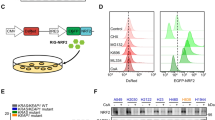
Abstract
Liver cancer stem cells (LCSCs) contribute to tumor recurrence and cancer cell proliferation in patients with hepatocellular carcinoma (HCC). METTL3-catalyzed m6A modification is relevant to the cancer stem cell (CSC) phenotype, including LCSCs. LCSCs were isolated from MHCC-97H and HepG2 cells through flow cytometry. UALCAN data were used to analyze the expression of METTL3 in liver hepatocellular carcinoma (LIHC) tissues. Loss- and gain-of-function experiments were utilized to assess the biological effects of METTL3 and SOCS3 on the proliferation and stemness phenotypes in vitro and in vivo. The mechanisms underlying the impact of METTL3 were explored using qPCR, MeRIP-qPCR, dual-luciferase reporter, and western blot assays. METTL3 was significantly upregulated in LIHC tissues according to the UALCAN database. METTL3 was highly expressed in LIHC and was significantly correlated with individual cancer stage, tumor grade and lymph node metastasis. Patients with low METTL3 expression had a longer overall survival time based on the data from UALCAN. In addition, the level of METTL3 was enhanced in LCSCs and decreased in non-LCSCs compared to HCC cells. Moreover, overexpression of METTL3 stimulated the proliferation and stemness of LCSCs in vitro and in vivo, while loss of METTL3 impeded it. Bioinformatics analysis combined with validation experiments determined that m6A was modified by METTL3-targeting SOCS3 mRNA. METTL3 had side effects regarding the stability of SOCS3 mRNA. SOCS3 overexpression impaired and SOCS3 depletion facilitated the development of LCSCs via the JAK2/STAT3 pathway. Furthermore, METTL3 depletion suppressed proliferation and stemness in LCSCs, which was restored by SOCS3 knockdown or colivelin treatment. We discovered that METTL3 facilitated the stemness and tumorigenicity of LCSCs by modifying SOCS3 mRNA with m6A.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The datasets used or analyzed during this study are available from the corresponding author upon reasonable request.
References
Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358–80.
Lee J, Kim JS, Cho HI, Jo SR, Jang YK. JIB-04, a Pan-inhibitor of histone demethylases, targets histone-lysine-demethylase-dependent AKT pathway, leading to cell cycle arrest and inhibition of cancer stem-like cell properties in hepatocellular carcinoma cells. Int J Mol Sci. 2022;23:7657.
Yao CY, Gao ZX, Hou LL, Fang D. DKK1 promotes NUAK1 transcriptional expression through the activation Akt in hepatocellular carcinoma. Cell Biol Int. 2022;47:383–93.
Kong H, Sun J, Zhang W, Zhang H, Li H. Long intergenic non-protein coding RNA 1273 confers sorafenib resistance in hepatocellular carcinoma via regulation of methyltransferase 3. Bioengineered. 2022;13:3108–21.
Wong HL, Ng LPW, Koh SP, Chan LWC, Wong EYK, Xue VW, et al. Hotspot KRAS exon 2 mutations in CD166 positive colorectal cancer and colorectal adenoma cells. Oncotarget. 2018;9:20426–38.
Mansour FA, Al-Mazrou A, Al-Mohanna F, Al-Alwan M, Ghebeh H. PD-L1 is overexpressed on breast cancer stem cells through notch3/mTOR axis. Oncoimmunology. 2020;9:1729299.
Park S-R, Kim S-R, Hong I-S, Lee H-Y. A novel therapeutic approach for colorectal cancer stem cells: blocking the PI3K/Akt signaling axis with caffeic acid. Front Cell Dev Biol. 2020;8:585987.
Chhabra R. let-7i-5p, miR-181a-2-3p and EGF/PI3K/SOX2 axis coordinate to maintain cancer stem cell population in cervical cancer. Sci Rep. 2018;8:1–9.
Zhang L, Liang H, Zhang J, Yang Y, Ling X, Jiang H. Long non-coding RNA SNHG16 facilitates esophageal cancer cell proliferation and self-renewal through the microRNA-802/PTCH1 axis. Curr Med Chem. 2022;29:6084–99.
Chen W, Dong J, Haiech J, Kilhoffer MC, Zeniou M. Cancer stem cell quiescence and plasticity as major challenges in cancer therapy. Stem Cells Int. 2016;2016:1740936.
Chen XY, Zhang J, Zhu JS. The role of m(6)A RNA methylation in human cancer. Mol Cancer. 2019;18:103.
Lan Q, Liu PY, Haase J, Bell JL, Huttelmaier S, Liu T. The critical role of RNA m(6)A methylation in cancer. Cancer Res. 2019;79:1285–92.
Li H, Wang C, Lan L, Yan L, Li W, Evans I, et al. METTL3 promotes oxaliplatin resistance of gastric cancer CD133+ stem cells by promoting PARP1 mRNA stability. Cell Mol Life Sci. 2022;79:1–22.
Wang G, Dai Y, Li K, Cheng M, Xiong G, Wang X, et al. Deficiency of Mettl3 in bladder cancer stem cells inhibits bladder cancer progression and angiogenesis. Front Cell Dev Biol. 2021;9:627706.
Yoshimura A, Naka T, Kubo M. SOCS proteins, cytokine signalling and immune regulation. Nat Rev Immunol. 2007;7:454–65.
Yang S, Wang B, Liu C, Wang Q, Wang R, Su W, et al. THAP9-AS1 promotes tumorigenesis and reduces ROS generation through the JAK2/STAT3 signaling pathway by increasing SOCS3 promoter methylation in osteosarcoma. Oxid Med Cell Longev. 2021;2021:5620475.
Jiang L, Chen T, Xiong L, Xu JH, Gong AY, Dai B, et al. Knockdown of m6A methyltransferase METTL3 in gastric cancer cells results in suppression of cell proliferation. Oncol Lett. 2020;20:2191–8.
Wang J, Zhou H, Han Y, Liu X, Wang M, Wang X, et al. SOCS3 methylation in synergy with Reg3A overexpression promotes cell growth in pancreatic cancer. J Mol Med. 2014;92:1257–69.
Chen H, Zhang C, Sheng Y, Yao S, Liu Z, Zhang C, et al. Frequent SOCS3 and 3OST2 promoter methylation and their epigenetic regulation in endometrial carcinoma. Am J Cancer Res. 2015;5:180.
Jiang B-G, Wang N, Huang J, Yang Y, Sun L-L, Pan Z-Y, et al. Tumor SOCS3 methylation status predicts the treatment response to TACE and prognosis in HCC patients. Oncotarget. 2017;8:28621.
Yang F, Zhang A. Involvement of METTL3 in arsenite-induced skin lesions by targeting the SOCS3/STAT3/Krt signaling pathway. Environ Pollut. 2023;316:120634.
Wu R, Liu Y, Zhao Y, Bi Z, Yao Y, Liu Q, et al. m6A methylation controls pluripotency of porcine induced pluripotent stem cells by targeting SOCS3/JAK2/STAT3 pathway in a YTHDF1/YTHDF2-orchestrated manner. Cell Death Dis. 2019;10:1–15.
Wei X, You X, Zhang J, Zhou C. MicroRNA-1305 inhibits the stemness of LCSCs and tumorigenesis by repressing the UBE2T-dependent Akt-signaling pathway. Mol Ther Nucleic Acids. 2019;16:721–32.
Tang Q, Chen J, Di Z, Yuan W, Zhou Z, Liu Z, et al. TM4SF1 promotes EMT and cancer stemness via the Wnt/beta-catenin/SOX2 pathway in colorectal cancer. J Exp Clin Cancer Res. 2020;39:232.
Lv Y, Lv X, Yang H, Qi X, Wang X, Li C, et al. LncRNA SNHG6/miR-125b-5p/BMPR1B axis: a new therapeutic target for triple-negative breast cancer. Front Oncol. 2021;11:678474.
Liu ZX, Li LM, Sun HL, Liu SM. Link between m6A modification and cancers. Front Bioeng Biotechnol. 2018;6:89.
Xu H, Wang H, Zhao W, Fu S, Li Y, Ni W, et al. SUMO1 modification of methyltransferase-like 3 promotes tumor progression via regulating Snail mRNA homeostasis in hepatocellular carcinoma. Theranostics. 2020;10:5671–86.
Dominguez E, Mauborgne A, Mallet J, Desclaux M, Pohl M. SOCS3-mediated blockade of JAK/STAT3 signaling pathway reveals its major contribution to spinal cord neuroinflammation and mechanical allodynia after peripheral nerve injury. J Neurosci. 2010;30:5754–66.
Wang Q, Geng W, Guo H, Wang Z, Xu K, Chen C, et al. Emerging role of RNA methyltransferase METTL3 in gastrointestinal cancer. J Hematol Oncol. 2020;13:1–10.
Gao Q, Zheng J, Ni Z, Sun P, Yang C, Cheng M, et al. The m6A methylation-regulated AFF4 promotes self-renewal of bladder cancer stem cells. Stem cells Int. 2020;2020:8849218.
Yao QJ, Sang L, Lin M, Yin X, Dong W, Gong Y, et al. Mettl3-Mettl14 methyltransferase complex regulates the quiescence of adult hematopoietic stem cells. Cell Res. 2018;28:952–4.
Iaiza A, Tito C, Ianniello Z, Ganci F, Laquintana V, Gallo E, et al. METTL3-dependent MALAT1 delocalization drives c-Myc induction in thymic epithelial tumors. Clin Epigenet. 2021;13:173.
Pan J, Liu F, Xiao X, Xu R, Dai L, Zhu M, et al. METTL3 promotes colorectal carcinoma progression by regulating the m6A-CRB3-Hippo axis. J Exp Clin Cancer Res. 2022;41:19.
Xie H, Li J, Ying Y, Yan H, Jin K, Ma X, et al. METTL3/YTHDF2 m6A axis promotes tumorigenesis by degrading SETD7 and KLF4 mRNAs in bladder cancer. J Cell Mol Med. 2020;24:4092–104.
Wang J, Zhao H, Zheng L, Zhou Y, Wu L, Xu Y, et al. FGF19/SOCE/NFATc2 signaling circuit facilitates the self-renewal of liver cancer stem cells. Theranostics. 2021;11:5045.
Song RH, Du P, Gao CQ, Liu XR, Zhang JA. METTL3 is involved in the development of graves’ disease by inducing SOCS mRNA m6A modification. Front Endocrinol. 2021;12:666393.
Zhang S, Ji WW, Wei W, Zhan LX, Huang X. Long noncoding RNA Meg3 sponges miR-708 to inhibit intestinal tumorigenesis via SOCS3-repressed cancer stem cells growth. Cell Death Dis. 2021;13:25.
Chu J, Hu X-C, Li C-C, Li T-Y, Fan H-W, Jiang G-Q. KLF14 alleviated breast cancer invasion and M2 macrophages polarization through modulating SOCS3/RhoA/Rock/STAT3 signaling. Cell Signal. 2022;92:110242.
Li M, Zheng R, Yuan F. MiR-410 affects the proliferation and apoptosis of lung cancer A549 cells through regulation of SOCS3/JAK-STAT signaling pathway. Eur Rev Med Pharm Sci. 2018;22:5987–93.
Lei Z, Duan H, Zhao T, Zhang Y, Li G, Meng J, et al. PARK2 inhibits osteosarcoma cell growth through the JAK2/STAT3/VEGF signaling pathway. Cell Death Dis. 2018;9:375.
Zeng YT, Liu XF, Yang WT, Zheng PS. REX1 promotes EMT-induced cell metastasis by activating the JAK2/STAT3-signaling pathway by targeting SOCS1 in cervical cancer. Oncogene. 2019;38:6940–57.
Acknowledgements
We would like to thank the anonymous reviewers who have helped to improve the paper.
Funding
This work was supported by the Ningbo Natural Science Foundation (Grant No.2022J248), Key Research Foundation of Ningbo No. 2 Hospital, China (Grant No. 2022HMZD08), the Public Welfare Foundation of Ningbo (2021S108), Ningbo leading Medical & Health Discipline-Anorectal Surgery (2022-B11), and the Science and Research Project of Ningbo City College of Vocational Technology (ZZX22230).
Author information
Authors and Affiliations
Contributions
HY guaranteed the integrity of the entire study; JH designed the study and literature research; HY defined the intellectual content; KC and FH performed the experiments; GG collected the data; XD analyzed the data; and HY wrote the main manuscript and prepared the figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was approved by the ethics committee of Ningbo No. 2 Hospital. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, J., Chen, K., Hong, F. et al. METTL3 facilitates stemness properties and tumorigenicity of cancer stem cells in hepatocellular carcinoma through the SOCS3/JAK2/STAT3 signaling pathway. Cancer Gene Ther 31, 228–236 (2024). https://doi.org/10.1038/s41417-023-00697-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-023-00697-w