Abstract
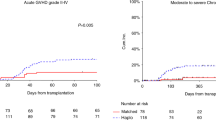
Posttransplant cyclophosphamide (PTCy)-based graft-versus-host disease (GVHD) prophylaxis has been increasingly used in HLA-haploidentical transplantation and recent studies also demonstrated the efficacy of PTCy in HLA-matched transplantation. We conducted a prospective multicenter phase II study to evaluate the safety and efficacy of PTCy with tacrolimus and mycophenolate mofetil in 43 patients who underwent HLA-matched (n = 21), 1 allele mismatched (n = 20), or 2 allele mismatched (n = 2) peripheral blood stem cell transplantation (PBSCT) following myeloablative (n = 28) or reduced-intensity (n = 15) conditioning. The incidence of grade III–IV acute GVHD at 100 days was 2.3%. The incidences of grades II–IV acute GVHD, all grade chronic GVHD, and moderate to severe chronic GVHD at 2 years were 16.3%, 14.0%, and 4.7%, respectively. Overall survival, disease-free survival, and non-relapse mortality at 2 years were 75.3%, 74.0%, and 7.0%, respectively. GVHD-free, relapse-free survival at 2 years was 67.0%. The rate of off-immunosuppressants in patients who survived without relapse at 2 years was 85.4%. These results indicate that PTCy is a valid option for GVHD prophylaxis in both HLA-matched and HLA 1–2 allele mismatched PBSCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The dataset supporting the conclusions of this article is available upon request to the corresponding author.
References
Bolaños-Meade J, Hamadani M, Wu J, Al Malki MM, Martens MJ, Runaas L, et al. Post-transplantation cyclophosphamide-based graft-versus-host disease prophylaxis. N Engl J Med. 2023;388:2338–48.
Broers AEC, de Jong CN, Bakunina K, Hazenberg MD, van Marwijk Kooy M, de Groot MR, et al. Posttransplant cyclophosphamide for prevention of graft-versus-host disease: results of the prospective randomized HOVON-96 trial. Blood Adv. 2022;6:3378–85.
Luznik L, Pasquini MC, Logan B, Soiffer RJ, Wu J, Devine SM, et al. Randomized phase III BMT CTN trial of calcineurin inhibitor-free chronic graft-versus-host disease interventions in myeloablative hematopoietic cell transplantation for hematologic malignancies. J Clin Oncol. 2022;40:356–68.
Shaw BE, Jimenez-Jimenez AM, Burns LJ, Logan BR, Khimani F, Shaffer BC, et al. National marrow donor program-sponsored multicenter, phase II trial of HLA-mismatched unrelated donor bone marrow transplantation using post-transplant cyclophosphamide. J Clin Oncol. 2021;39:1971–82.
Battipaglia G, Galimard J-E, Labopin M, Raiola AM, Blaise D, Ruggeri A, et al. Post-transplant cyclophosphamide in one-antigen mismatched unrelated donor transplantation versus haploidentical transplantation in acute myeloid leukemia: a study from the Acute Leukemia Working Party of the EBMT. Bone Marrow Transplant. 2022;57:562–71.
Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, Sanz G, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood. 1997;89:2079–88.
Malcovati L, Germing U, Kuendgen A, Della Porta MG, Pascutto C, Invernizzi R, et al. Time-dependent prognostic scoring system for predicting survival and leukemic evolution in myelodysplastic syndromes. J Clin Oncol. 2007;25:3503–10.
Harris AC, Young R, Devine S, Hogan WJ, Ayuk F, Bunworasate U, et al. International, multicenter standardization of acute graft-versus-host disease clinical data collection: a report from the mount sinai acute GVHD International Consortium. Biol Blood Marrow Transplant. 2016;22:4–10.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National Institutes of Health Consensus Development Project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant. 2015;21:389–401.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123:3664–71.
Sugita J, Kawashima N, Fujisaki T, Kakihana K, Ota S, Matsuo K, et al. HLA-haploidentical peripheral blood stem cell transplantation with post-transplant cyclophosphamide after busulfan-containing reduced-intensity conditioning. Biol Blood Marrow Transplant. 2015;21:1646–52.
Sugita J, Kagaya Y, Miyamoto T, Shibasaki Y, Nagafuji K, Ota S, et al. Myeloablative and reduced-intensity conditioning in HLA-haploidentical peripheral blood stem cell transplantation using post-transplant cyclophosphamide. Bone Marrow Transplant. 2019;54:432–41.
Nagafuji K, Matsuo K, Teshima T, Mori S-I, Sakamaki H, Hidaka M, et al. Peripheral blood stem cell versus bone marrow transplantation from HLA-identical sibling donors in patients with leukemia: a propensity score-based comparison from the Japan Society for Hematopoietic Stem Cell Transplantation registry. Int J Hematol. 2010;91:855–64.
Gooley TA, Leisenring W, Crowley J, Storer BE. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med. 1999;18:695–706.
Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988;16:1141–54.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Ruggeri A, Labopin M, Bacigalupo A, Afanasyev B, Cornelissen JJ, Elmaagacli A, et al. Post-transplant cyclophosphamide for graft-versus-host disease prophylaxis in HLA matched sibling or matched unrelated donor transplant for patients with acute leukemia, on behalf of ALWP-EBMT. J Hematol Oncol. 2018;11:40.
Kasamon YL, Ambinder RF, Fuchs EJ, Zahurak M, Rosner GL, Bolaños-Meade J, et al. Prospective study of nonmyeloablative, HLA-mismatched unrelated BMT with high-dose posttransplantation cyclophosphamide. Blood Adv. 2017;1:288–92.
Al Malki MM, Tsai N-C, Palmer J, Mokhtari S, Tsai W, Cao T, et al. Posttransplant cyclophosphamide as GVHD prophylaxis for peripheral blood stem cell HLA-mismatched unrelated donor transplant. Blood Adv. 2021;5:2650–9.
Rappazzo KC, Zahurak M, Bettinotti M, Ali SA, Ambinder AJ, Bolaños-Meade J, et al. Nonmyeloablative, HLA-mismatched unrelated peripheral blood transplantation with high-dose post-transplantation cyclophosphamide. Transplant Cell Ther. 2021;27:909.e1–6.
Jimenez Jimenez A, Komanduri K, Brown S, Wang T, Pereira D, Goodman M, et al. Improved GRFS after posttransplant cyclophosphamide-based vs ATG-based HLA-mismatched unrelated donor transplant. Blood Adv. 2022;6:4491–500.
Kanakry CG, Bolaños-Meade J, Kasamon YL, Zahurak M, Durakovic N, Furlong T, et al. Low immunosuppressive burden after HLA-matched related or unrelated BMT using posttransplantation cyclophosphamide. Blood. 2017;129:1389–93.
Rimando J, McCurdy SR, Luznik L. How I prevent GVHD in high-risk patients: posttransplant cyclophosphamide and beyond. Blood. 2023;141:49–59.
Luznik L, O’Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transplant. 2008;14:641–50.
Sugita J, Kamimura T, Ishikawa T, Ota S, Eto T, Kuroha T, et al. Reduced dose of posttransplant cyclophosphamide in HLA-haploidentical peripheral blood stem cell transplantation. Bone Marrow Transplant. 2021;56:596–604.
Duléry R, Goudet C, Mannina D, Bianchessi A, Granata A, Harbi S, et al. Reduced post-transplant cyclophosphamide doses in haploidentical hematopoietic cell transplantation for elderly patients with hematological malignancies. Bone Marrow Transplant. 2023;58:386–92.
McCurdy SR, Muth ST, Tsai HL, Symons HJ, Huff CA, Matsui WH, et al. Early fever after haploidentical bone marrow transplantation correlates with class II HLA-mismatching and myeloablation but not outcomes. Biol Blood Marrow Transplant. 2018;24:2056–64.
Solomon SR, Aubrey MT, Zhang X, Jackson KC, Morris LE, Holland HK, et al. Class II HLA mismatch improves outcomes following haploidentical transplantation with posttransplant cyclophosphamide. Blood Adv. 2020;4:5311–21.
Fleischhauer K. Haplo-PtCy: adjusting the HLA barrier. Blood. 2022;139:1431–3.
Fuchs EJ, McCurdy SR, Solomon SR, Wang T, Herr MR, Modi D, et al. HLA informs risk predictions after haploidentical stem cell transplantation with posttransplantation cyclophosphamide. Blood. 2022;139:1452–68.
Wiercinska E, Seifried E, Bonig H. CD3/CD19 depletion for T-cell reduction of allogeneic transplants: mostly efficient, but not robust. Clin Hematol Int. 2021;3:103–7.
Acknowledgements
This work was supported by Japan Agency for Medical Research and Development (AMED, 20ek0510025h0003).
Author information
Authors and Affiliations
Contributions
JS designed the study, recruited patients, collected, assembled, and analyzed data, performed the statistical analysis, and wrote the manuscript. TT designed the study, supervised research, analyzed data, and commented on the manuscript. KA, ST, and MH helped design the study, and commented on the manuscript. IY analyzed data, performed the statistical analysis, and commented on the manuscript. TK, JI, TE, and KF recruited patients and commented on the manuscript. All authors reviewed the manuscript and approved its submission for publication purposes.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sugita, J., Kuroha, T., Ishikawa, J. et al. Posttransplant cyclophosphamide in unrelated and related peripheral blood stem cell transplantation from HLA-matched and 1 allele mismatched donor. Bone Marrow Transplant 59, 344–349 (2024). https://doi.org/10.1038/s41409-023-02162-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-023-02162-6