and discusses the challenges with product crossover.
Oener, S. Z., Ardo, S. & Boettcher, S. W. Ionic processes in water electrolysis: the role of ion-selective membranes. ACS Energy Lett. 2, 2625–2634 (2017).
Aeshala, L. M., Uppaluri, R. & Verma, A. Electrochemical conversion of CO2 to fuels: tuning of the reaction zone using suitable functional groups in a solid polymer electrolyte. Phys. Chem. Chem. Phys. 16, 17588–17594 (2014).
Merle, G., Wessling, M. & Nijmeijer, K. Anion exchange membranes for alkaline fuel cells: a review. J. Membr. Sci. 377, 1–35 (2011).
Holdcroft, S. & Fan, J. Sterically-encumbered ionenes as hydroxide ion-conducting polymer membranes. Curr. Opin. Electrochem. 18, 99–105 (2019).
Varcoe, J. R. et al. Anion-exchange membranes in electrochemical energy systems. Energy Environ. Sci. 7, 3135–3191 (2014). Perspective paper that discusses the key concepts, misconceptions, technological and scientific limitations, and research priorities for anion exchange membranes in electrochemical devices.
Ramaswamy, N. & Mukerjee, S. Alkaline anion-exchange membrane fuel cells: challenges in electrocatalysis and interfacial charge transfer. Chem. Rev. 119, 11945–11979 (2019).
Gottesfeld, S. et al. Anion exchange membrane fuel cells: current status and remaining challenges. J. Power Sources 375, 170–184 (2018).
Noh, S., Jeon, J. Y., Adhikari, S., Kim, Y. S. & Bae, C. Molecular engineering of hydroxide conducting polymers for anion exchange membranes in electrochemical energy conversion technology. Acc. Chem. Res. 52, 2745–2755 (2019).
Yin, Z. et al. An alkaline polymer electrolyte CO2 electrolyzer operated with pure water. Energy Environ. Sci. 12, 2455–2246 (2019). Research paper that describes a QAPPT AEM CO2 electrolyser for CO production that uses pure water as an electrolyte.
Zawodzinski, T. A., Neeman, M., Sillerud, L. O. & Gottesfeld, S. Determination of water diffusion coefficients in perfluorosulfonate ionomeric membranes. J. Phys. Chem. 95, 6040–6044 (1991).
Tuckerman, M. E., Marx, D. & Parrinello, M. The nature and transport mechanism of hydrated hydroxide ions in aqueous solution. Nature 417, 925–929 (2002).
Kamcev, J. & Freeman, B. D. Charged polymer membranes for environmental/energy applications. Annu. Rev. Chem. Biomol. Eng. 7, 111–133 (2016).
Lee, K. M., Wycisk, R., Litt, M. & Pintauro, P. N. Alkaline fuel cell membranes from xylylene block ionenes. J. Membr. Sci. 383, 254–261 (2011).
Zheng, Y. et al. Water uptake study of anion exchange membranes. Macromolecules 51, 3264–3278 (2018).
Mangiagli, P. M., Ewing, C. S., Xu, K., Wang, Q. & Hickner, M. A. Dynamic water uptake of flexible ion-containing polymer networks. Fuel Cells 9, 432–438 (2009).
Li, Y. S., Zhao, T. S. & Chen, R. Cathode flooding behaviour in alkaline direct ethanol fuel cells. J. Power Sources 196, 133–139 (2011).
Zawodzinski, T. A., Davey, J., Valerio, J. & Gottesfeld, S. The water content dependence of electro-osmotic drag in proton-conducting polymer electrolytes. Electrochim. Acta 40, 297–302 (1995).
Xu, C. & Zhao, T. S. In situ measurements of water crossover through the membrane for direct methanol fuel cells. J. Power Sources 168, 143–153 (2007).
Liu, F., Lu, G. & Wang, C.-Y. Low crossover of methanol and water through thin membranes in direct methanol fuel cells. J. Electrochem. Soc. 153, A543–A553 (2006).
Zawodzinskijr, T., Springer, T., Uribe, F. & Gottesfeld, S. Characterization of polymer electrolytes for fuel cell applications. Solid State Ion. 60, 199–211 (1993).
Ziv, N. & Dekel, D. R. A practical method for measuring the true hydroxide conductivity of anion exchange membranes. Electrochem. Commun. 88, 109–113 (2018).
Larrazábal, G. O. et al. Analysis of mass flows and membrane cross-over in CO2 reduction at high current densities in an MEA-type electrolyzer. ACS Appl. Mater. Interfaces 11, 41281–41288 (2019). Research paper that analyses the mass balance in an AEM CO2 electrolyser and discusses CO2 crossover via carbonate formation.
Lu, X. et al. In-situ observation of the pH gradient near the gas diffusion electrode of CO2 reduction in alkaline electrolyte. J. Am. Chem. Soc. https://doi.org/10.1021/jacs.0c06779 (2020).
Pan, J. et al. High-performance alkaline polymer electrolyte for fuel cell applications. Adv. Funct. Mater. 20, 312–319 (2010).
Thieu, L. M., Zhu, L., Korovich, A. G., Hickner, M. A. & Madsen, L. A. Multiscale tortuous diffusion in anion and cation exchange membranes. Macromolecules 52, 24–35 (2019).
Disabb-Miller, M. L., Johnson, Z. D. & Hickner, M. A. Ion motion in anion and proton-conducting triblock copolymers. Macromolecules 46, 949–956 (2013).
Kim, Y. S. & Pivovar, B. S. Moving beyond mass-based parameters for conductivity analysis of sulfonated polymers. Annu. Rev. Chem. Biomol. Eng. 1, 123–148 (2010).
Dischinger, S. M., Gupta, S., Carter, B. M. & Miller, D. J. Transport of neutral and charged solutes in imidazolium-functionalized poly(phenylene oxide) membranes for artificial photosynthesis. Ind. Eng. Chem. Res. 59, 5257–5266 (2019).
Krödel, M. et al. Rational design of ion exchange membrane material properties limits the crossover of CO2 reduction products in artificial photosynthesis devices. ACS Appl. Mater. Interfaces 12, 12030–12042 (2020).
Nishimura, Y. et al. Solid polymer electrolyte CO2 reduction. Energy Convers. Manag. 36, 629–632 (1995).
Narayanan, S. R., Haines, B., Soler, J. & Valdez, T. I. Electrochemical conversion of carbon dioxide to formate in alkaline polymer electrolyte membrane cells. J. Electrochem. Soc. 158, A167–A173 (2011).
Aeshala, L. M., Rahman, S. U. & Verma, A. Effect of solid polymer electrolyte on electrochemical reduction of CO2. Sep. Purif. Technol. 94, 131–137 (2012).
Li, Y. C. et al. Bipolar membranes inhibit product crossover in CO2 electrolysis cells. Adv. Sustain. Syst. 2, 1700187 (2018).
Reyes, A. et al. Managing hydration at the cathode enables efficient CO2 electrolysis at commercially relevant current densities. ACS Energy Lett. 5, 1612–1618 (2020).
Weng, L.-C., Bell, A. T. & Weber, A. Z. Towards membrane-electrode assembly systems for CO2 reduction: a modeling study. Energy Environ. Sci. 12, 1950–1968 (2019).
Leonard, M. E., Clarke, L. E., Forner-Cuenca, A., Brown, S. M. & Brushett, F. R. Investigating electrode flooding in a flowing electrolyte, gas-fed carbon dioxide electrolyzer. ChemSusChem 89, 400–411 (2019).
Dinh, C.-T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Salvatore, D. & Berlinguette, C. P. Voltage matters when reducing CO2 in an electrochemical flow cell. ACS Energy Lett. 5, 215–220 (2020). Perspective paper that employs a reference electrode in situ of zero-gap CO2 electrolysers to measure voltage drops across membrane electrode assembly components.
Liu, Z. et al. Electrochemical generation of syngas from water and carbon dioxide at industrially important rates. J. CO2 Util. 15, 50–56 (2016).
Liu, Z., Yang, H., Kutz, R. & Masel, R. I. CO2 electrolysis to CO and O2 at high selectivity, stability and efficiency using sustainion membranes. J. Electrochem. Soc. 165, J3371 (2018).
Jiang, K. et al. Isolated Ni single atoms in graphene nanosheets for high-performance CO2 reduction. Energy Environ. Sci. https://doi.org/10.1039/C7EE03245E (2018).
Zheng, T. et al. Large-scale and highly selective CO2 electrocatalytic reduction on nickel single-atom catalyst. Joule 3, 265–278 (2019).
Peng, H. et al. Alkaline polymer electrolyte fuel cells stably working at 80 °C. J. Power Sources 390, 165–167 (2018).
Park, E. J. & Kim, Y. S. Quaternized aryl ether-free polyaromatics for alkaline membrane fuel cells: synthesis, properties, and performance — a topical review. J. Mater. Chem. A 6, 15456–15477 (2018).
Luo, X., Rojas-Carbonell, S., Yan, Y. & Kusoglu, A. Structure–transport relationships of poly(aryl piperidinium) anion-exchange membranes: effect of anions and hydration. J. Membr. Sci. 598, 117680 (2019).
Lee, J., Lim, J., Roh, C.-W., Whang, H. S. & Lee, H. Electrochemical CO2 reduction using alkaline membrane electrode assembly on various metal electrodes. J. CO2 Util. 31, 244–250 (2019).
Arquer, F. P. G. et al. CO2 electrolysis to multicarbon products at activities greater than 1 A cm−2. Science 367, 661–666 (2020).
Xu, Y. et al. Oxygen-tolerant electroproduction of C2 products from simulated flue gas. Energy Environ. Sci. 13, 554–561 (2020).
Khadke, P. S. & Krewer, U. Mass-transport characteristics of oxygen at Pt/anion exchange ionomer interface. J. Phys. Chem. C. 118, 11215–11223 (2014).
Zhang, T. et al. Nickel–nitrogen–carbon molecular catalysts for high rate CO2 electro-reduction to CO: on the role of carbon substrate and reaction chemistry. ACS Appl. Energy Mater. 3, 1617–1626 (2020).
Carter, B. M., Keller, L., Wessling, M. & Miller, D. J. Preparation and characterization of crosslinked poly(vinylimidazolium) anion exchange membranes for artificial photosynthesis. J. Mater. Chem. A https://doi.org/10.1039/C9TA00498J (2019).
Cui, L. et al. Novel sulfonated poly(ether ether ketone)/silica coated carbon nanotubes high-performance composite membranes for direct methanol fuel cell. Polym. Adv. Technol. 26, 457–464 (2015).
Su, Y.-H. et al. Proton exchange membranes modified with sulfonated silica nanoparticles for direct methanol fuel cells. J. Membr. Sci. 296, 21–28 (2007).
Si, K. et al. Rigid-rod poly(phenylenesulfonic acid) proton exchange membranes with cross-linkable biphenyl groups for fuel cell applications. Macromolecules 46, 422–433 (2013).
Weissbach, T. et al. Simultaneous, synergistic control of ion exchange capacity and cross-linking of sterically-protected poly(benzimidazolium)s. Chem. Mater. 28, 8060–8070 (2016).
Zhao, Y. et al. High-performance alkaline fuel cells using crosslinked composite anion exchange membrane. J. Power Sources 221, 247–251 (2013).
Katzfuß, A. et al. Methylated polybenzimidazole and its application as a blend component in covalently cross-linked anion-exchange membranes for DMFC. J. Membr. Sci. 465, 129–137 (2014).
Park, A. M., Turley, F. E., Wycisk, R. J. & Pintauro, P. N. Electrospun and cross-linked nanofiber composite anion exchange membranes. Macromolecules 47, 227–235 (2014).
Weng, L.-C., Bell, A. T. & Weber, A. Z. Modeling gas-diffusion electrodes for CO2 reduction. Phys. Chem. Chem. Phys. 20, 16973–16984 (2018).
Mohanty, A. D., Tignor, S. E., Krause, J. A., Choe, Y.-K. & Bae, C. Systematic alkaline stability study of polymer backbones for anion exchange membrane applications. Macromolecules 49, 3361–3372 (2016).
Arges, C. G. & Ramani, V. Investigation of cation degradation in anion exchange membranes using multi-dimensional NMR spectroscopy. J. Electrochem. Soc. 160, F1006–F1021 (2013).
Parrondo, J., Wang, Z., Jung, M.-S. J. & Ramani, V. Reactive oxygen species accelerate degradation of anion exchange membranes based on polyphenylene oxide in alkaline environments. Phys. Chem. Chem. Phys. 18, 19705–19712 (2016).
Klingele, M. et al. A completely spray-coated membrane electrode assembly. Electrochem. Commun. 70, 65–68 (2016).
Breitwieser, M., Klingele, M., Vierrath, S., Zengerle, R. & Thiele, S. Tailoring the membrane–electrode interface in PEM fuel cells: a review and perspective on novel engineering approaches. Adv. Energy Mater. 8, 1701257 (2018). Review and perspective paper on engineering the membrane–electrode interface to increase efficiency for PEM fuel cells; many of the same principles can be carried over to CO2 electrolysers.
Ito, H. et al. Experimental investigation of electrolytic solution for anion exchange membrane water electrolysis. Int. J. Hydrogen Energy 43, 17030–17039 (2018).
Li, J., Li, X., Gunathunge, C. M. & Waegele, M. M. Hydrogen bonding steers the product selectivity of electrocatalytic CO reduction. Proc. Natl Acad. Sci. USA 116, 9220–9229 (2019).
Ovalle, V. J. & Waegele, M. M. Understanding the impact of N-arylpyridinium ions on the selectivity of CO2 reduction at the Cu/electrolyte interface. J. Phys. Chem. C 123, 24453–24460 (2019).
Aeshala, L. M., Uppaluri, R. G. & Verma, A. Effect of cationic and anionic solid polymer electrolyte on direct electrochemical reduction of gaseous CO2 to fuel. J. CO2 Util. 3-4, 49–55 (2013).
Hou, P., Wang, X., Wang, Z. & Kang, P. Gas phase electrolysis of carbon dioxide to carbon monoxide using nickel nitride as the carbon enrichment catalyst. ACS Appl. Mater. Interfaces 10, 38024–38031 (2018).
Ma, C. et al. Carbon nanotubes with rich pyridinic nitrogen for gas phase CO2 electroreduction. Appl. Catal. B 250, 347–354 (2019).
Ju, W. et al. Electrocatalytic reduction of gaseous CO2 to CO on Sn/Cu-nanofiber-based gas diffusion electrodes. Adv. Energy Mater. 9, 1901514 (2019).
Endrődi, B. et al. Multilayer electrolyzer stack converts carbon dioxide to gas products at high pressure with high efficiency. ACS Energy Lett. 4, 1770–1777 (2019).
Jeong, H.-Y. et al. Achieving highly efficient CO2 to CO electroreduction exceeding 300 mA cm−2 with single-atom nickel electrocatalysts. J. Mater. Chem. A 7, 10651–10661 (2019).
Zhu, W., Kattel, S., Jiao, F. & Chen, J. G. Shape-controlled CO2 electrochemical reduction on nanosized Pd hydride cubes and octahedra. Adv. Energy Mater. 9, 1802840 (2019).
Wang, Y. et al. Catalyst synthesis under CO2 electroreduction favours faceting and promotes renewable fuels electrosynthesis. Nat. Catal. 3, 98–106 (2020).
Li, F. et al. Molecular tuning of CO2-to-ethylene conversion. Nature 577, 509–513 (2020).
Kaczur, J. J., Yang, H., Liu, Z., Sajjad, S. D. & Masel, R. I. Carbon dioxide and water electrolysis using new alkaline stable anion membranes. Front. Chem. 6, 263 (2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Energy thanks Zengcai Liu, Guenter Schmid and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Source Data Fig. 3
Detailed data and corresponding references for Fig. 3.
Rights and permissions
About this article
Cite this article
Salvatore, D.A., Gabardo, C.M., Reyes, A. et al. Designing anion exchange membranes for CO2 electrolysers. Nat Energy 6, 339–348 (2021). https://doi.org/10.1038/s41560-020-00761-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-020-00761-x
This article is cited by
-
Exploring CO2 reduction and crossover in membrane electrode assemblies
Nature Chemical Engineering (2024)
-
An organic proton cage that is ultra-resistant to hydroxide-promoted degradation
Nature Communications (2024)
-
High Surface Area Assemblies of Gold Nanoparticles on Hydrophilic Carbon Fiber Paper with Ionomer Overlayers for Aqueous CO2 Reduction Electrocatalysis to Clean Syngas
Topics in Catalysis (2024)
-
Electrochemical Carbon Dioxide Reduction in Acidic Media
Electrochemical Energy Reviews (2024)
-
Designing Membrane Electrode Assembly for Electrochemical CO2 Reduction: a Review
Transactions of Tianjin University (2024)