Key Points
-
In 2000, the view of Candida albicans as a purely asexual organism was overturned when C. albicans was shown to mate, both in a mouse host and on laboratory media. C. albicans has a highly efficient mating apparatus, which seems to have remained hidden from investigators.
-
In Saccharomyces cerevisiae, the mating locus called the MAT locus — encodes transcriptional regulatory proteins that are responsible for controlling the sexual cycle. The genes residing at the MAT locus can be either the a type or the α type. In C. albicans the mating locus — called the MTL (mating-type-like) locus — is much larger than the MAT locus of S. cerevisiae and contains more genes. However, the MTL locus does have orthologues of the S. cerevisiae MATa1, MATα2 and MATα1 genes, and these control mating in C. albicans. Similar to S. cerevisiae the C. albicans locus can be can be either the a type or the α type.
-
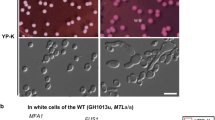
In C. albicans, a cells mate with α cells, but to do so, they must first undergo a switch from the white from to the opaque from. This transition involves changes in expression of several hundred genes. White-opaque awitching is controlled by the mating-type locus, and a and α, but not a/α cells, can undergo switching.White and opaque cells have different virulence profiles in an animal model, and this link between mating and virulence indicates that white-opaque switching (which is conspicuously absent in S. cerevisiae mating) might be a strategy to allow mating in the hostile environment of a mammalian host.
-
C. albicans is diploid, and a successful mating event produces an a/α (non-mating) tetraploid cell. These tetraploids can lose chromosomes in an apparently concerted manner, to become diploid (or at least near-diploid) a and α mating-competent cells. So, in C. albicans, chromosome loss might provide an alternative to meiosis thereby completing a parasexual cycle.
-
Important, but largely unanswered questions, are discussed, namely why is white-opaque switching a part of mating? What other aspects of C. albicans biology might the MTL locus control? What might be the purpose of mating in this human fungal pathogen?
Abstract
Candida albicans has maintained an elaborate — but largely hidden — mating apparatus, which shares some features with the closely related 'model' yeast Saccharomyces cerevisiae, but which also has some important differences. The differences are particularly noteworthy, as they could indicate the strategies that allow C. albicans to survive and mate in the hostile environment of a mammalian host. Indeed, some features of C. albicans mating seem to be intimately connected to its host.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Odds, F. C. in Candida and Candidosis. (Bailliere Tindall, London, UK, 1988). Beautifully written account of Candida research up to the late 1980s.
Calderone, R. A. in Candida and Candidiasis. (ed. Calderone, R. A.) 3–13 (ASM Press, Washington DC, 2002).
Tibayrenc, M. Are Candida albicans natural populations subdivided? Trends Microbiol. 5, 253–254 (1997).
Vilgalys, R., Graser, Y., Schonian, G. & Presber, W. Is Candida albicans clonal or recombining? Trends Microbiol. 5, 254–257 (1997). References 3 and 4 summarize the conclusions regarding sexual recombination reached from population studies of C. albicans.
Xu, J. & Mitchell, T. G. in Candida and Candidiasis. (ed. Calderone, R. A.) 55–64 (ASM Press, Washington DC, 2002).
Whelan, W. L. The genetics of medically important fungi. CRC Critical Reviews in Microbiology 14, 99–170 (1987).
Ayala, F.J. Is sex better? Parasites say 'no'. Proc. Natl Acad. Sci USA 95, 3346–3348 (1998).
Hull, C. M., Raisner, R. M. & Johnson, A. D. Evidence for mating of the 'asexual' yeast Candida albicans in a mammalian host. Science 289, 307–310 (2000).
Magee, B. B. & Magee, P. T. Induction of mating in Candida albicans by construction of MTLa and MTLα strains. Science 289, 310–313 (2000). References 8 and 9 are the first reports of mating in C. albicans.
Gargas, A., DePriest, P. T., Grube, M. & Tehler, A. Multiple origins of lichen symbioses in fungi suggested by SSU rDNA phylogeny. Science 268: 1492–1495 (1995).
Hedges, S. B. The origin and evolution of model organisms. Nature Rev. Genet. 3, 838–849 (2002).
Goffeau, A. et al. Life with 6000 genes. Science 274, 546–567 (1996).
Kellis, M., Patterson, N., Endrizzi, M., Birren, B. & Lander, E. S. Sequencing and comparison of yeast species to identify genes and regulatory elements. Nature 423, 241–254 (2003).
Magee, P. T. & Chibana, H. in Candida and Candidiasis. (ed. Calderone, R. A.) 293–304 (ASM Press, Washington DC, 2002).
Kadosh, D. and Johnson, A. D. Rfg1, a protein related to the Saccharomyces cerevisiae hypoxic regulator Rox1, controls filamentous growth and virulence in Candida albicans. Mol. Cell Biol. 21, 2496–2505 (2001).
Khalaf, R. A. & Zitomer, R. S. The DNA-binding protein Rfg1 is a repressor of filamentation in Candida albicans. Genetics 157, 1503–1512 (2001).
Olaiya, A. F. & Sogin, S. J. Ploidy determination of Candida albicans. J. Bacteriol. 140, 1043–1049 (1979).
Whelen, W. L. & Magee, P. T. Natural heterozygosity in Candida albicans. Mol Gen Genet. 180, 107–113 (1981).
Riggsby, W. S., Torres-Bauza, L. J., Wills, J. W. & Townes, T. M. DNA content, kinetic complexity, and the ploidy question in Candida albicans. Mol. Cell Biol. 2, 853–862 (1982).
Berman, J. & Sudbery, P. E. Candida albicans: A molecular revolution built on lessons from budding yeast. Nature 3, 918–930 (2002). Excellent review of recent developments in C. albicans research.
Scherer, S. in Candida and Candidiasis. (ed. Calderone, R. A.) 259–265 (ASM Press, Washington DC, 2002).
Yesland, K. & Fonzi, W. A. Allele-specific gene targeting in Candida albicans results from heterology between alleles. Microbiology 146, 2097–2104 (2000).
Herskowitz, I., Rine, J. & Strathern, J. in The Molecular Cellular Biology of the Yeast Saccharomyces. (eds Jones, E. W., Pringle, J. R. & Broach, J. R.) Vol. 2 (Cold Spring Harbor Laboratory Press, New York, 1992).
Johnson, A. D. Molecular mechanisms of cell-type determination in budding yeast. Curr. Opin. Genet. Dev. 5, 552–558 (1995).
Sadhu, C., Hoekstra, D., McEachern, M. J., Reed, S. I. & Hicks, J. B. A G-protein α-subunit from asexual Candida albicans functions in the mating signal transduction pathway of Saccharomyces cerevisiae and is regulated by the a1-α2 repressor. Mol. Cell Biol. 12, 1977–1985 (1992).
Liu, H., Kohler, J. & Fink, G. R. Suppression of hyphal formation in Candida albicans by mutation of a STE12 homolog. Science 266, 1723–1726 (1994).
Clark, K. L. et al. Constitutive activation of the Saccharomyces cerevisiae mating response pathway by a MAP kinase kinase from Candida albicans. Mol. Gen. Genet. 249, 609–621 (1995).
Diener, A. C. & Fink, G. R. DLH1 is a functional Candida albicans homologue of the meiosis-specific gene DMC1. Genetics 143, 769–776 (1996).
Leberer, E. et al. Signal transduction through homologs of the Ste20p and Ste7p protein kinase hyphal formation in the pathogenic fungus Candida albicans. Proc. Natl Acad. Sci. USA 93, 13217–13222 (1996).
Raymond, M. et al. A Ste6p/P-glycoprotein homologue from the asexual yeast Candida albicans transports the a-factor mating pheromone in Saccharomyces cerevisiae. Mol. Microbiol. 27, 587–598 (1998).
Hull, C. M. & Johnson, A. D. Identification of a mating type-like locus in the asexual pathogenic yeast Candida albicans. Science 285, 1271–1275 (1999). Description of the C. albicans mating-type-like ( MTL ) locus.
Odds, F. C., Brown, A. J. & Gow, N. A. Might Candida albicans be made to mate after all? Trends Microbiol. 8, 4–6 (2000).
Fonzi, W. A. & Irwin, M. Y. Isogenic strain construction and gene mapping in Candida albicans. Genetics 134, 717–728 (1993).
Janbon, G., Sherman, F. & Rustchenko, E. Monosomy of a specific chromosome determines L-sorbose utilization: a novel regulatory mechanism in Candida albicans. Proc. Natl Acad. Sci. USA 95, 5150–5155 (1998).
Janbon, G., Sherman, F. & Rustchenko, E. Appearance and properties of L-sorbose-utilizing mutants of Candida albicans obtained on a selective plate. Genetics 153, 653–664 (1999).
Gow, N. A., Brown, A. J. & Odds, F. C. Candida's arranged marriage. Science 289, 256–257 (2000).
Soll, D. R. in Candida and Candidiasis. (ed. Calderone, R. A.) 123–142 (ASM Press, Washington DC, 2002).
Soll, D. R., Lockhart, S. & Zhao, R. The relationship between switching and mating in Candida albicans. Eukaryot. Cell 2, 390–397 (2003).
Srikantha, T., Tsai, L., Daniels, K., Klar, A. J. & Soll, D. R. The histone deacetylase genes HDA1 and RPD3 play distinct roles in regulation of high-frequency phenotypic switching in Candida albicans. J. Bacteriol. 183, 4614–4625 (2001).
Lan, C. Y. et al. Metabolic specialization associated with phenotypic switching in Candida albicans. Proc. Natl Acad. Sci. USA 99, 14907–14912 (2002). Description of several hundred genes differentially expressed in white versus opaque cells.
Slutsky, B. et al. 'White-opaque transition': a second high-frequency switching system in Candida albicans. J. Bacteriol. 169, 189–197 (1987). Original description of white-opaque switching.
Miller, M. G. & Johnson, A. D. White-opaque switching in Candida albicans is controlled by mating-type locus homeodomain proteins and allows efficient mating. Cell 110, 293–302 (2002). First description of the role of white-opaque switching in mating.
Lockhart, S. R. et al. In Candida albicans, white-opaque switchers are homozygous for mating type. Genetics 162, 737–745 (2002).
Gow, N. A. Candida albicans switches mates. Mol. Cell 10, 217–218 (2002).
Sprague, G. F. Jr. in Guide to Yeast Genetics and Molecular Biology. (eds Guthrie, C. & Fink, G. R.) 77–93 (Academic Press, New York, 1991).
Anderson, J., Mihalik, R. & Soll, D. R. Ultrastructure and antigenicity of the unique cell wall pimple of the Candida opaque phenotype. J. Bacteriol. 172, 224–235 (1990).
Lockhart, S. R., Daniels, K. J., Zhao, R., Wessels, D. & Soll, D. R. Cell biology of mating in Candida albicans. Eukaryot. Cell. 2, 49–61 (2003). First visualization and description of the mating process in C. albicans.
Sprague, G. F. J. & Thorner, J. W. in The Molecular and Cellular Biology of the Yeast Saccharomyces (eds Jones, E. W., Pringle, J. R. & Broach, J. R) 657–744 (Cold Spring Harbor Laboratory Press, New York, 1992).
MacKay, V. L. in The Early Days of Yeast Genetics. (eds Hall, M. N. & Linder, P.) (Cold Spring Harbor Laboratory Press, New York, 1993).
Dutcher, S. K. Internuclear transfer of genetic information in kar1-1/KAR1 heterokaryons in Saccharomyces cerevisiae. Mol. Cell. Biol. 1, 245–253 (1981).
Chen, J., Chen, J., Lane, S. & Liu, H. A conserved mitogen-activated protein kinase pathway is required for mating in Candida albicans. Mol. Microbiol. 46: 1335–1344 (2002).
Magee, B. B., Legrand, M., Alarco, A. M., Raymond, M. & Magee, P. T. Many of the genes required for mating in Saccharomyces cerevisiae are also required for mating in Candida albicans. Mol. Microbiol. 46, 1345–1351 (2002).
Bennett, R. J. & Johnson, A. D. Completion of a parasexual cycle in Candida albicans by induced chromosome loss in tetraploid strains. EMBO J. 22, 2505–2515 (2003).
Tzung, K. W. et al. Genomic evidence for a complete sexual cycle in Candida albicans. Proc. Natl Acad. Sci. USA 98, 3249–3253 (2001).
Bennett, R. J., Uhl, M. A., Miller, M. G. & Johnson, A. D. Identification and characterization of a Candida albicans mating pheromone. Mol. Cell. Biol. (in the press).
Madhani, H. D. & G. R., Fink. The control of filamentous differentiation and virulence in fungi. Trends Cell Biol. 8, 348–353 (1998). Excellent general review on the relationship of filamentous growth to virulence in fungi.
Mitchell, A. P. Dimorphism and virulence in Candida albicans. Curr. Opin. Microbiol. 1, 687–692 (1998).
Whiteway, M. Transcriptional control of cell type and morphogenesis in Candida albicans. Curr. Opin. Microbiol. 3, 582–588 (2000).
Calderone, R. A. & Fonzi, W. A. Virulence factors of Candida albicans. Trends Microbiol. 9, 327–335 (2001).
Navarro-Garcia, F., M. Sanchez, Nombela, C. & Pla, J. Virulence genes in the pathogenic yeast Candida albicans. FEMS Microbiol. Rev. 25, 245–268 (2001).
Liu, H. Co-regulation of pathogenesis with dimorphism and phenotypic switching in Candida albicans, a commensal and a pathogen. Int. J. Med. Microbiol. 292, 299–311 (2002).
Brown, A. J. P. in Candida and Candidiasis. (ed. Calderone, R. A.) 87–93 (ASM Press, Washington, DC, 2002).
Brown, A. J. P. in Candida and Candidiasis. (ed. Calderone, R. A.) 95–106 (ASM Press, Washington, DC, 2002).
Roberts, C. J. et al. Signaling and circuitry of multiple MAPK pathways revealed by a matrix of global gene expression profiles. Science 287, 873–880 (2000).
Rustchenko, E. & Sherman, F. in Pathogenic Fungi in Humans and Animals. (ed. Howard, D.) (Marcel Dekker, New York, 2003).
Hilton, C., Markie, D., Corner, B., Rikkerink, E. & Poulter, R. Heat shock induces chromosome loss in the yeast Candida albicans. Mol. Gen. Genet. 200, 162–168 (1985).
Kolotila, M. P. & Diamond, R. D. Effects of neutrophils and in vitro oxidants on survival and phenotypic switching of Candida albicans WO-1. Infect. Immun. 58, 1174–1179 (1990).
Kvaal, C. et al. Misexpression of the opaque-phase-specific gene PEP1 (SAP1) in the white phase of Candida albicans confers increased virulence in a mouse model of cutaneous infection. Infect. Immun. 67, 6652–6662 (1999).
Lachke, S. A., Lockhart, S. R., Daniels, K. J. & Soll, D. R. Skin facilitates Candida albicans mating. Infect. Immun. 71, 4970–4976 (2003).
Hoyer, L. L. The ALS gene family of Candida albicans. Trends Microbiol. 9, 176–180 (2001).
Lehto, M. & Olkkonen, V. M. The OSBP-related proteins: a novel protein family involved in vesicle transport, cellular lipid metabolism, and cell signalling. Biochim. Biophys. Acta 1631, 1–11 (2003).
Hama, H., Scheiders, E. A., Thorner, J., Takemoto, J. Y. & DeWald, D. B. Direct involvement of phosphatidylinositol 4-phosphate in secretion in the yeast Saccharomyces cerevisiae. J. Biol. Chem. 274, 34294–34300 (1999).
Audhya, A., Foti, M. & Emr, S. D. Distinct roles for the yeast phosphatidylinositol 4-kinases, Stt4p and Pik1p, in secretion, cell growth, and organelle membrane dynamics. Mol. Biol. Cell 11, 2673–2689 (2000).
Lengeler, K. B. et al. Mating-type locus of Cryptococcus neoformans: a step in the evolution of sex chromosomes. Eukaryot. Cell 1, 704–718 (2002).
Peck, J. R. & Waxman, D. Mutation and sex in a competitive world. Nature 406, 399–404 (2000).
Graser, Y. et al. Molecular markers reveal that population structure of the human pathogen Candida albicans exhibits both clonality and recombination. Proc. Natl Acad. Sci. USA 93, 12473–12477 (1996).
Anderson, J. B. et al. Infrequent genetic exchange and recombination in the mitochondrial genome of Candida albicans. J. Bacteriol. 183, 865–872 (2001).
Acknowledgements
The author thanks M. Chiu, M. Miller, A. Tsong and R. Zordan for critical comments and for help in preparing the manuscript and figures. Sequence data for Candida albicans was obtained from the Stanford Genome Technology Center (see Online links). Sequencing of Candida albicans was accomplished with the support of the NIDR and the Burroughs Wellcome fund. The author gratefully acknowledges the valuable comments made by the anonymous reviewers.
Author information
Authors and Affiliations
Glossary
- HOMEODOMAIN
-
A 60 amino acid protein domain that folds into a compact three-helix structure and binds to specific sequences of DNA. Homeodomain proteins are found in virtually all eukaryotes, where they specify cell identity.
- HETEROKARYON
-
A cell that has two or more genetically distinct nuclei.
Rights and permissions
About this article
Cite this article
Johnson, A. The biology of mating in Candida albicans. Nat Rev Microbiol 1, 106–116 (2003). https://doi.org/10.1038/nrmicro752
Issue Date:
DOI: https://doi.org/10.1038/nrmicro752
This article is cited by
-
Is There a Relationship Between Mating and Pathogenesis in Two Human Fungal Pathogens, Candida albicans and Candida glabrata?
Current Clinical Microbiology Reports (2023)
-
Morphogenic plasticity: the pathogenic attribute of Candida albicans
Current Genetics (2023)
-
Mechanism of Candida pathogenesis: revisiting the vital drivers
European Journal of Clinical Microbiology & Infectious Diseases (2020)
-
Proteins that physically interact with the phosphatase Cdc14 in Candida albicans have diverse roles in the cell cycle
Scientific Reports (2019)
-
Diversity of A mating type in Lentinula edodes and mating type preference in the cultivated strains
Journal of Microbiology (2018)