Abstract
Background/objective
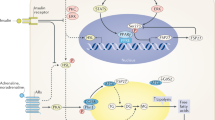
The recognized association between male hypogonadism and obesity has multifactorial implications on adipose tissue (AT) physiology. The fat solubility of testosterone (T) suggests a sequestration process in fat depots, leading to reduced circulating levels of T in obesity. Several evidence suggest that steroids play a two-sided inhibitory role on adipogenesis by locally decreasing lipid accumulation and by stimulating lipolysis. The current study investigates T trafficking and activity in dysfunctional AT.
Subjects/methods
Samples of subcutaneous AT (SAT) were obtained from explants from lipoaspirate plastic surgery in six obese and six normal weight male patients. Experimental procedures on both SAT explants and insulin-resistant (IR) 3T3-L1 adipocytes were performed, including real-time PCR and mass-spectrometry quantification.
Results
A significant deregulation of gene responsiveness to androgens in IR cells and obese SAT was observed (all p < 0.05), together with reduced T release after adrenergic stimulation (−10% compared with −55% in lean SAT, p = 0.021). Higher concentrations of intracellular T and estradiol in obese SAT were also observed (2.4 vs. 1.3 ng/g, p = 0.013 and 0.075 vs. 0.22 ng/g, p = 0.004, respectively). Testosterone accumulation resulted in even lower expression in androgen-responsive genes involved in lipolytic and anti-adipogenic pathways from both in vitro and ex vivo experiments.
Conclusions
These results suggest an altered response of dysfunctional fat cells to testosterone stimulation, which normally favors lipolysis and induces an anti-adipogenic effect. The considerable reduction of lipolytic T release after adrenergic stimulation in obese SAT contributes to AT dysfunction, in a feedforward loop further reducing T levels in obese hypogonadal males.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zitzmann M. Testosterone deficiency, insulin resistance and the metabolic syndrome. Nat Rev Endocrinol. 2009;5:673–81.
Mauvais-Jarvis F. Estrogen and androgen receptors: regulators of fuel homeostasis and emerging targets for diabetes and obesity. Trends Endocrinol Metab. 2011;22:24–33.
Basaria S, Lieb J, Tang AM, DeWeese T, Carducci M, Eisenberger M, et al. Long-term effects of androgen deprivation therapy in prostate cancer patients. Clin Endocrinol. 2002;56:779–86.
Zitzmann M, Faber S, Nieschlag E. Association of specific symptoms and metabolic risks with serum testosterone in older men. J Clin Endocrinol Metab. 2006;91:4335–43.
Mårin P. Testosterone and regional fat distribution. Obes Res. 1995;3 Suppl 4:609S–12S.
Wu FCW, Tajar A, Pye SR, Silman AJ, Finn JD, O’Neill TW, et al. Hypothalamic-pituitary-testicular axis disruptions in older men are differentially linked to age and modifiable risk factors: the European male aging study. J Clin Endocrinol Metab. 2008;93:2737–45.
Rastrelli G, Carter EL, Ahern T, Finn JD, Antonio L, O’Neill TW, et al. Development of and recovery from secondary hypogonadism in aging men: prospective results from the EMAS. J Clin Endocrinol Metab. 2015;100:3172–82.
Hammoud A, Gibson M, Hunt SC, Adams TD, Carrell DT, Kolotkin RL, et al. Effect of Roux-en-Y gastric bypass surgery on the sex steroids and quality of life in obese men. J Clin Endocrinol Metab. 2009;94:1329–32.
O’Reilly MW, House PJ, Tomlinson JW. Understanding androgen action in adipose tissue. J Steroid Biochem Mol Biol. 2014;143:277–84.
Blouin K, Boivin A, Tchernof A. Androgens and body fat distribution. J Steroid Biochem Mol Biol. 2008;108:272–80.
Zhang Y, Calvo E, Martel C, Luu-The V, Labrie F, Tchernof A. Response of the adipose tissue transcriptome to dihydrotestosterone in mice. Physiol Genomics. 2008;35:254–61.
Lafontan M, Sengenes C, Galitzky J, Berlan M, De Glisezinski I, Crampes F, et al. Recent developments on lipolysis regulation in humans and discovery of a new lipolytic pathway. Int J Obes Relat Metab Disord. 2000;24 Suppl 4:S47–52.
Ryden M, Backdahl J, Petrus P, Thorell A, Gao H, Coue M, et al. Impaired atrial natriuretic peptide-mediated lipolysis in obesity. Int J Obes. 2016;40:714–20.
Reynisdottir S, Wahrenberg H, Carlström K, Rössner S, Arner P. Catecholamine resistance in fat cells of women with upper-body obesity due to decreased expression of beta 2-adrenoceptors. Diabetologia. 1994;37:428–35.
Di Nisio A, De Toni L, Sabovic I, Rocca MS, De Filippis V, Opocher G, et al. Impaired release of vitamin D in dysfunctional adipose tissue: new cues on vitamin D supplementation in obesity. J Clin Endocrinol Metab. 2017;102:2564–74.
Langin D, Dicker A, Hoffstedt J, Mairal A, Arner E, Sicard A, et al. Obesity defects of adipose lipase. Diabetes. 2005;54:3190–97.
Bélanger C, Luu-The V, Dupont P, Tchernof A. Adipose tissue intracrinology: potential importance of local androgen/estrogen metabolism in the regulation of adiposity. Horm Metab Res. 2002;34:737–45.
Bélanger C, Hould F-S, Lebel S, Biron S, Brochu G, Tchernof A. Omental and subcutaneous adipose tissue steroid levels in obese men. Steroids. 2006;71:674–82.
O’Reilly MW, Kempegowda P, Walsh M, Taylor AE, Manolopoulos KN, Allwood JW, et al. AKR1C3-mediated adipose androgen generation drives lipotoxicity in women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2017;102:3327–39.
Drincic AT, Armas LAG, Van Diest EE, Heaney RP. Volumetric dilution, rather than sequestration best explains the low vitamin D Status of obesity. Obesity. 2012;20:1444–8.
George AM, Jones PM. Perspectives on the structure–function of ABC transporters: the switch and constant contact models. Prog Biophys Mol Biol. 2012;109:95–107.
Tarling EJ, de Aguiar Vallim TQ, Edwards PA. Role of ABC transporters in lipid transport and human disease. Trends Endocrinol Metab. 2013;24:342–50.
Sivils JC, Gonzalez I, Bain LJ. Mice lacking Mrp1 have reduced testicular steroid hormone levels and alterations in steroid biosynthetic enzymes. Gen Comp Endocrinol. 2010;167:51–9.
Dankers ACA, Roelofs MJE, Piersma AH, Sweep FCGJ, Russel FGM, van den Berg M, et al. Endocrine disruptors differentially target ATP-binding cassette transporters in the blood-testis barrier and affect Leydig cell testosterone secretion in vitro. Toxicol Sci. 2013;136:382–91.
Deslypere JP, Verdonck L, Vermeulen A. Fat tissue: a steroid reservoir and site of steroid metabolism. J Clin Endocrinol Metab. 1985;61:564–70.
Joyner J, Hutley L, Cameron D. Intrinsic regional differences in androgen receptors and dihydrotestosterone metabolism in human preadipocytes. Horm Metab Res. 2002;34:223–8.
Blouin K, Richard C, Brochu G, Hould FS, Lebel S, Marceau S, et al. Androgen inactivation and steroid-converting enzyme expression in abdominal adipose tissue in men. J Endocrinol. 2006;191:637–49.
Zhang J, Hupfeld CJ, Taylor SS, Olefsky JM, Tsien RY. Insulin disrupts beta-adrenergic signalling to protein kinase A in adipocytes. Nature. 2005;437:569–73.
Arner P, Langin D. Lipolysis in lipid turnover, cancer cachexia, and obesity-induced insulin resistance. Trends Endocrinol Metab. 2014;25:255–62.
Blouin K, Nadeau M, Perreault M, Veilleux A, Drolet R, Marceau P, et al. Effects of androgens on adipocyte differentiation and adipose tissue explant metabolism in men and women. Clin Endocrinol. 2010;72:176–88.
Singh R, Artaza JN, Taylor WE, Gonzalez-Cadavid NF, Bhasin S. Androgens stimulate myogenic differentiation and inhibit adipogenesis in C3H 10T1/2 pluripotent cells through an androgen receptor-mediated pathway. Endocrinology. 2003;144:5081–8.
Hoffstedt J, Arner P, Schalling M, Pedersen NL, Sengul S, Ahlberg S, et al. A common hormone-sensitive lipase i6 gene polymorphism is associated with decreased human adipocyte lipolytic function. Diabetes. 2001;50:2410–3.
Holland AM, Roberts MD, Mumford PW, Mobley CB, Kephart WC, Conover CF, et al. Testosterone inhibits expression of lipogenic genes in visceral fat by an estrogen-dependent mechanism. J Appl Physiol. 2016;121:792–805.
Anderson LA, McTernan PG, Harte AL, Barnett AH, Kumar S. The regulation of HSL and LPL expression by DHT and flutamide in human subcutaneous adipose tissue. Diabetes Obes Metab. 2002;4:209–13.
Dieudonné M-N, Sammari A, Dos Santos E, Leneveu M-C, Giudicelli Y, Pecquery R. Sex steroids and leptin regulate 11β-hydroxysteroid dehydrogenase I and P450 aromatase expressions in human preadipocytes: sex specificities. J Steroid Biochem Mol Biol. 2006;99:189–96.
Morris PG, Hudis CA, Giri D, Morrow M, Falcone DJ, Zhou XK, et al. Inflammation and increased aromatase expression occur in the breast tissue of obese women with breast cancer. Cancer Prev Res. 2011;4:1021–9.
Subbaramaiah K, Morris PG, Zhou XK, Morrow M, Du B, Giri D, et al. Increased Levels of COX-2 and prostaglandin E 2 contribute to elevated aromatase expression in inflamed breast tissue of obese women. Cancer Discov. 2012;2:356–65.
Polari L, Yatkin E, Martínez Chacón MG, Ahotupa M, Smeds A, Strauss L, et al. Weight gain and inflammation regulate aromatase expression in male adipose tissue, as evidenced by reporter gene activity. Mol Cell Endocrinol. 2015;412:123–30.
Calle EE, Kaaks R. Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat Rev Cancer. 2004;4:579–91.
Finkelstein JS, Lee H, Burnett-Bowie SAM, Pallais JC, Yu EW, Borges LF, et al. Gonadal steroids and body composition, strength, and sexual function in men. N Engl J Med. 2013;369:1011–22.
Colleluori G, Aguirre LE, Qualls C, Chen R, Napoli N, Villareal DT, et al. Adipocytes ESR1 expression, body fat and response to testosterone therapy in hypogonadal men vary according to estradiol levels. Nutrients. 2018;10:1226.
Aversa A, Caprio M, Antelmi A, Armani A, Brama M, Greco EA, et al. Exposure to phosphodiesterase type 5 inhibitors stimulates aromatase expression in human adipocytes in vitro. J Sex Med. 2011;8:696–704.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Di Nisio, A., Sabovic, I., De Toni, L. et al. Testosterone is sequestered in dysfunctional adipose tissue, modifying androgen-responsive genes. Int J Obes 44, 1617–1625 (2020). https://doi.org/10.1038/s41366-020-0568-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-020-0568-9
This article is cited by
-
Obesity, type 2 diabetes, and testosterone in ageing men
Reviews in Endocrine and Metabolic Disorders (2022)
-
Relationship of Vitamin D status with testosterone levels: a systematic review and meta-analysis
Endocrine (2021)
-
Testosterone treatment is associated with reduced adipose tissue dysfunction and nonalcoholic fatty liver disease in obese hypogonadal men
Journal of Endocrinological Investigation (2021)