Abstract
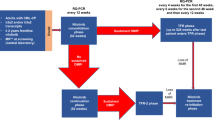
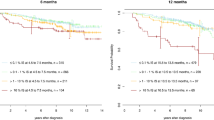
Following the achievement of deep molecular response on tyrosine kinase inhibitors (TKIs), approximately half of patients with chronic myeloid leukemia (CML) can discontinue TKI and remain in treatment-free remission (TFR). The ALLG CML8 study enrolled 40 imatinib-treated patients with undetectable BCR-ABL1 mRNA (approximately MR4.5). Molecular relapse was defined as detectable BCR-ABL1 on two consecutive tests or any single value >0.1%. With a median follow-up of 8.6 years (range 5.7–11.2 years), 18 patients remain in continuous TFR (45.0%; 95% confidence interval 31.9−63.4%). The latest relapse detected was 27 months after stopping imatinib. No patient progressed to advanced phase. Twenty-two patients met criteria for imatinib re-treatment and all regained undetectable molecular response. Nine patients in long-term TFR were monitored by highly sensitive individualized BCR-ABL1 DNA PCR in a sufficient number of samples to enable more precise quantification of residual leukemia. BCR-ABL1 DNA decreased from a median of MR5.0 in the first year of TFR to MR6.1 in the sixth year of TFR. Our results support the long-term safety and remarkable stability of response after imatinib discontinuation in appropriately selected CML patients. Serial high sensitivity testing provides a new and unexpected finding of gradually reducing CML cells in patients in long-term TFR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ross DM, Branford S, Seymour JF, Schwarer AP, Arthur C, Yeung DT, et al. Safety and efficacy of imatinib cessation for CML patients with stable undetectable minimal residual disease: results from the TWISTER study. Blood. 2013;122:515–22.
Mahon FX, Rea D, Guilhot J, Guilhot F, Huguet F, Nicolini F, et al. Discontinuation of imatinib in patients with chronic myeloid leukaemia who have maintained complete molecular remission for at least 2 years: the prospective, multicentre Stop Imatinib (STIM) trial. Lancet Oncol. 2010;11:1029–35.
Rousselot P, Charbonnier A, Cony-Makhoul P, Agape P, Nicolini FE, Varet B, et al. Loss of major molecular response as a trigger for restarting tyrosine kinase inhibitor therapy in patients with chronic-phase chronic myelogenous leukemia who have stopped imatinib after durable undetectable disease. J Clin Oncol. 2014;32:424–30.
Hughes TP, Ross DM. Moving treatment-free remission into mainstream clinical practice in CML. Blood. 2016;128:17–23.
Saussele S, Richter J, Hochhaus A, Mahon FX. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638–47.
Villemagne Sanchez LA, O’Callaghan C, Gough K, Hall K, Kashima Y, Seymour JF, et al. Patient perceptions of treatment-free remission in chronic myeloid leukemia. Leuk Lymphoma. 2018;59:406–15.
Hochhaus A, Saussele S, Rosti G, Mahon FX, Janssen J, Hjorth-Hansen H, et al. Chronic myeloid leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(suppl_4):iv41–iv51.
Etienne G, Guilhot J, Rea D, Rigal-Huguet F, Nicolini F, Charbonnier A, et al. Long-term follow-up of the French Stop Imatinib (STIM1) study in patients with chronic myeloid leukemia. J Clin Oncol. 2017;35:298–305.
Ross DM, Branford S, Seymour JF, Schwarer AP, Arthur C, Bartley PA, et al. Patients with chronic myeloid leukemia who maintain a complete molecular response after stopping imatinib treatment have evidence of persistent leukemia by DNA PCR. Leukemia. 2010;24:1719–24.
Ross DM, O’Hely M, Bartley PA, Dang P, Score J, Goyne JM, et al. Distribution of genomic breakpoints in chronic myeloid leukemia: analysis of 308 patients. Leukemia. 2013;27:2105–7.
Branford S, Fletcher L, Cross NC, Muller MC, Hochhaus A, Kim DW, et al. Desirable performance characteristics for BCR-ABL measurement on an international reporting scale to allow consistent interpretation of individual patient response and comparison of response rates between clinical trials. Blood. 2008;112:3330–8.
Branford S, Hughes TP, Rudzki Z. Monitoring chronic myeloid leukaemia therapy by real-time quantitative PCR in blood is a reliable alternative to bone marrow cytogenetics. Br J Haematol. 1999;107:587–99.
Gabert J, Beillard E, van der Velden VH, Bi W, Grimwade D, Pallisgaard N, et al. Standardization and quality control studies of ‘real-time’ quantitative reverse transcriptase polymerase chain reaction of fusion gene transcripts for residual disease detection in leukemia—a Europe Against Cancer program. Leukemia. 2003;17:2318–57.
Pagani IS, Dang P, Kommers IO, Goyne JM, Nicola M, Saunders VA, et al. BCR-ABL1 genomic DNA PCR response kinetics during first-line imatinib treatment of chronic myeloid leukemia. Haematologica. 2018. 189787. https://doi.org/10.3324/haematol.2018.189787. [Epub ahead of print].
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem. 2009;55:611–22.
Sekhri A, Liu D, Rasul M, Ahmed N, Ahmed T, Seiter K. Very late relapse of chronic myelogenous leukemia after allogeneic bone marrow transplantation. Leuk Res. 2009;33:1291–3.
Goldman JM, Majhail NS, Klein JP, Wang Z, Sobocinski KA, Arora M, et al. Relapse and late mortality in 5-year survivors of myeloablative allogeneic hematopoietic cell transplantation for chronic myeloid leukemia in first chronic phase. J Clin Oncol. 2010;28:1888–95.
Cross NC, White HE, Colomer D, Ehrencrona H, Foroni L, Gottardi E, et al. Laboratory recommendations for scoring deep molecular responses following treatment for chronic myeloid leukemia. Leukemia. 2015;29:999–1003.
Rawer D, Borkhardt A, Wilda M, Kropf S, Kreuder J. Influence of stochastics on quantitative PCR in the detection of minimal residual disease. Leukemia. 2003;17:2527–8. author reply 2528−31
Hughes A, Clarson J, Tang C, Vidovic L, White DL, Hughes TP, et al. CML patients with deep molecular responses to TKI have restored immune effectors and decreased PD-1 and immune suppressors. Blood. 2017;129:1166–76.
Ohyashiki K, Katagiri S, Tauchi T, Ohyashiki JH, Maeda Y, Matsumura I, et al. Increased natural killer cells and decreased CD3(+)CD8(+)CD62L(+) T cells in CML patients who sustained complete molecular remission after discontinuation of imatinib. Br J Haematol. 2012;157:254–6.
Ilander M, Olsson-Stromberg U, Schlums H, Guilhot J, Bruck O, Lahteenmaki H, et al. Increased proportion of mature NK cells is associated with successful imatinib discontinuation in chronic myeloid leukemia. Leukemia. 2017;31:1108–16.
Imagawa J, Tanaka H, Okada M, Nakamae H, Hino M, Murai K, et al. Discontinuation of dasatinib in patients with chronic myeloid leukaemia who have maintained deep molecular response for longer than 1 year (DADI trial): a multicentre phase 2 trial. Lancet Haematol. 2015;2:e528–535.
Rea D, Henry G, Khaznadar Z, Etienne G, Guilhot F, Nicolini F, et al. Natural killer-cell counts are associated with molecular relapse-free survival after imatinib discontinuation in chronic myeloid leukemia: the IMMUNOSTIM study. Haematologica. 2017;102:1368–77.
Traulsen A, Lenaerts T, Pacheco JM, Dingli D. On the dynamics of neutral mutations in a mathematical model for a homogeneous stem cell population. J R Soc Interface. 2013;10:20120810.
Roeder I, Horn M, Glauche I, Hochhaus A, Mueller MC, Loeffler M. Dynamic modeling of imatinib-treated chronic myeloid leukemia: functional insights and clinical implications. Nat Med. 2006;12:1181–4.
Michor F, Hughes TP, Iwasa Y, Branford S, Shah NP, Sawyers CL, et al. Dynamics of chronic myeloid leukaemia. Nature. 2005;435:1267–70.
Tang M, Gonen M, Quintas-Cardama A, Cortes J, Kantarjian H, Field C, et al. Dynamics of chronic myeloid leukemia response to long-term targeted therapy reveal treatment effects on leukemic stem cells. Blood. 2011;118:1622–31.
Legros L, Nicolini FE, Etienne G, Rousselot P, Rea D, Giraudier S, et al. Second tyrosine kinase inhibitor discontinuation attempt in patients with chronic myeloid leukemia. Cancer. 2017;123:4403–10.
Saussele S, Richter J, Guilhot J, Gruber FX, Hjorth-Hansen H, Almeida A, et al. Discontinuation of tyrosine kinase inhibitor therapy in chronic myeloid leukaemia (EURO-SKI): a prespecified interim analysis of a prospective, multicentre, non-randomised, trial. Lancet Oncol. 2018;19:747–57.
Acknowledgements
The authors acknowledge the important contribution of the TWISTER study patients and coordinators, and the ALLG as the study sponsor. This research was funded by the Australian National Health & Medical Research Council (Project grant #1051965), the Royal Adelaide Hospital Health Services Charitable Gifts Board, and Novartis Pharmaceuticals.
Author information
Authors and Affiliations
Consortia
Contributions
DMR designed and supervised the study, performed experiments, analysed data, and prepared the manuscript; ISP performed the experiments, analysed data, and prepared the manuscript; NS, JFS, AKM, RJF CKA, ASY, APG, and APS contributed essential clinical data and reviewed the manuscript; PD and VAS performed experiments and reviewed the manuscript; JB contributed RQ-PCR data and reviewed the manuscript; CHK and DTY analysed data, and reviewed the manuscript; DLW supervised research and reviewed the manuscript; SB contributed RQ-PCR data, supervised research, and reviewed the manuscript; TPH designed the study, supervised research, and reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
DMR has received research funding from Novartis and Celgene, and honoraria from Novartis and BMS. AKM received honoraria from Novartis, BMS, and Specialised Therapeutics. ASY has received research funding from Novartis, BMS and Celgene, and honoraria from Novartis and BMS. DTY and TPH have received research funding and honoraria from Novartis, BMS, and Ariad. APG is an advisory board member for Novartis, BMS, Roche, Takeda, and MSD. APS has received honoraria from Novartis, BMS, Celgene, Roche, Amgen, and Specialised Therapeutics. SB has received research funding from Novartis, Ariad and Otsuka, honoraria from Novartis, BMS, Otsuka, Qiagen and Ariad, and is an advisory board member for Novartis and Qiagen. The remaining authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ross, D.M., Pagani, I.S., Shanmuganathan, N. et al. Long-term treatment-free remission of chronic myeloid leukemia with falling levels of residual leukemic cells. Leukemia 32, 2572–2579 (2018). https://doi.org/10.1038/s41375-018-0264-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-018-0264-0
This article is cited by
-
Computational modeling reveals key factors driving treatment-free remission in chronic myeloid leukemia patients
npj Systems Biology and Applications (2024)
-
Tyrosine Kinase Inhibitor Discontinuation in Chronic Myeloid Leukemia: Strategies to Optimize Success and New Directions
Current Hematologic Malignancy Reports (2024)
-
Single-cell analysis of immune recognition in chronic myeloid leukemia patients following tyrosine kinase inhibitor discontinuation
Leukemia (2024)
-
Cardiovascular Adverse Events of Tyrosine Kinase Inhibitors in Chronic Myeloid Leukemia: Clinical Relevance, Impact on Outcome, Preventive Measures and Treatment Strategies
Current Treatment Options in Oncology (2023)
-
Guidelines for the treatment of chronic myeloid leukemia from the NCCN and ELN: differences and similarities
International Journal of Hematology (2023)