Abstract
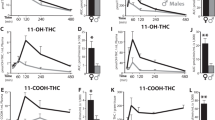
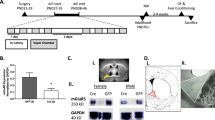
Despite the belief that cannabis is relatively harmless, exposure during adolescence is associated with increased risk of developing several psychopathologies in adulthood. In addition to the high levels of use amongst teenagers, the potency of ∆-9-tetrahydrocannabinol (THC) has increased more than fourfold compared to even twenty years ago, and it is unclear whether potency influences the presentation of THC-induced behaviors. Expanded knowledge about the impact of adolescent THC exposure, especially high dose, is important to delineating neural networks and molecular mechanisms underlying psychiatric risk. Here, we observed that repeated exposure to low (1.5 mg/kg) and high (5 mg/kg) doses of THC during adolescence in male rats produced divergent effects on behavior in adulthood. Whereas low dose rats showed greater sensitivity to reward devaluation and also self-administered more heroin, high dose animals were significantly more reactive to social isolation stress. RNA sequencing of the basolateral amygdala, a region linked to reward processing and stress, revealed significant perturbations in transcripts and gene networks related to synaptic plasticity and HPA axis that were distinct to THC dose as well as stress. In silico single-cell deconvolution of the RNAseq data revealed a significant reduction of astrocyte-specific genes related to glutamate regulation in stressed high dose animals, a result paired anatomically with greater astrocyte-to-neuron ratios and hypotrophic astrocytes. These findings emphasize the importance of dose and behavioral state on the presentation of THC-related behavioral phenotypes in adulthood and dysregulation of astrocytes as an interface for the protracted effects of high dose THC and subsequent stress sensitivity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
UNODC. World Drug Report 2019: United Nations Office on Drugs and Crime; 2019. Report no. Sales No. E.19.XI.8.
Johnston LD, Miech, RA, O’Malley, PM, Bachman, JG, Schulenberg, JE, Patrick, ME. Monitoring the Future national survey results on drug use, 1975–2019: Overview, key findings on adolescent drug use. Ann Arbor, MI: The University of Michigan; 2020.
Schulenberg J, Johnston L, O’Malley P, Bachman J, Miech R, Patrick ME. Monitoring the Future national survey results on drug use, 1975–2019: Volume II, college students and adults ages 19–60. Institute for Social Research. Ann Arbor, MI: University of Michigan; 2020.
Renard J, Krebs MO, Le Pen G, Jay TM. Long-term consequences of adolescent cannabinoid exposure in adult psychopathology. Front Neurosci. 2014;8:361.
Renard J, Rosen LG, Loureiro M, De Oliveira C, Schmid S, Rushlow WJ, et al. Adolescent cannabinoid exposure induces a persistent sub-cortical hyper-dopaminergic state and associated molecular adaptations in the prefrontal cortex. Cereb Cortex. 2017;27:1297–310.
Renard J, Szkudlarek HJ, Kramar CP, Jobson CEL, Moura K, Rushlow WJ, et al. Adolescent THC exposure causes enduring prefrontal cortical disruption of GABAergic inhibition and dysregulation of sub-cortical dopamine function. Sci Rep. 2017;7:11420.
Rubino T, Vigano D, Realini N, Guidali C, Braida D, Capurro V, et al. Chronic delta 9-tetrahydrocannabinol during adolescence provokes sex-dependent changes in the emotional profile in adult rats: behavioral and biochemical correlates. Neuropsychopharmacology. 2008;33:2760–71.
Prini P, Rusconi F, Zamberletti E, Gabaglio M, Penna F, Fasano M, et al. Adolescent THC exposure in female rats leads to cognitive deficits through a mechanism involving chromatin modifications in the prefrontal cortex. J Psychiatry Neurosci. 2017;42:170082.
Stopponi S, Soverchia L, Ubaldi M, Cippitelli A, Serpelloni G, Ciccocioppo R. Chronic THC during adolescence increases the vulnerability to stress-induced relapse to heroin seeking in adult rats. Eur Neuropsychopharmacol. 2014;24:1037–45.
Zamberletti E, Beggiato S, Steardo L Jr., Prini P, Antonelli T, Ferraro L, et al. Alterations of prefrontal cortex GABAergic transmission in the complex psychotic-like phenotype induced by adolescent delta-9-tetrahydrocannabinol exposure in rats. Neurobiol Dis. 2014;63:35–47.
Abela AR, Rahbarnia A, Wood S, Le AD, Fletcher PJ. Adolescent exposure to Delta9-tetrahydrocannabinol delays acquisition of paired-associates learning in adulthood. Psychopharmacology. 2019;236:1875–86.
Llorente-Berzal A, Puighermanal E, Burokas A, Ozaita A, Maldonado R, Marco EM, et al. Sex-dependent psychoneuroendocrine effects of THC and MDMA in an animal model of adolescent drug consumption. PLoS One. 2013;8:e78386.
Rubino T, Realini N, Braida D, Guidi S, Capurro V, Vigano D, et al. Changes in hippocampal morphology and neuroplasticity induced by adolescent THC treatment are associated with cognitive impairment in adulthood. Hippocampus. 2009;19:763–72.
Ellgren M, Spano SM, Hurd YL. Adolescent cannabis exposure alters opiate intake and opioid limbic neuronal populations in adult rats. Neuropsychopharmacology. 2007;32:607–15.
Lecca D, Scifo A, Pisanu A, Valentini V, Piras G, Sil A, et al. Adolescent cannabis exposure increases heroin reinforcement in rats genetically vulnerable to addiction. Neuropharmacology. 2020;166:107974.
Tomasiewicz HC, Jacobs MM, Wilkinson MB, Wilson SP, Nestler EJ, Hurd YL. Proenkephalin mediates the enduring effects of adolescent cannabis exposure associated with adult opiate vulnerability. Biol Psychiatry. 2012;72:803–10.
Scherma M, Dessi C, Muntoni AL, Lecca S, Satta V, Luchicchi A, et al. Adolescent Delta(9)-Tetrahydrocannabinol exposure alters WIN55,212-2 self-administration in adult rats. Neuropsychopharmacology. 2016;41:1416–26.
Friedman AL, Meurice C, Jutkiewicz EM. Effects of adolescent Delta9-tetrahydrocannabinol exposure on the behavioral effects of cocaine in adult Sprague–Dawley rats. Exp Clin Psychopharmacol. 2019;27:326–37.
Chandra S, Radwan MM, Majumdar CG, Church JC, Freeman TP, ElSohly MA. New trends in cannabis potency in USA and Europe during the last decade (2008–2017). Eur Arch Psychiatry Clin Neurosci. 2019;269:5–15.
ElSohly MA, Mehmedic Z, Foster S, Gon C, Chandra S, Church JC. Changes in cannabis potency over the last 2 decades (1995–2014): analysis of current data in the United States. Biol Psychiatry. 2016;79:613–9.
Cash MC, Cunnane K, Fan C, Romero-Sandoval EA. Mapping cannabis potency in medical and recreational programs in the United States. PLoS One. 2020;15:e0230167.
Keeley RJ, Trow J, Bye C, McDonald RJ. Part II: strain- and sex-specific effects of adolescent exposure to THC on adult brain and behaviour: variants of learning, anxiety and volumetric estimates. Behav Brain Res. 2015;288:132–52.
Silva L, Black R, Michaelides M, Hurd YL, Dow-Edwards D. Sex and age specific effects of delta-9-tetrahydrocannabinol during the periadolescent period in the rat: the unique susceptibility of the prepubescent animal. Neurotoxicol Teratol. 2016;58:88–100.
Kruse LC, Cao JK, Viray K, Stella N, Clark JJ. Voluntary oral consumption of Delta(9)-tetrahydrocannabinol by adolescent rats impairs reward-predictive cue behaviors in adulthood. Neuropsychopharmacology. 2019;44:1406–14.
Weidenfeld J, Feldman S, Mechoulam R. Effect of the brain constituent anandamide, a cannabinoid receptor agonist, on the hypothalamo-pituitary-adrenal axis in the rat. Neuroendocrinology. 1994;59:110–2.
DeVuono MV, La Caprara O, Sullivan MT, Bath A, Petrie GN, Limebeer CL, et al. Role of the stress response and the endocannabinoid system in Delta(9)-tetrahydrocannabinol (THC)-induced nausea. Psychopharmacology 2020;237:2187–99.
McLaughlin RJ, Hill MN, Gorzalka BB. Monoaminergic neurotransmission contributes to cannabinoid-induced activation of the hypothalamic-pituitary-adrenal axis. Eur J Pharm. 2009;624:71–6.
Miller ML, Chadwick B, Dickstein DL, Purushothaman I, Egervari G, Rahman T, et al. Adolescent exposure to Delta(9)-tetrahydrocannabinol alters the transcriptional trajectory and dendritic architecture of prefrontal pyramidal neurons. Mol Psychiatry. 2019;24:588–600.
Saravia R, Ten-Blanco M, Julia-Hernandez M, Gagliano H, Andero R, Armario A, et al. Concomitant THC and stress adolescent exposure induces impaired fear extinction and related neurobiological changes in adulthood. Neuropharmacology. 2019;144:345–57.
Walker EF, Sabuwalla Z, Huot R. Pubertal neuromaturation, stress sensitivity, and psychopathology. Dev Psychopathol. 2004;16:807–24.
Allsop SA, Vander Weele CM, Wichmann R, Tye KM. Optogenetic insights on the relationship between anxiety-related behaviors and social deficits. Front Behav Neurosci. 2014;8:241.
Greenwood BN, Strong PV, Fleshner M. Lesions of the basolateral amygdala reverse the long-lasting interference with shuttle box escape produced by uncontrollable stress. Behav Brain Res. 2010;211:71–6.
Wassum KM, Izquierdo A. The basolateral amygdala in reward learning and addiction. Neurosci Biobehav Rev. 2015;57:271–83.
Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014;15:550.
Kuleshov MV, Jones MR, Rouillard AD, Fernandez NF, Duan Q, Wang Z, et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016;44:W90–97.
Song WM, Zhang B. Multiscale embedded gene co-expression network analysis. PLoS Comput Biol. 2015;11:e1004574.
Newman AM, Liu CL, Green MR, Gentles AJ, Feng W, Xu Y, et al. Robust enumeration of cell subsets from tissue expression profiles. Nat Methods. 2015;12:453–7.
Newman AM, Steen CB, Liu CL, Gentles AJ, Chaudhuri AA, Scherer F, et al. Determining cell type abundance and expression from bulk tissues with digital cytometry. Nat Biotechnol. 2019;37:773–82.
Zhang WH, Zhang JY, Holmes A, Pan BX. Amygdala circuit substrates for stress adaptation and adversity. Biol Psychiatry. 2021;89:847–56.
Zhang Y, Chen K, Sloan SA, Bennett ML, Scholze AR, O’Keeffe S, et al. An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J Neurosci. 2014;34:11929–47.
Bhattacherjee A, Djekidel MN, Chen R, Chen W, Tuesta LM, Zhang Y. Cell type-specific transcriptional programs in mouse prefrontal cortex during adolescence and addiction. Nat Commun. 2019;10:4169.
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods. 2012;9:676–82.
Team J. JASP. 0.14.1 edn: Amsterdam, The Netherlands, 2020.
Badiani A, Caprioli D, De Pirro S. Opposite environmental gating of the experienced utility (‘liking’) and decision utility (‘wanting’) of heroin versus cocaine in animals and humans: implications for computational neuroscience. Psychopharmacology. 2019;236:2451–71.
Acuff SF, Amlung M, Dennhardt AA, MacKillop J, Murphy JG. Experimental manipulations of behavioral economic demand for addictive commodities: a meta-analysis. Addiction. 2020;115:817–31.
Ruiz CM, Torrens A, Castillo E, Perrone CR, Cevallos J, Inshishian VC, et al. Pharmacokinetic, behavioral, and brain activity effects of Delta(9)-tetrahydrocannabinol in adolescent male and female rats. Neuropsychopharmacology. 2021;46:959–69.
Turner AI, Smyth N, Hall SJ, Torres SJ, Hussein M, Jayasinghe SU, et al. Psychological stress reactivity and future health and disease outcomes: A systematic review of prospective evidence. Psychoneuroendocrinology. 2020;114:104599.
Koob GF. The dark side of emotion: the addiction perspective. Eur J Pharm. 2015;753:73–87.
Bara A, Ferland JN, Rompala G, Szutorisz H, Hurd YL, Cannabis and synaptic reprogramming of the developing brain. Nat Rev Neurosci. 2021;22:423–38.
Felix-Ortiz AC, Burgos-Robles A, Bhagat ND, Leppla CA, Tye KM. Bidirectional modulation of anxiety-related and social behaviors by amygdala projections to the medial prefrontal cortex. Neuroscience. 2016;321:197–209.
Young CE, Tong Q. Corticotropin releasing hormone signaling in the bed nuclei of the Stria terminalis as a link to maladaptive behaviors. Front Neurosci. 2021;15:642379.
Kofuji P, Araque A. G-Protein-Coupled receptors in astrocyte-neuron communication. Neuroscience 2020;456:71–84.
Navarrete M, Araque A. Endocannabinoids mediate neuron-astrocyte communication. Neuron. 2008;57:883–93.
Durkee CA, Araque A. Diversity and specificity of astrocyte-neuron communication. Neuroscience. 2019;396:73–8.
Murphy-Royal C, Gordon GR, Bains JS. Stress-induced structural and functional modifications of astrocytes-Further implicating glia in the central response to stress. Glia. 2019;67:1806–20.
Cordeiro JL, Neves JD, Vizuete AF, Aristimunha D, Pedroso TA, Sanches EF, et al. Arundic Acid (ONO-2506), an inhibitor of S100B protein synthesis, prevents neurological deficits and brain tissue damage following intracerebral hemorrhage in male Wistar rats. Neuroscience. 2020;440:97–112.
O’Leary LA, Mechawar N. Implication of cerebral astrocytes in major depression: a review of fine neuroanatomical evidence in humans. Glia. 2021;69:2077–99.
Chye Y, Christensen E, Yucel M. Cannabis use in adolescence: a review of neuroimaging findings. J Dual Diagn. 2020;16:83–105.
Munshi S, Rosenkranz JA, Caccamise A, Wolf ME, Corbett CM, Loweth JA. Cocaine and chronic stress exposure produce an additive increase in neuronal activity in the basolateral amygdala. Addict Biol. 2021;26:e12848.
Glass MJ, Kruzich PJ, Colago EE, Kreek MJ, Pickel VM. Increased AMPA GluR1 receptor subunit labeling on the plasma membrane of dendrites in the basolateral amygdala of rats self-administering morphine. Synapse. 2005;58:1–12.
Shiina N, Yamaguchi K, Tokunaga M. RNG105 deficiency impairs the dendritic localization of mRNAs for Na+/K+ ATPase subunit isoforms and leads to the degeneration of neuronal networks. J Neurosci. 2010;30:12816–30.
Kusakawa Y, Mikawa S, Sato K. BMP5 expression in the adult rat brain. Neuroscience. 2015;284:972–87.
Lee SE, Lee Y, Lee GH. The regulation of glutamic acid decarboxylases in GABA neurotransmission in the brain. Arch Pharm Res. 2019;42:1031–9.
Lv QY, Chen MM, Li Y, Yu Y, Liao H, Brain circuit dysfunction in specific symptoms of depression. Eur J Neurosci. 2021. https://doi.org/10.1111/ejn.15221. Epub ahead of print.
Stevens LH, Spencer HM, Turkington D. Identifying four subgroups of trauma in psychosis: vulnerability, psychopathology, and treatment. Front Psychiatry. 2017;8:21.
Roozendaal B, McReynolds JR, McGaugh JL. The basolateral amygdala interacts with the medial prefrontal cortex in regulating glucocorticoid effects on working memory impairment. J Neurosci. 2004;24:1385–92.
Gunduz-Cinar O, Hill MN, McEwen BS, Holmes A. Amygdala FAAH and anandamide: mediating protection and recovery from stress. Trends Pharm Sci. 2013;34:637–44.
Perusini JN, Meyer EM, Long VA, Rau V, Nocera N, Avershal J, et al. Induction and expression of fear sensitization caused by acute traumatic stress. Neuropsychopharmacology. 2016;41:45–57.
Levesque D, Rouillard C. Nur77 and retinoid X receptors: crucial factors in dopamine-related neuroadaptation. Trends Neurosci. 2007;30:22–30.
Quinnies KM, Doyle TJ, Kim KH, Rissman EF. Transgenerational effects of Di-(2-Ethylhexyl) Phthalate (DEHP) on stress hormones and behavior. Endocrinology. 2015;156:3077–83.
Dieterich A, Floeder J, Stech K, Lee J, Srivastava P, Barker DJ, et al. Activation of Basolateral amygdala to nucleus accumbens projection neurons attenuates chronic corticosterone-induced behavioral deficits in male mice. Front Behav Neurosci. 2021;15:643272.
Pecina S, Schulkin J, Berridge KC. Nucleus accumbens corticotropin-releasing factor increases cue-triggered motivation for sucrose reward: paradoxical positive incentive effects in stress? BMC Biol. 2006;4:8.
Sharp BM. Basolateral amygdala and stress-induced hyperexcitability affect motivated behaviors and addiction. Transl Psychiatry. 2017;7:e1194.
Garrido Zinn C, Clairis N, Silva Cavalcante LE, Furini CR, de Carvalho Myskiw J, Izquierdo I. Major neurotransmitter systems in dorsal hippocampus and basolateral amygdala control social recognition memory. Proc Natl Acad Sci USA. 2016;113:E4914–4919.
Kash TL, Nobis WP, Matthews RT, Winder DG. Dopamine enhances fast excitatory synaptic transmission in the extended amygdala by a CRF-R1-dependent process. J Neurosci. 2008;28:13856–65.
Puaud M, Higuera-Matas A, Brunault P, Everitt BJ, Belin D. The Basolateral amygdala to nucleus accumbens core circuit mediates the conditioned reinforcing effects of cocaine-paired cues on cocaine seeking. Biol Psychiatry. 2021;89:356–65.
Keistler CR, Hammarlund E, Barker JM, Bond CW, DiLeone RJ, Pittenger C, et al. Regulation of alcohol extinction and cue-induced reinstatement by specific projections among medial prefrontal cortex, nucleus accumbens, and Basolateral amygdala. J Neurosci. 2017;37:4462–71.
Jouroukhin Y, Zhu X, Shevelkin AV, Hasegawa Y, Abazyan B, Saito A, et al. Adolescent Delta(9)-tetrahydrocannabinol exposure and astrocyte-specific genetic vulnerability converge on nuclear factor-kappaB-Cyclooxygenase-2 signaling to impair memory in adulthood. Biol Psychiatry. 2019;85:891–903.
Zamberletti E, Gabaglio M, Grilli M, Prini P, Catanese A, Pittaluga A, et al. Long-term hippocampal glutamate synapse and astrocyte dysfunctions underlying the altered phenotype induced by adolescent THC treatment in male rats. Pharm Res. 2016;111:459–70.
Zuchero JB, Barres BA. Glia in mammalian development and disease. Development. 2015;142:3805–9.
Sollner JF, Leparc G, Hildebrandt T, Klein H, Thomas L, Stupka E, et al. An RNA-Seq atlas of gene expression in mouse and rat normal tissues. Sci Data. 2017;4:170185.
Zhang X, Alnafisah RS, Hamoud AA, Shukla R, Wen Z, McCullumsmith RE, et al. Role of astrocytes in major neuropsychiatric disorders. Neurochem Res. 2021;46:2715–30.
Batiuk MY, Martirosyan A, Wahis J, de Vin F, Marneffe C, Kusserow C, et al. Identification of region-specific astrocyte subtypes at single cell resolution. Nat Commun. 2020;11:1220.
Livezey GT, Miller JM, Vogel WH. Plasma norepinephrine, epinephrine and corticosterone stress responses to restraint in individual male and female rats, and their correlations. Neurosci Lett. 1985;62:51–6.
Mathews IZ, Wilton A, Styles A, McCormick CM. Increased depressive behaviour in females and heightened corticosterone release in males to swim stress after adolescent social stress in rats. Behav Brain Res. 2008;190:33–40.
Dearing C, Morano R, Ptaskiewicz E, Mahbod P, Scheimann JR, Franco-Villanueva A, et al. Glucoregulation and coping behavior after chronic stress in rats: sex differences across the lifespan. Horm Behav. 2021;136:105060.
Przybyl KJ, Jenz ST, Lim PH, Ji MT, Wert SL, Luo W, et al. Genetic stress-reactivity, sex, and conditioning intensity affect stress-enhanced fear learning. Neurobiol Learn Mem. 2021;185:107523.
Schaack AK, Mocchi M, Przybyl KJ, Redei EE. Immediate stress alters social and object interaction and recognition memory in nearly isogenic rat strains with differing stress reactivity. Stress 2021:1–9.
Cacioppo JT, Cacioppo S. Social relationships and health: the toxic effects of perceived social isolation. Soc Pers Psychol Compass. 2014;8:58–72.
Ferland CL, Schrader LA. Cage mate separation in pair-housed male rats evokes an acute stress corticosterone response. Neurosci Lett. 2011;489:154–8.
Panksepp J. Brief social isolation, pain responsiveness, and morphine analgesia in young rats. Psychopharmacology. 1980;72:111–2.
Niesink RJ, van Ree JM. Short-term isolation increases social interactions of male rats: a parametric analysis. Physiol Behav. 1982;29:819–25.
Acknowledgements
We thank Drs. Qian Wang and Bin Zhang for their intellectual input on the MEGENA gene network analysis and Nayana Patel for assistance with the plasma corticosterone analysis.
Author information
Authors and Affiliations
Contributions
JMF and YLH designed the experiments. JMF, JAL, JEC, AL, MDF, and TOU conducted the experiments. RJE and GR provided computational genomics analyses for RNAseq data. JMF completed all behavioral and molecular analyses. JMF and YLH wrote the manuscript and all authors reviewed the manuscript and provided comments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ferland, JM.N., Ellis, R.J., Rompala, G. et al. Dose mediates the protracted effects of adolescent THC exposure on reward and stress reactivity in males relevant to perturbation of the basolateral amygdala transcriptome. Mol Psychiatry 28, 2583–2593 (2023). https://doi.org/10.1038/s41380-022-01467-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-022-01467-0
This article is cited by
-
Astrocytic transcriptional and epigenetic mechanisms of drug addiction
Journal of Neural Transmission (2024)
-
Long-term effects of THC exposure on reward learning and motivated behavior in adolescent and adult male rats
Psychopharmacology (2023)
-
Chronic adolescent exposure to cannabis in mice leads to sex-biased changes in gene expression networks across brain regions
Neuropsychopharmacology (2022)