Abstract
Background
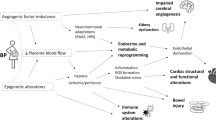
Intrauterine exposure to hypertensive disorders of pregnancy (HDP) may increase the risk of neuropsychiatric disorders. This investigation examined for associations between maternal HDP and febrile seizures (FS) in offspring by the age of three years.
Methods
The present cohort study analyzed data from the Japan Environment and Children’s Study, a large national birth cohort. We included mother-child pairs recruited between January 2011 and March 2014. Information regarding maternal HDP, the presence of FS in offspring up to 3 years of age, and potential confounding factors were assessed using written questionnaires administered to mothers.
Results
A total of 77,699 mother-child dyads were analyzed. The prevalence of FS was 8.4% in children without HDP exposure, 10.6% in those exposed to mild HDP, and 10.4% in those with severe HDP exposure. Among children with full-term birth, logistic regression analysis indicated that exposure to mild or severe HDP was significantly associated with a higher incidence of FS (adjusted odds ratio [95% confidence interval]: 1.27 [1.05–1.53] and 1.27 [0.90–1.78], respectively, P for trend = 0.008), compared with children without HDP exposure.
Conclusion
In children with full-term birth, intrauterine exposure to HDP was significantly associated with FS by the age of three years.
Impact
-
This study revealed a significant association between intrauterine exposure to hypertensive disorders of pregnancy (HDP) and the subsequent development of febrile seizures (FS) in offspring by three years.
-
This increased incidence of FS by HDP was independent of preterm birth status.
-
This is the first large nationwide birth cohort study showing the impact of intrauterine exposure to HDP on FS in early childhood.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 14 print issues and online access
$259.00 per year
only $18.50 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
Data are unsuitable for public deposition due to ethical restrictions and legal framework of Japan. It is prohibited by the Act on the Protection of Personal Information (Act No. 57 of 30 May 2003, amendment on 9 September 2015) to publicly deposit the data containing personal information. Ethical Guidelines for Medical and Health Research Involving Human Subjects enforced by the Japan Ministry of Education, Culture, Sports, Science and Technology and the Ministry of Health, Labour and Welfare also restricts the open sharing of the epidemiologic data. All inquiries about access to data should be sent to: jecs-en@nies.go.jp. The person responsible for handling enquiries sent to this e-mail address is Dr. Shoji F. Nakayama, JECS Programme Office, National Institute for Environmental Studies.
References
Duffner, P. K. et al. Neurodiagnostic evaluation of the child with a simple febrile seizure. Pediatrics 127, 389–394 (2011).
Sugai, K. Current management of febrile seizures in Japan: An overview. Brain Dev. 32, 64–70 (2010).
Tsuboi, T. Epidemiology of febrile and afebrile convulsions in children in Japan. Neurology 34, 175–181 (1984).
Vestergaard, M., Pedersen, C. B., Sidenius, P., Olsen, J. & Christensen, J. The long-term risk of epilepsy after febrile seizures in susceptible subgroups. Am. J. Epidemiol. 165, 911–918 (2007).
Annegers, J. F., Hauser, W. A., Shirts, S. B. & Kurland, L. T. Factors Prognostic of Unprovoked Seizures after Febrile Convulsions. N. Engl. J. Med. 316, 493–498 (1987).
Vestergaard, M. & Christensen, J. Register-based studies on febrile seizures in Denmark. Brain Dev. 31, 372–377 (2009).
Ateşoğlu, M., İnce, T., Lüleci, D., Ergör, A. & Aydın, A. Sociodemographic risk factors for febrile seizures: A school-based study from Izmir, Turkey. Seizure 61, 45–49 (2018).
Umesawa, M. & Kobashi, G. Epidemiology of hypertensive disorders in pregnancy: prevalence, risk factors, predictors and prognosis. Hypertens. Res. 40, 213–220 (2017).
Gumusoglu, S. B., Chilukuri, A. S. S., Santillan, D. A., Santillan, M. K. & Stevens, H. E. Neurodevelopmental Outcomes of Prenatal Preeclampsia Exposure. Trends Neurosci. 43, 253–268 (2020).
Vestergaard, M. et al. Pre-eclampsia and febrile convulsions. Arch. Dis. Child 88, 726–727 (2003).
Ishitsuka, K. et al. Japan Environment and Children’s Study: backgrounds, activities, and future directions in global perspectives. Environ. Health Prev. Med. 22, 61 (2017).
Kawamoto, T. et al. Rationale and study design of the Japan environment and children’s study (JECS). BMC Public Health 14, 25 (2014).
Michikawa, T. et al. Baseline Profile of Participants in the Japan Environment and Children’s Study (JECS). J. Epidemiol. 28, 99–104 (2018).
Watanabe, K., Naruse, K., Tanaka, K., Metoki, H. & Suzuki, Y. Outline of Definition and Classification of “Pregnancy induced Hypertension (PIH)”. Hypertens. Res. Pregnancy 1, 3–4 (2013).
Choi, Y. J. et al. Febrile seizures: Are they truly benign? Longitudinal analysis of risk factors and future risk of afebrile epileptic seizure based on the national sample cohort in South Korea, 2002–2013. Seizure 64, 77–83 (2019).
Vahidnia, F., Eskenazi, B. & Jewell, N. Maternal smoking, alcohol drinking, and febrile convulsion. Seizure - Eur. J. Epilepsy 17, 320–326 (2008).
Dalbem, J. S., Siqueira, H. H., Espinosa, M. M. & Alvarenga, R. P. Febrile seizures: A population-based study. J. Pediatr. (Rio J.) 91, 529–534 (2015).
Pavlidou, E., Hagel, C. & Panteliadis, C. Febrile seizures: Recent developments and unanswered questions. Child’s Nerv. Syst. 29, 2011–2017 (2013).
Newman, T. B., Wu, Y. W., Kuzniewicz, M. W., Grimes, B. A. & Mcculloch, C. E. Childhood Seizures After Phototherapy. Pediatrics 142, e20180648 (2018).
McNulty, H. et al. Effect of continued folic acid supplementation beyond the first trimester of pregnancy on cognitive performance in the child: a follow-up study from a randomized controlled trial (FASSTT Offspring Trial). BMC Med. 17, 196 (2019).
Huang, C. C. et al. Risk factors for a first febrile convulsion in children: A population study in southern Taiwan. Epilepsia 40, 719–725 (1999).
Gillberg, C., Lundström, S., Fernell, E., Nilsson, G. & Neville, B. Febrile Seizures and Epilepsy: Association With Autism and Other Neurodevelopmental Disorders in the Child and Adolescent Twin Study in Sweden. Pediatr. Neurol. 74, 80–86 (2017).
Okai, T. Standardization for ultrasonographic measurement of fetus and Japanese standard. Jpn J. Med Ultrason. 30, 415–440 (2003).
Mitsuda, N., Hosokawa, T., Eitoku, M., Fujieda, M. & Suganuma, N. Breastfeeding and risk of febrile seizures in infants: The Japan Environment and Children’s Study. Brain Dev. 41, 839–847 (2019).
Nakamura, M. et al. Association of cesarean birth with prevalence of functional constipation in toddlers at 3 years of age: results from the Japan Environment and Children’s Study (JECS). BMC Pediatr. 21, 419 (2021).
Goldenberg, R. L., Culhane, J. F., Iams, J. D. & Romero, R. Epidemiology and causes of preterm birth. Lancet 371, 75–84 (2008).
Dachew, B. A., Mamun, A., Maravilla, J. C. & Alati, R. Pre-eclampsia and the risk of autism-spectrum disorder in offspring: Meta-analysis. Br. J. Psychiatry 212, 142–147 (2018).
Mann, J. R., McDermott, S., Bao, H., Hardin, J. & Gregg, A. Pre-eclampsia, birth weight, and autism spectrum disorders. J. Autism Dev. Disord. 40, 548–554 (2010).
Dachew, B. A., Scott, J. G., Mamun, A. & Alati, R. Pre-eclampsia and the risk of attention-deficit/hyperactivity disorder in offspring: Findings from the ALSPAC birth cohort study. Psychiatry Res. 272, 392–397 (2019).
Morsing, E. & Maršál, K. Pre-eclampsia-An additional risk factor for cognitive impairment at school age after intrauterine growth restriction and very preterm birth. Early Hum. Dev. 90, 99–101 (2014).
Johnson, S. et al. Neurodevelopmental outcomes following late and moderate prematurity: A population-based cohort study. Arch. Dis. Child Fetal Neonatal. Ed. 100, F301–F308 (2015).
Heikura, U. et al. Maternal hypertensive disorders during pregnancy and mild cognitive limitations in the offspring. Paediatr. Perinat. Epidemiol. 27, 188–198 (2013).
Nahum Sacks, K. et al. Long-term neuropsychiatric morbidity in children exposed prenatally to preeclampsia. Early Hum. Dev. 130, 96–100 (2019).
Kajantie, E., Eriksson, J. G., Osmond, C., Thornburg, K. & Barker, D. J. P. Pre-eclampsia is associated with increased risk of stroke in the adult offspring the Helsinki birth cohort study. Stroke 40, 1176–1180 (2009).
Rees, S., Harding, R. & Walker, D. An adverse intrauterine environment: implications for injury and altered development of the brain. Int. J. Dev. Neurosci. 26, 3–11 (2008).
Walker, C. K. et al. Preeclampsia, placental insufficiency, and autism spectrum disorder or developmental delay. JAMA Pediatr. 169, 154–162 (2015).
Camfield P. R., et al. Febrile seizures and genetic epilepsy with febrile seizures plus. In Epileptic syndrome in infancy, Childhood and Adolescence (6th ed) (Bureau M., Genton P., Dravet C., eds) 119-137 (John Libbey Eurotext Ltd, 2019).
Seckl, J. R. & Holmes, M. C. Mechanisms of disease: glucocorticoids, their placental metabolism and fetal ‘programming’ of adult pathophysiology. Nat. Clin. Pr. Endocrinol. Metab. 3, 479–488 (2007).
Aufdenblatten, M. et al. Prematurity Is Related to High Placental Cortisol in Preeclampsia. Pediatr. Res. 65, 198–202 (2009).
McCalla, C. O., Nacharaju, V. L., Muneyyirci-Delale, O., Glasgow, S. & Feldman, J. G. Placental 11-beta-hydroxysteroid dehydrogenase activity in normotensive and pre-eclamptic pregnancies. Steroids 63, 511–515 (1998).
Thébault-Dagher, F. et al. Febrile seizure incidence and age at first occurrence are associated with changes in placental normalized gene expression: the ‘3D’ pregnancy cohort study. J. Neuroendocrinol. 33, e13046 (2021).
Kay, V. R., Rätsep, M. T., Figueiró-Filho, E. A. & Croy, B. A. Preeclampsia may influence offspring neuroanatomy and cognitive function: A role for placental growth factor. Biol. Reprod. 101, 271–283 (2019).
Acknowledgements
We sincerely thank all of the participants in this study and all individuals involved in data collection. The Japan Environment and Children’s Study was funded by the Ministry of the Environment, Japan. The findings and conclusions of this study are solely those of the authors and do not represent the official views of the above government.
Author information
Authors and Affiliations
Consortia
Contributions
T.N. contributed to the data collection. M.Y., N.M., and T.F. designed the study. M.Y. and N.M. performed statistical analysis and interpretation of data. M.Y. drafted the article. T.F., N.M., T.S., H.T., K.H., T.T., T.N., and the members of the Japan Environment and Children’s Study Group revised the manuscript critically for important intellectual content. All members reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
Written informed consent was obtained from all participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yabe, M., Fukuyama, T., Motoki, N. et al. Hypertensive disorders of pregnancy increase the incidence of febrile seizures in offspring. Pediatr Res (2024). https://doi.org/10.1038/s41390-024-03057-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41390-024-03057-y