Abstract
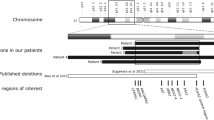
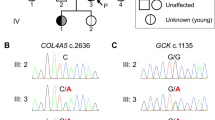
Severe insulin resistance syndromes result from primary insulin signaling defects, adipose tissue abnormalities or other complex syndromes. Mutations in TBC1D4 lead to partial insulin signaling defects, characterized mainly by postprandial insulin resistance. We describe an individual with severe insulin-resistant diabetes unresponsive to multiple therapies, in whom exome and genome analyses identified a complex rearrangement in TBC1D4. The rearrangement was of the pattern DUP-TRP/INV-DUP, with mutational signatures suggestive of replicative repair and Alu-Alu recombination as the underlying mechanisms. TBC1D4 encodes the TBC1D4/AS160 RabGTPase activating protein (RabGAP) involved in the translocation of glucose transporter 4 (GLUT4) from the cytosol to the cell membrane. Although the precise functional mechanism underlying insulin resistance in the proband is yet to be determined, this case provides further support for the link between TBC1D4 and hereditary insulin-resistant diabetes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data generated in this study are found in the published article. Additional data are available from the corresponding author on reasonable request.
References
Bonnefond A, Unnikrishnan R, Doria A, Vaxillaire M, Kulkarni RN, Mohan V, et al. Monogenic diabetes. Nat Rev Dis Prim. 2023;9:12.
Samadli S, Zhou Q, Zheng B, Gu W, Zhang A. From glucose sensing to exocytosis: takes from maturity onset diabetes of the young. Front Endocrinol (Lausanne). 2023;14:1188301.
Greeley SAW, Polak M, Njølstad PR, Barbetti F, Williams R, Castano L, et al. ISPAD Clinical Practice Consensus Guidelines 2022: the diagnosis and management of monogenic diabetes in children and adolescents. Pediatr Diabetes. 2022;23:1188–211.
Angelidi AM, Filippaios A, Mantzoros CS. Severe insulin resistance syndromes. J Clin Invest. 2021;131:e142245.
Iqbal J, Jiang HL, Wu HX, Li L, Zhou YH, Hu N, et al. Hereditary severe insulin resistance syndrome: pathogenesis, pathophysiology, and clinical management. Genes Dis. 2023;10:1846–56.
George S, Rochford JJ, Wolfrum C, Gray SL, Schinner S, Wilson JC, et al. A family with severe insulin resistance and diabetes due to a mutation in AKT2. Science. 2004;304:1325–8.
Moltke I, Grarup N, Jørgensen ME, Bjerregaard P, Treebak JT, Fumagalli M, et al. A common Greenlandic TBC1D4 variant confers muscle insulin resistance and type 2 diabetes. Nature. 2014;512:190–3.
Leto D, Saltiel AR. Regulation of glucose transport by insulin: traffic control of GLUT4. Nat Rev Mol Cell Biol. 2012;13:383–96.
Dahary D, Golan Y, Mazor Y, Zelig O, Barshir R, Twik M, et al. Genome analysis and knowledge-driven variant interpretation with Tgex. BMC Med Genomics. 2019;12:200.
Mölder F, Jablonski KP, Letcher B, Hall MB, Tomkins-Tinch CH, Sochat V, et al. Sustainable data analysis with Snakemake. F1000Res. 2021;10:33.
Hastings PJ, Ira G, Lupski JR. A microhomology-mediated break-induced replication model for the origin of human copy number variation. PLoS Genet. 2009;5:e1000327.
Carvalho CM, Ramocki MB, Pehlivan D, Franco LM, Gonzaga-Jauregui C, Fang P, et al. Inverted genomic segments and complex triplication rearrangements are mediated by inverted repeats in the human genome. Nat Genet. 2011;43:1074–81.
Beck CR, Carvalho CM, Banser L, Gambin T, Stubbolo D, Yuan B, et al. Complex genomic rearrangements at the PLP1 locus include triplication and quadruplication. PLoS Genet. 2015;11:e1005050.
Bahrambeigi V, Song X, Sperle K, Beck CR, Hijazi H, Grochowski CM, et al. Distinct patterns of complex rearrangements and a mutational signature of microhomeology are frequently observed in PLP1 copy number gain structural variants. Genome Med. 2019;11:80.
Ishmukhametova A, Chen JM, Bernard R, de Massy B, Baudat F, Boyer A, et al. Dissecting the structure and mechanism of a complex duplication-triplication rearrangement in the DMD gene. Hum Mutat. 2013;34:1080–4.
Kane S, Sano H, Liu SC, Asara JM, Lane WS, Garner CC, et al. A method to identify serine kinase substrates. Akt phosphorylates a novel adipocyte protein with a Rab GTPase-activating protein (GAP) domain. J Biol Chem. 2002;277:22115–8.
Ng Y, Ramm G, Lopez JA, James DE. Rapid activation of Akt2 is sufficient to stimulate GLUT4 translocation in 3T3-L1 adipocytes. Cell Metab. 2008;7:348–56.
Wang HY, Ducommun S, Quan C, Xie B, Li M, Wasserman DH, et al. AS160 deficiency causes whole-body insulin resistance via composite effects in multiple tissues. Biochem J. 2013;449:479–89.
Arias EB, Zheng X, Agrawal S, Cartee GD. Whole body glucoregulation and tissue-specific glucose uptake in a novel Akt substrate of 160 kDa knockout rat model. PLoS One. 2019;14:e0216236.
Dash S, Sano H, Rochford JJ, Semple RK, Yeo G, Hyden CS, et al. A truncation mutation in TBC1D4 in a family with acanthosis nigricans and postprandial hyperinsulinemia. Proc Natl Acad Sci USA. 2009;106:9350–5.
Dash S, Langenberg C, Fawcett KA, Semple RK, Romeo S, Sharp S, et al. Analysis of TBC1D4 in patients with severe insulin resistance. Diabetologia. 2010;53:1239–42.
Bouzakri K, Ribaux P, Tomas A, Parnaud G, Rickenbach K, Halban PA. Rab GTPase-activating protein AS160 is a major downstream effector of protein kinase B/Akt signaling in pancreatic beta-cells. Diabetes. 2008;57:1195–204.
Acknowledgements
The authors would like to thank the patient for his participation in this study.
Funding
No specific funding sources to declare.
Author information
Authors and Affiliations
Contributions
AC, HM-S and TH conceptualized the study and wrote the manuscript with input from all authors. AC and RP collected and analyzed clinical information. HM-S, BT, GS and ES analyzed genomic information. HR-F, SY-D and TH analyzed genomic data, and designed and performed molecular experiments.
Corresponding author
Ethics declarations
Competing interests
BT is an employee of Pacific Bioscience, GS and ES are employees of Ocean Genomics, and HM-S is an employee of Geneyx Genomix. The other authors declare no conflict of interest.
Ethics approval and consent for publication
The study was conducted in accordance with IRB-approved protocol 0306-10-HMO. Consent for publication was confirmed.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cahn, A., Mor-Shaked, H., Rosenberg-Fogler, H. et al. Complex rearrangement in TBC1D4 in an individual with diabetes due to severe insulin resistance syndrome. Eur J Hum Genet 32, 232–237 (2024). https://doi.org/10.1038/s41431-023-01512-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41431-023-01512-8