Abstract
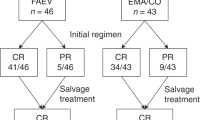
The management of chronic myeloid leukemia (CML) diagnosed during pregnancy is a rare and challenging situation. We report the treatment and outcome of 87 cases diagnosed in chronic phase from 2001–2022 derived from the largest international observational registry, supported by the European LeukemiaNet (ELN), of 400 pregnancies in 299 CML women. Normal childbirth occurred in 76% without an increased rate of birth abnormalities or life-threatening events, including in patients untreated or treated with interferon-α and/or imatinib in 2nd–3rd trimester. The low birth weight rate of 12% was comparable to that seen in the normal population. Elective and spontaneous abortions occurred in 21% and 3%, respectively. The complete hematologic response rate before labor was 95% with imatinib and 47% with interferon only. No disease progression during pregnancy was observed, 28% of the patients switched their therapy at varying times after delivery. Treatment options balance the efficacy and safety for mother and infant: interferon-α can commence in the 1st trimester and continued throughout in cases of good disease control and tolerability. Because of limited placental crossing, selected tyrosine kinase inhibitors (imatinib and nilotinib) seem to be safe and effective options in 2nd and 3rd trimester while hydroxycarbamide offers few benefits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The data supporting the findings of this study are not openly available to preserve individuals privacy under the European General Data Protection Regulation. However, they can be obtained from the corresponding author upon reasonable request and permission.
References
Chronic granulocytic leukaemia: comparison of radiotherapy and busulphan therapy. Report of the Medical Research Council’s working party for therapeutic trials in leukaemia. Br Med J. 1968;1:201–8
Law AD, Kim DDH, Lipton JH. Pregnancy: part of life in chronic myelogenous leukemia. Leuk Lymphoma. 2017;58:280–7.
Robertson HF, Apperley JF. Treatment of CML in pregnancy. Hematol Am Soc Hematol Educ Program. 2022;2022:123–8.
Abruzzese E, Mauro M, Apperley J, Chelysheva E. Tyrosine kinase inhibitors and pregnancy in chronic myeloid leukemia: opinion, evidence, and recommendations. Ther Adv Hematol. 2020;11:2040620720966120.
Chelysheva E, Turkina A. Risks and challenges of CML management during pregnancy: looking for a balanced decision. Eur J Hematol. 2019;102:378–9.
Berdman E. Family planning and pregnancy in patients with chronic myeloid leukemia. Curr Hematol Malignancy Rep. 2023;18:1–7.
Pye SM, Cortes J, Ault P, Hatfield A, Kantarjian H, Pilot R, et al. The effects of imatinib on pregnancy outcome. Blood. 2008;111:5505–8.
Cortes JE, Abruzzese E, Chelysheva E, Guha M, Wallis N, Apperley JF. The impact of dasatinib on pregnancy outcomes. Am J Hematol. 2015;90:1111–5.
Abruzzese E, Trawinska MM, de Fabritiis P, Baccarani M. Management of pregnant chronic myeloid leukemia patients. Expert Rev Hematol. 2016;9:781–91.
Cole S, Kantarjian H, Ault P, Cortés JE. Successful completion of pregnancy in a patient with chronic myeloid leukemia without active intervention: a case report and review of the literature. Clin Lymphoma Myeloma. 2009;9:324–7.
Iqbal J, Ali Z, Khan AU, Aziz Z. Pregnancy outcomes in patients with chronic myeloid leukemia treated with imatinib mesylate: short report from a developing country. Leuk Lymphoma. 2014;55:2109–13.
Jovelet C, Seck A, Mir O, Simasotchi C, Broutin S, Goffinet F, et al. Variation in transplacental transfer of tyrosine kinase inhibitors in the human perfused cotyledon model. Ann Oncol. 2015;26:1500–4.
Chelysheva E, Turkina A, Polushkina E, Shmakov R, Zeifman A, Aleshin S, et al. Placental transfer of tyrosine kinase inhibitors used for chronic myeloid leukemia treatment. Leuk Lymphoma. 2018;59:733–8.
Russell MA, Carpenter MW, Akhtar MS, Lagattuta TF, Egorin MJ. Imatinib mesylate and metabolite concentrations in maternal blood, umbilical cord blood, placenta and breast milk. J Perinatol. 2007;27:241–3.
Abruzzese E, Aureli S, Bondanini F, Ciccarone M, Cortis E, Di Paolo A, et al. Chronic myeloid leukemia and pregnancy: when dreams meet reality. State of the art, management and outcome of 41 cases, nilotinib placental transfer. J Clin Med. 2022;11:1801.
Cortes JE, Gambacorti-Passerini C, Deininger M, Abruzzese E, De Annuntis L, Brümmendorf TH. Pregnancy outcomes in patients treated with bosutinib. Int J Hematol Oncol. 2020;9:IJH26.
Alizadeh H, Jaafar H, Rajnics P, Khan MI, Kajtár B. Outcome of pregnancy in chronic myeloid leukaemia patients treated with tyrosine kinase inhibitors: short report from a single centre. Leuk Res. 2015;39:47–51.
Berveiller P, Andreoli A, Mir O, Anselem O, Delezoide AL, Sauvageon H, et al. A dramatic fetal outcome following transplacental transfer of dasatinib. Anticancer Drugs. 2012;23:754–7.
Lasica M, Willcox A, Burbury K, Ross DM, Branford S, Butler J, et al. The effect of tyrosine kinase inhibitor interruption and interferon use on pregnancy outcomes and long-term disease control in chronic myeloid leukemia. Leuk Lymphoma. 2019;60:1796–802.
Celiloglu M, Altunyurt S, Undar B. Hydroxyurea treatment for chronic myeloid leukemia during pregnancy. Acta Obstet Gynecol Scand. 2000;79:803–4.
Jain D, Atmapoojya P, Colah R, Lodha P. Sickle cell disease and pregnancy. Mediterr J Hematol Infect Dis. 2019;11:e2019040.
Rohilla M, Rai R, Yanamandra U, Chaudhary N, Malhotra P, Varma N, et al. Obstetric complications and management in chronic myeloid leukemia. Indian J Hematol Blood Transfus. 2016;32:62–6.
Hochhaus A, Baccarani M, Silver RT, Schiffer C, Apperley JF, Cervantes F, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966–84.
Assi R, Kantarjian H, Keating M, Pemmaraju N, Verstovsek S, Garcia-Manero G, et al. Management of chronic myeloid leukemia during pregnancy among patients treated with a tyrosine kinase inhibitor: a single-Center experience. Leuk Lymphoma. 2021;62:909–17.
Claudiani S, Gatenby A, Szydlo R, Nesr G, Abulafia AS, Palanicawandar R, et al. MR4 sustained for 12 months is associated with stable deep molecular responses in chronic myeloid leukemia. Haematologica. 2019;104:2206–14.
Pfirrmann M, Baccarani M, Saussele S, Guilhot J, Cervantes F, Ossenkoppele G, et al. Prognosis of long-term survival considering disease-specific death in patients with chronic myeloid leukemia. Leukemia. 2016;30:48–56.
Ali R, Ozkalemkas F, Kimya Y, Koksal N, Ozkocaman V, Gulten T, et al. Imatinib use during pregnancy and breast feeding: a case report and review of the literature. Arch Gynecol Obstet. 2009;280:169–75.
Burwick RM, Kuo K, Brewer D, Druker BJ. Maternal, fetal, and neonatal imatinib levels with treatment of chronic myeloid leukemia in pregnancy. Obstet Gynecol. 2017;129:831–4.
Egbe A, Uppu S, Lee S, Stroustrup A, Ho D, Srivastava S. Congenital malformations in the newborn population: a population study and analysis of the effect of sex and prematurity. Pediatr Neonatol. 2015;56:25–30.
Homma S, Messé SR, Rundek T, Sun YP, Franke J, Davidson K, et al. Patent foramen ovale. Nat Rev Dis Prim. 2016;2:15086.
Tripathi R, Mazmudar RS, Knusel KD, Ezaldein HH, Belazarian LT, Bordeaux JS, et al. Impact of congenital cutaneous hemangiomas on newborn care in the United States. Arch Dermatol Res. 2021;313:641–51.
Stirnemann J, Villar J, Salomon LJ, Ohuma E, Ruyan P, Altman DG, et al. International estimated fetal weight standards of the INTERGROWTH-21st Project. Ultrasound Obstet Gynecol. 2017;49:478–86.
Damhuis SE, Ganzevoort W, Gordijn SJ. Abnormal fetal growth: small for gestational age, fetal growth restriction, large for gestational age: definitions and epidemiology. Obstet Gynecol Clin N Am. 2021;48:267–79.
Al Kindi S, Dennison D, Pathare A. Imatinib in pregnancy. Eur J Haematol. 2005;74:535–7.
Barron SL. Birthweight and ethnicity. Br J Obstet Gynaecol. 1983;90:289–90.
Urquia ML. Variability in birthweight, birthweight charts, and adverse outcomes: Is the “right size” the right question? Paediatr Perinat Epidemiol. 2019;33:433–5.
Nardozza LM, Caetano AC, Zamarian AC, Mazzola JB, Silva CP, Marçal VM, et al. Fetal growth restriction: current knowledge. Arch Gynecol Obstet. 2017;295:1061–77.
Romo A, Carceller R, Tobajas J. Intrauterine growth retardation (IUGR): epidemiology and etiology. Pediatr Endocrinol Rev. 2009;6:332–6.
Hehlmann R, Heimpel H, Hasford J, Kolb HJ, Pralle H, Hossfeld DK, et al. Randomized comparison of interferon-alpha with busulfan and hydroxyurea in chronic myelogenous leukemia. The German CML Study Group. Blood. 1994;84:4064–77.
Hehlmann R, Lauseker M, Saussele S, Pfirrmann M, Krause S, Kolb HJ, et al. Assessment of imatinib as first-line treatment of chronic myeloid leukemia: 10-year survival results of the randomized CML study IV and impact of non-CML determinants. Leukemia. 2017;31:2398–406.
Hochhaus A, Saglio G, Hughes TP, Larson RA, Kim DW, Issaragrisil S, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30:1044–54.
Cortes JE, Saglio G, Kantarjian HM, Baccarani M, Mayer J, Boque C, et al. Final 5-year study results of DASISION: the dasatinib versus imatinib study in treatment-naive chronic myeloid leukemia patients trial. J Clin Oncol. 2016;34:2333–40.
Baccarani M, Deininger MW, Rosti G, Hochhaus A, Soverini S, Apperley JF, et al. EuropeanLeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood. 2013;122:872–84.
Wood WA, Lee SJ. Malignant hematologic diseases in adolescents and young adults. Blood. 2011;117:5803–15.
Pemmaraju N, Cortes J. Chronic myeloid leukemia in adolescents and young adults: patient characteristics, outcomes and review of the literature. Acta Haematol. 2014;132:298–306.
Kalmanti L, Saussele S, Lauseker M, Proetel U, Müller MC, Hanfstein B, et al. Younger patients with chronic myeloid leukemia do well in spite of poor prognostic indicators: results from the randomized CML study IV. Ann Hematol. 2014;93:71–80.
Staley EM, Simmons SC, Feldman AZ, Lorenz RG, Marques MB, Williams LA 3rd, et al. Management of chronic myeloid leukemia in the setting of pregnancy: when is leukocytapheresis appropriate? A case report and review of the literature. Transfusion. 2018;58:456–60.
Gangat N, Tefferi A. Myeloproliferative neoplasms and pregnancy: Overview and practice recommendations. Am J Hematol. 2021;96:354–66.
Talpaz M, Mercer J, Hehlmann R. The interferon-alpha revival in CML. Ann Hematol. 2015;94:S195–207.
Mubarak AA, Kakil IR, Awidi A, Al-Homsi U, Fawzi Z, Kelta M, et al. Normal outcome of pregnancy in chronic myeloid leukemia treated with interferon-alpha in 1st trimester: report of 3 cases and review of the literature. Am J Hematol. 2002;69:115–8.
Kantarjian HM, Deisseroth A, Kurzrock R, Estrov Z, Talpaz M. Chronic myelogenous leukemia: a concise update. Blood. 1993;82:691–703.
Quinta´s-Cardama A, Kantarjian HM, Giles F, Verstovsek S. Pegylated interferon therapy for patients with Philadelphia chromosome-negative myeloproliferative disorders. Thromb Hemost. 2006;32:409–16.
Varytė G, Arlauskienė A, Ramašauskaitė D. Pregnancy and multiple sclerosis: an update. Curr Opin Obstet Gynecol. 2021;33:378–83.
Acknowledgements
The authors express their gratitude to the colleagues who treated and consulted patients within the multidisciplinary team. Our warm gratitude to the patients and their children. On behalf of ELN Pregnancy Registry Committee we express our special gratitude for the contribution to collecting cases for the ELN Pregnancy Registry to Dr. Dong-Wook Kim from Catholic Hematology Hospital, Seoul St. Mary’s Hospital, Seoul, S. Korea, Dr. Konstantin Kotlyarchuk from SI ‘Institute of Blood Pathology and Transfusion Medicine UAMS, Lviv, Ukraine and Dr. Penka Ganeva from Specilaized Hospital for Active Treatment of Hematological Diseases, Sofia, Bulgaria. We are grateful to Sandro Pittori for management of the REDCap database in the international registry. We thank Mr. Christophe Bouvier, datamanager, for data collection in Lyon, France. We thank Yulia Chabaeva and Sergey Kulikov from National Medical Hematology Research Center, Moscow, for the data management and express our special gratitude to «National Hematological Society» for promoting the development of hematology, transfusiology and bone marrow transplantation in Russian Federation. JFA acknowledges the support of the Imperial College NIHR Biomedical Research Centre Our special thanks to Professor Rudiger Hehlmann for many years of inspiring support for ELN international cooperation.
Funding
The study was supported by European LeukemiaNet.
Author information
Authors and Affiliations
Contributions
EC and EA were responsible for designing the study protocol, collecting and extracting the data and interpreting results. EC, JA, AT and EA were responsible for preparing the initial report. All the co-authors contributed into data collection and were involved into manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
EC – speaker: Novartis, Pfizer, R-Pharm, consultancy: Ascentage Pharma, JA – speaker: Novartis, Bristol Myers Squibb, Incyte, honoraria: Novartis, Bristol Myers Squibb, Incyte, Research Funding: Pfizer, Incyte, AT – speaker: Novartis, Pfizer, R-Pharm, Amgen, consultancy/advisory board: Novartis, DR – Novartis, Incyte, Pfizer, Terns, FEN – consultancy: Sun Pharma Ltd, Novartis, speaker: Incyte Biosciences, board entity: Incyte Biosciences, Pfizer, Novartis, BMS-Celgene, research fundings: Incyte Biosciences, Novartis, ASA – consultancy: Novartis, JB – honoraria: Novartis, Incyte, Pfizer, MC – advisory board: Insight, Novartis, Italfarmaco, speaker: Servier, Abbvie, Janssen, PF – consultancy: Jazz, Jannsen, Abbvie, MMT – consultancy: Novartis, Takeda, EA – consultancy: BMS, Novaris, Incyte, Pfizer, advisory: BMS, Novaris, Incyte, Pfizer, SAK, MAY, DB, KhK, SS, HFR, RS and EP – nothing to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chelysheva, E., Apperley, J., Turkina, A. et al. Chronic myeloid leukemia diagnosed in pregnancy: management and outcome of 87 patients reported to the European LeukemiaNet international registry. Leukemia 38, 788–795 (2024). https://doi.org/10.1038/s41375-024-02183-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-024-02183-0