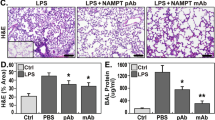
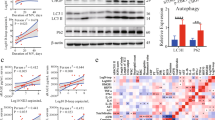
Abstract
Acetaldehyde dehydrogenase 2 (ALDH2) mutations are commonly found in a subgroup of the Asian population. However, the role of ALDH2 in septic acute respiratory distress syndrome (ARDS) remains unknown. Here, we showed that human subjects carrying the ALDH2rs671 mutation were highly susceptible to developing septic ARDS. Intriguingly, ALDH2rs671-ARDS patients showed higher levels of blood cell-free DNA (cfDNA) and myeloperoxidase (MPO)-DNA than ALDH2WT-ARDS patients. To investigate the mechanisms underlying ALDH2 deficiency in the development of septic ARDS, we utilized Aldh2 gene knockout mice and Aldh2rs671 gene knock-in mice. In clinically relevant mouse sepsis models, Aldh2-/- mice and Aldh2rs671 mice exhibited pulmonary and circulating NETosis, a specific process that releases neutrophil extracellular traps (NETs) from neutrophils. Furthermore, we discovered that NETosis strongly promoted endothelial destruction, accelerated vascular leakage, and exacerbated septic ARDS. At the molecular level, ALDH2 increased K48-linked polyubiquitination and degradation of peptidylarginine deiminase 4 (PAD4) to inhibit NETosis, which was achieved by promoting PAD4 binding to the E3 ubiquitin ligase CHIP. Pharmacological administration of the ALDH2-specific activator Alda-1 substantially alleviated septic ARDS by inhibiting NETosis. Together, our data reveal a novel ALDH2-based protective mechanism against septic ARDS, and the activation of ALDH2 may be an effective treatment strategy for sepsis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Bos LDJ, Ware LB. Acute respiratory distress syndrome: causes, pathophysiology, and phenotypes. Lancet. 2022;400:1145–56.
ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526–33.
Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315:788–800.
Fan E, Brodie D, Slutsky AS. Acute respiratory distress syndrome: advances in diagnosis and treatment. JAMA. 2018;319:698–710.
Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M, et al. Incidence and outcomes of acute lung injury. N. Engl J Med. 2005;353:1685–93.
Joffre J, Hellman J, Ince C, Ait-Oufella H. Endothelial responses in sepsis. Am J Respir Crit Care Med. 2020;202:361–70.
Jaffee W, Hodgins S, McGee WT. Tissue Edema, fluid balance, and patient outcomes in severe sepsis: an organ systems review. J Intensive Care Med. 2018;33:502–9.
Chang R, Holcomb JB. Choice of fluid therapy in the initial management of sepsis, severe sepsis, and septic shock. Shock. 2016;46:17–26.
Zhang J, Guo Y, Zhao X, Pang J, Pan C, Wang J, et al. The role of aldehyde dehydrogenase 2 in cardiovascular disease. Nat Rev Cardiol. 2023;20:495–509.
Zhang J, Zhao X, Guo Y, Liu Z, Wei S, Yuan Q, et al. Macrophage ALDH2 (Aldehyde Dehydrogenase 2) stabilizing Rac2 is required for efferocytosis internalization and reduction of atherosclerosis development. Arterioscler Thromb Vasc Biol. 2022;42:700–16.
Zhang Y, Lv Y, Zhang Q, Wang X, Han Q, Liang Y, et al. ALDH2 attenuates myocardial pyroptosis through breaking down Mitochondrion-NLRP3 inflammasome pathway in septic shock. Front Pharm. 2023;14:1125866.
Millwood IY, Walters RG, Mei XW, Guo Y, Yang L, Bian Z, et al. Conventional and genetic evidence on alcohol and vascular disease aetiology: a prospective study of 500 000 men and women in China. Lancet. 2019;393:1831–42.
Chen CH, Ferreira JC, Gross ER, Mochly-Rosen D. Targeting aldehyde dehydrogenase 2: new therapeutic opportunities. Physiol Rev. 2014;94:1–34.
Hsu LA, Tsai FC, Yeh YH, Chang CJ, Kuo CT, Chen WJ, et al. Aldehyde Dehydrogenase 2 ameliorates chronic alcohol consumption-induced atrial fibrillation through detoxification of 4-HNE. Int J Mol Sci. 2020;21:6678.
Chang JS, Hsiao JR, Chen CH. ALDH2 polymorphism and alcohol-related cancers in Asians: a public health perspective. J Biomed Sci. 2017;24:19.
Perez-Miller S, Younus H, Vanam R, Chen CH, Mochly-Rosen D, Hurley TD. Alda-1 is an agonist and chemical chaperone for the common human aldehyde dehydrogenase 2 variant. Nat Struct Mol Biol. 2010;17:159–64.
Sidramagowda Patil S, Hernández-Cuervo H, Fukumoto J, Krishnamurthy S, Lin M, Alleyn M, et al. Alda-1 attenuates hyperoxia-induced acute lung injury in mice. Front Pharm. 2020;11:597942.
Kuroda A, Hegab AE, Jingtao G, Yamashita S, Hizawa N, Sakamoto T, et al. Effects of the common polymorphism in the human aldehyde dehydrogenase 2 (ALDH2) gene on the lung. Respir Res. 2017;18:69.
Oka Y, Hamada M, Nakazawa Y, Muramatsu H, Okuno Y, Higasa K, et al. Digenic mutations in ALDH2 and ADH5 impair formaldehyde clearance and cause a multisystem disorder, AMeD syndrome. Sci Adv. 2020;6:eabd7197.
Hu Y, Yan JB, Zheng MZ, Song XH, Wang LL, Shen YL, et al. Mitochondrial aldehyde dehydrogenase activity protects against lipopolysaccharide‑induced cardiac dysfunction in rats. Mol Med Rep. 2015;11:1509–15.
Pang J, Zheng Y, Han Q, Zhang Y, Sun R, Wang J, et al. The role of ALDH2 in sepsis and the to-be-discovered mechanisms. Adv Exp Med Biol. 2019;1193:175–94.
Mantovani A, Cassatella MA, Costantini C, Jaillon S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat Rev Immunol. 2011;11:519–31.
Liew PX, Kubes P. The neutrophil’s role during health and disease. Physiol Rev. 2019;99:1223–48.
Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, et al. Neutrophil extracellular traps kill bacteria. Science. 2004;303:1532–5.
Fuchs TA, Abed U, Goosmann C, Hurwitz R, Schulze I, Wahn V, et al. Novel cell death program leads to neutrophil extracellular traps. J Cell Biol. 2007;176:231–41.
Yipp BG, Kubes P. NETosis: how vital is it? Blood. 2013;122:2784–94.
Thiam HR, Wong SL, Wagner DD, Waterman CM. Cellular Mechanisms of NETosis. Annu Rev Cell Dev Biol. 2020;36:191–218.
Chen L, Zhao Y, Lai D, Zhang P, Yang Y, Li Y, et al. Neutrophil extracellular traps promote macrophage pyroptosis in sepsis. Cell Death Dis. 2018;9:597.
Perdomo J, Leung H, Ahmadi Z, Yan F, Chong J, Passam FH, et al. Neutrophil activation and NETosis are the major drivers of thrombosis in heparin-induced thrombocytopenia. Nat Commun. 2019;10:1322.
Mattox AK, Douville C, Wang Y, Popoli M, Ptak J, Silliman N, et al. The origin of highly elevated cell-free DNA in healthy individuals and patients with pancreatic, colorectal, lung, or ovarian cancer. Cancer Discov. 2023;13:2166–2179.
Thiam HR, Wong SL, Qiu R, Kittisopikul M, Vahabikashi A, Goldman AE, et al. NETosis proceeds by cytoskeleton and endomembrane disassembly and PAD4-mediated chromatin decondensation and nuclear envelope rupture. Proc Natl Acad Sci USA. 2020;117:7326–37.
Neubert E, Meyer D, Rocca F, Günay G, Kwaczala-Tessmann A, Grandke J, et al. Chromatin swelling drives neutrophil extracellular trap release. Nat Commun. 2018;9:3767.
Li P, Li M, Lindberg MR, Kennett MJ, Xiong N, Wang Y. PAD4 is essential for antibacterial innate immunity mediated by neutrophil extracellular traps. J Exp Med. 2010;207:1853–62.
Martinod K, Demers M, Fuchs TA, Wong SL, Brill A, Gallant M, et al. Neutrophil histone modification by peptidylarginine deiminase 4 is critical for deep vein thrombosis in mice. Proc Natl Acad Sci USA. 2013;110:8674–9.
Biron BM, Chung CS, Chen Y, Wilson Z, Fallon EA, Reichner JS, et al. PAD4 deficiency leads to decreased organ dysfunction and improved survival in a dual insult model of hemorrhagic shock and sepsis. J Immunol. 2018;200:1817–28.
Alsabani M, Abrams ST, Cheng Z, Morton B, Lane S, Alosaimi S, et al. Reduction of NETosis by targeting CXCR1/2 reduces thrombosis, lung injury, and mortality in experimental human and murine sepsis. Br J Anaesth. 2022;128:283–93.
Pham T, Rubenfeld GD. Fifty years of research in ARDS. The epidemiology of acute respiratory distress syndrome. a 50th birthday review. Am J Respir Crit Care Med. 2017;195:860–70.
Rezoagli E, Fumagalli R, Bellani G. Definition and epidemiology of acute respiratory distress syndrome. Ann Transl Med. 2017;5:282.
Matthay MA, Zemans RL, Zimmerman GA, Arabi YM, Beitler JR, Mercat A, et al. Acute respiratory distress syndrome. Nat Rev Dis Prim. 2019;5:18.
Parotto M, Gyöngyösi M, Howe K, Myatra SN, Ranzani O, Shankar-Hari M, et al. Post-acute sequelae of COVID-19: understanding and addressing the burden of multisystem manifestations. Lancet Respir Med. 2023;11:739–54.
Carmona-Rivera C, Zhao W, Yalavarthi S, Kaplan MJ. Neutrophil extracellular traps induce endothelial dysfunction in systemic lupus erythematosus through the activation of matrix metalloproteinase-2. Ann Rheum Dis. 2015;74:1417–24.
Kato N, Takeuchi F, Tabara Y, Kelly TN, Go MJ, Sim X, et al. Meta-analysis of genome-wide association studies identifies common variants associated with blood pressure variation in east Asians. Nat Genet. 2011;43:531–8.
Ma H, Guo R, Yu L, Zhang Y, Ren J. Aldehyde dehydrogenase 2 (ALDH2) rescues myocardial ischaemia/reperfusion injury: role of autophagy paradox and toxic aldehyde. Eur Heart J. 2011;32:1025–38.
Dingler FA, Wang M, Mu A, Millington CL, Oberbeck N, Watcham S, et al. Two aldehyde clearance systems are essential to prevent lethal formaldehyde accumulation in mice and humans. Mol Cell. 2020;80:996–1012.e9.
Liu H, Hu Q, Ren K, Wu P, Wang Y, Lv C. ALDH2 mitigates LPS-induced cardiac dysfunction, inflammation, and apoptosis through the cGAS/STING pathway. Mol Med. 2023;29:171.
Jin J, Chang RS, Xu S, Xia G, Wong J, Fang Y, et al. Aldehyde Dehydrogenase 2 ameliorates LPS-induced acute kidney injury through detoxification of 4-HNE and suppression of the MAPK pathway. J Immunol Res. 2023;2023:5513507.
Ji W, Wan T, Zhang F, Zhu X, Guo S, Mei X. Aldehyde Dehydrogenase 2 protects against lipopolysaccharide-induced myocardial injury by suppressing mitophagy. Front Pharm. 2021;12:641058.
Ning L, Wei W, Wenyang J, Rui X, Qing G. Cytosolic DNA-STING-NLRP3 axis is involved in murine acute lung injury induced by lipopolysaccharide. Clin Transl Med. 2020;10:e228.
Rittirsch D, Huber-Lang MS, Flierl MA, Ward PA. Immunodesign of experimental sepsis by cecal ligation and puncture. Nat Protoc. 2009;4:31–6.
Ho J, Chan H, Liang Y, Liu X, Zhang L, Li Q, et al. Cathelicidin preserves intestinal barrier function in polymicrobial sepsis. Crit Care. 2020;24:47.
Han S, Lee SJ, Kim KE, Lee HS, Oh N, Park I, et al. Amelioration of sepsis by TIE2 activation-induced vascular protection. Sci Transl Med. 2016;8:335ra55.
Peng X, Hassoun PM, Sammani S, McVerry BJ, Burne MJ, Rabb H, et al. Protective effects of sphingosine 1-phosphate in murine endotoxin-induced inflammatory lung injury. Am J Respir Crit Care Med. 2004;169:1245–51.
Kulkarni HS, Lee JS, Bastarache JA, Kuebler WM, Downey GP, Albaiceta GM, et al. Update on the features and measurements of experimental acute lung injury in animals: An Official American Thoracic Society Workshop Report. Am J Respir Cell Mol Biol. 2022;66:e1–e14.
Jin H, Aziz M, Murao A, Kobritz M, Shih AJ, Adelson RP, et al. Antigen-presenting aged neutrophils induce CD4+ T cells to exacerbate inflammation in sepsis. J Clin Invest. 2023;133:e164585.
Kim B, Jang C, Dharaneeswaran H, Li J, Bhide M, Yang S, et al. Endothelial pyruvate kinase M2 maintains vascular integrity. J Clin Invest. 2018;128:4543–56.
Newman AM, Bratman SV, To J, Wynne JF, Eclov NC, Modlin LA, et al. An ultrasensitive method for quantitating circulating tumor DNA with broad patient coverage. Nat Med. 2014;20:548–54.
Middleton EA, He XY, Denorme F, Campbell RA, Ng D, Salvatore SP, et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood. 2020;136:1169–79.
Najmeh, S, Cools-Lartigue J, Giannias B, Spicer J, Ferri LE, Simplified Human Neutrophil Extracellular Traps (NETs) Isolation and Handling. J Vis Exp. 2015;98:52687.
Deng M, Tang Y, Li W, Wang X, Zhang R, Zhang X, et al. The endotoxin delivery protein HMGB1 mediates Caspase-11-dependent lethality in sepsis. Immunity. 2018;49:740–753.e7.
Acknowledgements
This study was supported by the State Key Program of the National Natural Science Foundation of China (82030059), the National Science Fund for Distinguished Young Scholars (82325031), the National Natural Science Regional Innovation Fund Joint Fund Key Support Projects (U23A20485), the National Natural Science Foundation of China (82072144, 82172127), the National Key R&D Program of China (2020YFC1512700, 2020YFC1512705, 2020YFC1512703), the Key R&D Program of Shandong Province (2021ZLGX02, 2021SFGC0503, 2022ZLGX03), the Taishan Pandeng Scholar Program of Shandong Province (tspd20181220), the Taishan Young Scholar Program of Shandong Province (tsqn202211312), the Clinical Research Project of Shandong University (2021SDUCRCC006), and the Interdisciplinary Young Researcher Groups Program of Shandong University (2020QNQT004). Illustrations were made using BioRender.
Author information
Authors and Affiliations
Contributions
Y Chen and JP conceived and designed the experiments. CX and LZ performed most of the experiments with the help of SX, ZW, QH, YL, XW, XZ, QZ, YZ, SH, and QY CX, LZ, SX, ZW, and YB performed the data analysis. Y Cao and JP assisted in revising the draft. CL, JW, and FX provided valuable experimental advice and guidance. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Supplementary information
41423_2024_1146_MOESM1_ESM.docx
Sup Fig1-11, Tab1-5, unprocessed original images (Supplementary Materials for Neutrophil ALDH2 is a new therapeutic target for effective treatment of sepsis-triggered ARDS)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, C., Zhang, L., Xu, S. et al. Neutrophil ALDH2 is a new therapeutic target for the effective treatment of sepsis-induced ARDS. Cell Mol Immunol 21, 510–526 (2024). https://doi.org/10.1038/s41423-024-01146-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-024-01146-w