Abstract
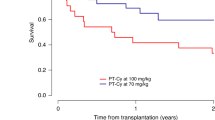
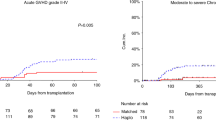
Although post-transplant cyclophosphamide (PT-Cy) is effective for graft-versus-host disease (GVHD) prophylaxis, it is associated with toxicities, which might be dose-dependent. We compared the outcomes with PT-Cy at 80 mg/kg to those with PT-Cy at 100 mg/kg in elderly patients undergoing haploidentical hematopoietic cell transplantation (HCT). Inclusion criteria included peripheral blood stem cells, hematological malignancy, and age>65 years (or age>60 years if cardiac event history). Thirty-eight patients received PT-Cy at 80 mg/kg and 55 100 mg/kg, divided in two doses. The cumulative incidences (CI) of acute grade II–IV, acute grade III–IV, and moderate/severe chronic GVHD were 32%, 16%, and 13% with PT-Cy at 80 mg/kg compared to 33%, 13%, and 16% with 100 mg/kg, respectively. In multivariable analysis, reducing PT-Cy dose had no significant impact on GVHD. Neutrophil and platelet engraftments were significantly improved, and CI of BK virus-associated hemorrhagic cystitis was reduced with 80 mg/kg of PT-Cy compared to 100 mg/kg. At 2 years, non-relapse mortality was 16% and 31%, progression-free survival 65% and 49%, overall survival 70% and 56%, and GVHD-free, relapse-free survival 52% and 36% with 80 mg/kg and 100 mg/kg, respectively. Reducing PT-Cy dose to 80 mg/kg is safe and associated with improved hematological recovery and lower CI of hemorrhagic cystitis in elderly patients undergoing haploidentical HCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The dataset supporting the conclusions of this article is available upon demand to the corresponding author.
References
Passweg JR, Baldomero H, Chabannon C, Basak GW, de la Cámara R, Corbacioglu S, et al. Hematopoietic cell transplantation and cellular therapy survey of the EBMT: monitoring of activities and trends over 30 years. Bone Marrow Transpl. 2021;56:1651–64.
Muffly L, Pasquini MC, Martens M, Brazauskas R, Zhu X, Adekola K, et al. Increasing use of allogeneic hematopoietic cell transplantation in patients aged 70 years and older in the United States. Blood. 2017;130:1156–64.
Robinson TM, O’Donnell PV, Fuchs EJ, Luznik L. Haploidentical bone marrow and stem cell transplantation: experience with post-transplantation cyclophosphamide. Semin Hematol. 2016;53:90–7.
Castagna L, Devillier R, Vey N, Blaise D. T-cell-replete haploidentical transplantation in acute myeloid leukemia. Exp Hematol. 2018;58:5–16.
Liu JH, Kanakry CG, Luznik L. Have haploidentical transplants replaced umbilical cord transplants for acute leukemias? Curr Opin Hematol. 2018;25:103–11.
Passweg JR, Baldomero H, Bader P, Basak GW, Bonini C, Duarte R, et al. Is the use of unrelated donor transplantation leveling off in Europe? The 2016 European Society for Blood and Marrow Transplant activity survey report. Bone Marrow Transpl. 2018;53:1139–48.
Luznik L, O’Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2008;14:641–50.
Raiola AM, Dominietto A, di Grazia C, Lamparelli T, Gualandi F, Ibatici A, et al. Unmanipulated haploidentical transplants compared with other alternative donors and matched sibling grafts. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2014;20:1573–9.
Ruggeri A, Labopin M, Sanz G, Piemontese S, Arcese W, Bacigalupo A, et al. Comparison of outcomes after unrelated cord blood and unmanipulated haploidentical stem cell transplantation in adults with acute leukemia. Leukemia. 2015;29:1891–900.
Ciurea SO, Zhang MJ, Bacigalupo AA, Bashey A, Appelbaum FR, Aljitawi OS, et al. Haploidentical transplant with posttransplant cyclophosphamide vs matched unrelated donor transplant for acute myeloid leukemia. Blood. 2015;126:1033–40.
Bashey A, Zhang X, Jackson K, Brown S, Ridgeway M, Solh M, et al. Comparison of outcomes of hematopoietic cell transplants from T-replete haploidentical donors using post-transplantation cyclophosphamide with 10 of 10 HLA-A, -B, -C, -DRB1, and -DQB1 allele-matched unrelated donors and HLA-identical sibling donors: a multivariable analysis including disease risk index. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2016;22:125–33.
Martínez C, Gayoso J, Canals C, Finel H, Peggs K, Dominietto A, et al. Post-transplantation cyclophosphamide-based haploidentical transplantation as alternative to matched sibling or unrelated donor transplantation for hodgkin lymphoma: a registry study of the lymphoma working party of the european society for blood and marrow transplantation. J Clin Oncol J Am Soc Clin Oncol. 2017;35:3425–32.
Gauthier J, Poiré X, Gac AC, Leclerc M, Guillaume T, Chalandon Y, et al. Better outcome with haploidentical over HLA-matched related donors in patients with Hodgkin’s lymphoma undergoing allogeneic haematopoietic cell transplantation-a study by the Francophone Society of Bone Marrow Transplantation and Cellular Therapy. Bone Marrow Transpl. 2018;53:400–09.
Duléry R, Ménard AL, Chantepie S, Cheikh JE, François S, Delage J, et al. Sequential conditioning with thiotepa in T cell- replete hematopoietic stem cell transplantation for the treatment of refractory hematologic malignancies: comparison with matched related, haplo-mismatched, and unrelated donors. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2018;24:1013–21.
Cytryn S, Abdul-Hay M. Haploidentical hematopoietic stem cell transplantation followed by ‘post-cyclophosphamide’: the future of allogeneic stem cell transplant. Clin Hematol Int. 2020;2:49–58.
Stocker N, Gaugler B, Labopin M, Farge A, Ye Y, Ricard L, et al. High-dose post-transplant cyclophosphamide impairs γδ T-cell reconstitution after haploidentical haematopoietic stem cell transplantation using low-dose antithymocyte globulin and peripheral blood stem cell graft. Clin Transl Immunol. 2020;9:e1171.
Rambaldi B, Kim HT, Reynolds C, Chamling Rai S, Arihara Y, Kubo T, et al. Impaired T- and NK-cell reconstitution after haploidentical HCT with posttransplant cyclophosphamide. Blood Adv. 2021;5:352–64.
Duléry R, Mohty R, Labopin M, Sestili S, Malard F, Brissot E, et al. Early cardiac toxicity associated with post-transplant cyclophosphamide in allogeneic stem cell transplantation. JACC CardioOncol. 2021;3:250–9.
Solomon SR, Sizemore CA, Sanacore M, Zhang X, Brown S, Holland HK, et al. Haploidentical transplantation using T cell replete peripheral blood stem cells and myeloablative conditioning in patients with high-risk hematologic malignancies who lack conventional donors is well tolerated and produces excellent relapse-free survival: results of a prospective phase II trial. Biol Blood Marrow Transplant J Am Soc Blood Marrow Transpl. 2012;18:1859–66.
Ruggeri A, Roth-Guepin G, Battipaglia G, Mamez AC, Malard F, Gomez A, et al. Incidence and risk factors for hemorrhagic cystitis in unmanipulated haploidentical transplant recipients. Transpl Infect Dis J Transpl Soc. 2015;17:822–30.
Copelan OR, Sanikommu SR, Trivedi JS, Butler C, Ai J, Ragon BK, et al. Higher incidence of hemorrhagic cystitis following haploidentical related donor transplantation compared with matched related donor transplantation. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2019;25:785–90.
Mohty R, Brissot E, Battipaglia G, Ruggeri A, Dulery R, Bonnin A, et al. Infectious complications after post-transplantation cyclophosphamide and anti-thymocyte globulin-based haploidentical stem cell transplantation. Br J Haematol. 2019;187:e64–8.
Ishida S, Doki N, Shingai N, Yoshioka K, Kakihana K, Sakamaki H, et al. The clinical features of fatal cyclophosphamide-induced cardiotoxicity in a conditioning regimen for allogeneic hematopoietic stem cell transplantation (allo-HSCT). Ann Hematol. 2016;95:1145–50.
Braverman AC, Antin JH, Plappert MT, Cook EF, Lee RT. Cyclophosphamide cardiotoxicity in bone marrow transplantation: a prospective evaluation of new dosing regimens. J Clin Oncol J Am Soc Clin Oncol. 1991;9:1215–23.
Emadi A, Jones RJ, Brodsky RA. Cyclophosphamide and cancer: golden anniversary. Nat Rev Clin Oncol. 2009;6:638–47.
Wachsmuth LP, Patterson MT, Eckhaus MA, Venzon DJ, Kanakry CG. Optimized timing of post-transplantation cyclophosphamide in mhc-haploidentical murine hematopoietic cell transplantation. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2020;26:230–41.
Sugita J, Kamimura T, Ishikawa T, Ota S, Eto T, Kuroha T, et al. Reduced dose of posttransplant cyclophosphamide in HLA-haploidentical peripheral blood stem cell transplantation. Bone Marrow Transpl. 2021;56:596–604.
Soltermann Y, Heim D, Medinger M, Baldomero H, Halter JP, Gerull S, et al. Reduced dose of post-transplantation cyclophosphamide compared to ATG for graft-versus-host disease prophylaxis in recipients of mismatched unrelated donor hematopoietic cell transplantation: a single-center study. Ann Hematol. 2019;98:1485–93.
Zhang W, Gui R, Zu Y, Zhang B, Li Z, Zhang Y, et al. Reduced-dose post-transplant cyclophosphamide plus low-dose post-transplant anti-thymocyte globulin as graft-versus-host disease prophylaxis with fludarabine-busulfan-cytarabine conditioning in haploidentical peripheral blood stem cell transplantation: a multicentre, randomized controlled clinical trial. Br J Haematol. 2022; https://doi.org/10.1111/bjh.18483.
Kanate AS, Nagler A, Savani B. Summary of scientific and statistical methods, study endpoints and definitions for observational and registry-based studies in hematopoietic cell transplantation. Clin Hematol Int. 2019;2:2–4.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15:825–8.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National institutes of health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. the 2014 diagnosis and staging working group report. Biol Blood Marrow Transplant J Am Soc Blood Marrow Transpl. 2015;21:389–401.
Plana JC, Galderisi M, Barac A, Ewer MS, Ky B, Scherrer-Crosbie M, et al. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2014;15:1063–93.
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2019;25:625–38.
Bedi A, Miller CB, Hanson JL, Goodman S, Ambinder RF, Charache P, et al. Association of BK virus with failure of prophylaxis against hemorrhagic cystitis following bone marrow transplantation. J Clin Oncol J Am Soc Clin Oncol. 1995;13:1103–9.
Cesaro S, Dalianis T, Hanssen Rinaldo C, Koskenvuo M, Pegoraro A, Einsele H, et al. ECIL guidelines for the prevention, diagnosis and treatment of BK polyomavirus-associated haemorrhagic cystitis in haematopoietic stem cell transplant recipients. J Antimicrob Chemother. 2018;73:12–21.
Ruggeri A, Labopin M, Ciceri F, Mohty M, Nagler A. Definition of GvHD-free, relapse-free survival for registry-based studies: an ALWP-EBMT analysis on patients with AML in remission. Bone Marrow Transpl. 2016;51:610–1.
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME. et al. Validation and refinement of the disease risk index for allogeneic stem cell transplantation. Blood. 2014;123:3664–71.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Fine J, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
Raiola AM, Dominietto A, Ghiso A, Di Grazia C, Lamparelli T, Gualandi F, et al. Unmanipulated haploidentical bone marrow transplantation and posttransplantation cyclophosphamide for hematologic malignancies after myeloablative conditioning. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2013;19:117–22.
Kasamon YL, Bolaños-Meade J, Prince GT, Tsai HL, McCurdy SR, Kanakry JA, et al. Outcomes of nonmyeloablative HLA-haploidentical blood or marrow transplantation with high-dose post-transplantation cyclophosphamide in older adults. J Clin Oncol J Am Soc Clin Oncol. 2015;33:3152–61.
Santoro N, Labopin M, Giannotti F, Ehninger G, Niederwieser D, Brecht A, et al. Unmanipulated haploidentical in comparison with matched unrelated donor stem cell transplantation in patients 60 years and older with acute myeloid leukemia: a comparative study on behalf of the ALWP of the EBMT. J Hematol OncolJ Hematol Oncol. 2018;11:55.
Bashey ZA, Zhang X, Brown S, Jackson K, Morris LE, Holland HK, et al. Comparison of outcomes following transplantation with T-replete HLA-haploidentical donors using post-transplant cyclophosphamide to matched related and unrelated donors for patients with AML and MDS aged 60 years or older. Bone Marrow Transpl. 2018;53:756–63.
Devillier R, Legrand F, Rey J, Castagna L, Fürst S, Granata A, et al. HLA-Matched Sibling versus Unrelated versus haploidentical related donor allogeneic hematopoietic stem cell transplantation for patients aged over 60 years with acute myeloid leukemia: a single-center donor comparison. Biol Blood Marrow Transplant J Am Soc Blood Marrow Transpl. 2018;24:1449–54.
Duléry R, Bastos J, Paviglianiti A, Malard F, Brissot E, Battipaglia G, et al. Thiotepa, Busulfan, and Fludarabine conditioning regimen in T cell-replete HLA-haploidentical hematopoietic stem cell transplantation. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2019;25:1407–15.
Memoli M, Paviglianiti A, Malard F, Battipaglia G, Brissot E, Médiavilla C, et al. Thiotepa-busulfan-fludarabine as a conditioning regimen for patients with myelofibrosis undergoing allogeneic hematopoietic transplantation: a single center experience. Leuk Lymphoma. 2021;62:419–27.
Peric Z, Mohty R, Bastos J, Brissot E, Battipaglia G, Belhocine R, et al. Thiotepa and antithymocyte globulin-based conditioning prior to haploidentical transplantation with posttransplant cyclophosphamide in high-risk hematological malignancies. Bone Marrow Transpl. 2020;55:763–72.
El-Cheikh J, Devillier R, Dulery R, Massoud R, Al Chami F, Ghaoui N, et al. Impact of adding antithymocyte globulin to posttransplantation cyclophosphamide in haploidentical stem-cell transplantation. Clin Lymphoma Myeloma Leuk. 2020;20:617–23.
Acknowledgements
We thank all the clinicians, pharmacists, and the nursing staff for providing excellent care for our patients, especially Agnès Bonnin, Chiara De Philippis, Iacopo Mariotti, Barbara Sarina, and Armando Santoro.
Author information
Authors and Affiliations
Contributions
R Duléry and R Devillier designed the study, collected, assembled, and analyzed data, performed statistical analysis, and wrote the manuscript. MM, DB, and SF designed the study, supervised research, analyzed data, and commented on the manuscript. CG, DM, and AB collected, assembled, and analyzed data, recruited patients, and commented on the manuscript. SE helped designing the study, analyzed data, and commented on the manuscript. AG, SH, VM, CC, FM, EB, SS, AB, ZVW, RB, LC, and SB recruited patients and commented the manuscript. All authors reviewed the manuscript and approved submission of the manuscript for publication purposes.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Duléry, R., Goudet, C., Mannina, D. et al. Reduced post-transplant cyclophosphamide doses in haploidentical hematopoietic cell transplantation for elderly patients with hematological malignancies. Bone Marrow Transplant 58, 386–392 (2023). https://doi.org/10.1038/s41409-022-01908-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01908-y
This article is cited by
-
Posttransplant cyclophosphamide in unrelated and related peripheral blood stem cell transplantation from HLA-matched and 1 allele mismatched donor
Bone Marrow Transplantation (2024)
-
Post-transplant cyclophosphamide at 80 mg/kg with low dose post-engraftment anti-thymocyte globulin in haploidentical transplantation with myeloablative conditioning
Bone Marrow Transplantation (2024)
-
Posttransplant cyclophosphamide beyond haploidentical transplantation
Annals of Hematology (2024)
-
Peripheral blood haploidentical hematopoietic cell transplantation for patients aged 70 years and over with acute myeloid leukemia or high-risk myelodysplastic syndrome
Bone Marrow Transplantation (2024)
-
Reduced post-transplant cyclophosphamide dose with antithymocyte globulin in peripheral blood stem cell haploidentical transplantation
Bone Marrow Transplantation (2023)