Key Points
-
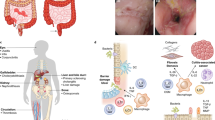
Through crosstalk with the intestinal microbiota, the innate immune system regulates intestinal homeostasis by conditioning the immune response both at mucosal sites and systemically, and by providing instructive signals that drive intestinal epithelial cell turnover and mucosal regeneration. The innate immune system is also a crucial modifier of bacterial handling through autophagy and a determinant of intestinal epithelial cell survival and apoptosis.
-
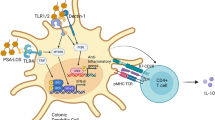
Although excessive Toll-like receptor (TLR) activation in the gut is pathogenic, sensing of the commensal flora by these receptors, particularly TLR2 and TLR4, is required for intestinal homeostasis and control of tissue repair. In addition, apical TLR9 stimulation triggers mucosal tolerance through induction of type I interferons that have been proposed to protect from colonic inflammation.
-
Nucleotide-binding oligomerization domain (NOD) receptors have a paramount role in intestinal homeostasis, and genetic mutations and single-nucleotide polymorphisms (SNPs) in their genes are associated with Crohn's disease. It is generally accepted that alterations in NOD2 function affect susceptibility to IBD because of the key role of this receptor in linking innate signals to the induction of adaptive immune tolerance to the intestinal microbiota. NOD2 modulates TLR signalling, regulates the production of defensins and IL-10, and polarizes the adaptive immune response towards a TH2-type response. More recently, NOD2 was shown to trigger autophagy in response to bacterial sensing through an interaction with the autophagy protein ATG16L1 (autophagy-related 16-like 1).
-
Genetic studies of Crohn's disease, including genome-wide association studies, uncovered two autophagy loci — ATG16L1 and IRGM — that are linked to Crohn's disease susceptibility. Deficiency in ATG16L1 function not only affects the ability of NOD2 to trigger autophagy, but also affects Paneth cells and renders macrophages hyperresponsive to TLR agonists, which leads to excessive inflammasome activation. This suggests that aberrant bacterial handling could act as a trigger for inflammation in Crohn's disease.
-
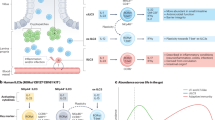
A SNP in the human NOD-, LRR- and pyrin domain-containing 3(NLRP3) region that downregulates NLRP3 expression is associated with Crohn's disease susceptibility. Consistently, the NLRP3 inflammasome–caspase 1–IL-18–IL-18R–MYD88 axis of inflammation is required for tissue repair following injury and is protective from colitis and colitis-associated colorectal cancer in mouse models.
-
Negative regulators of innate immunity pathways, including SIGIRR (single immunoglobulin IL-1-related receptor), SOCS1 (suppressor of cytokine signalling 1) and caspase 12, are important effectors of intestinal immune tolerance. Deletion of their respective genes leads to amplification of the intestinal inflammatory response and promotion of colorectal cancer.
Abstract
The innate immune system provides first-line defences in response to invading microorganisms and endogenous danger signals by triggering robust inflammatory and antimicrobial responses. However, innate immune sensing of commensal microorganisms in the intestinal tract does not lead to chronic intestinal inflammation in healthy individuals, reflecting the intricacy of the regulatory mechanisms that tame the inflammatory response in the gut. Recent findings suggest that innate immune responses to commensal microorganisms, although once considered to be harmful, are necessary for intestinal homeostasis and immune tolerance. This Review discusses recent findings that identify a crucial role for innate immune effector molecules in protection against colitis and colitis-associated colorectal cancer and the therapeutic implications that ensue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Economou, M. & Pappas, G. New global map of Crohn's disease: genetic, environmental, and socioeconomic correlations. Inflamm. Bowel Dis. 14, 709–720 (2008).
Shanahan, F. & Bernstein, C. N. The evolving epidemiology of inflammatory bowel disease. Curr. Opin. Gastroenterol. 25, 301–305 (2009).
Cohen, R. D. The pharmacoeconomics of biologic therapy for IBD. Nature Rev. Gastroenterol. Hepatol. 7, 103–109 (2010).
de Silva, S., Devlin, S. & Panaccione, R. Optimizing the safety of biologic therapy for IBD. Nature Rev. Gastroenterol. Hepatol. 7, 93–101 (2010).
Yan, F. & Polk, D. B. Probiotics: progress toward novel therapies for intestinal diseases. Curr. Opin. Gastroenterol. 26, 95–101 (2010).
Weinstock, J. V. & Elliott, D. E. Helminths and the IBD hygiene hypothesis. Inflamm. Bowel Dis. 15, 128–133 (2009).
Eaden, J. A., Abrams, K. R. & Mayberry, J. F. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut 48, 526–535 (2001).
von Roon, A. C. et al. The risk of cancer in patients with Crohn's disease. Dis. Colon Rectum 50, 839–855 (2007).
Rutter, M. D. et al. Thirty-year analysis of a colonoscopic surveillance program for neoplasia in ulcerative colitis. Gastroenterology 130, 1030–1038 (2006).
Feagins, L. A., Souza, R. F. & Spechler, S. J. Carcinogenesis in IBD: potential targets for the prevention of colorectal cancer. Nature Rev. Gastroenterol. Hepatol. 6, 297–305 (2009).
Kamangar, F., Dores, G. M. & Anderson, W. F. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J. Clin. Oncol. 24, 2137–2150 (2006).
Tomatis, L. (ed.) Cancer: Causes, Occurrence and Control. (IARC Press, 1990).
Mazmanian, S. K., Round, J. L. & Kasper, D. L. A microbial symbiosis factor prevents intestinal inflammatory disease. Nature 453, 620–625 (2008).
Rescigno, M. Before they were gut dendritic cells. Immunity 31, 454–456 (2009).
Hand, T. & Belkaid, Y. Microbial control of regulatory and effector T cell responses in the gut. Curr. Opin. Immunol. 22, 63–72 (2010).
Clarke, T. B. et al. Recognition of peptidoglycan from the microbiota by Nod1 enhances systemic innate immunity. Nature Med. 16, 228–231 (2010).
Hirayama, K. et al. Effects of human intestinal flora on mutagenicity of and DNA adduct formation from food and environmental mutagens. Carcinogenesis 21, 2105–2111 (2000).
Garrett, W. S. et al. Colitis-associated colorectal cancer driven by T-bet deficiency in dendritic cells. Cancer Cell 16, 208–219 (2009).
Garrett, W. S. et al. Communicable ulcerative colitis induced by T-bet deficiency in the innate immune system. Cell 131, 33–45 (2007). This paper demonstrates that alterations in innate and adaptive immune responses affect the composition of the commensal flora, resulting in the development of colitis.
Garrett, W. S. et al. Enterobacteriaceae act in concert with the gut microbiota to induce spontaneous and maternally transmitted colitis. Cell Host Microbe 8, 292–300 (2010). This paper demonstrates that the presence of specific bacterial species in immunodeficient mice induces colitis and colitis-associated cancer by a mechanism that also involves the normal gut microbiota.
Ivanov, I. I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009). This paper shows that the presence of SFB in the intestine is not by itself pathogenic but is responsible for a shift of the mucosal immune response towards a T H 17-type response.
Wu, H. J. et al. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 32, 815–827 (2010).
Westbrook, A. M., Wei, B., Braun, J. & Schiestl, R. H. More damaging than we think: systemic effects of intestinal inflammation. Cell Cycle 8, 2482–2483 (2009).
Backhed, F., Manchester, J. K., Semenkovich, C. F. & Gordon, J. I. Mechanisms underlying the resistance to diet-induced obesity in germ-free mice. Proc. Natl Acad. Sci. USA 104, 979–984 (2007).
Brown, S. L. et al. Myd88-dependent positioning of Ptgs2-expressing stromal cells maintains colonic epithelial proliferation during injury. J. Clin. Invest. 117, 258–269 (2007).
Walker, M. R., Patel, K. K. & Stappenbeck, T. S. The stem cell niche. J. Pathol. 217, 169–180 (2009).
Mankertz, J. & Schulzke, J. D. Altered permeability in inflammatory bowel disease: pathophysiology and clinical implications. Curr. Opin. Gastroenterol. 23, 379–383 (2007).
Su, L. et al. Targeted epithelial tight junction dysfunction causes immune activation and contributes to development of experimental colitis. Gastroenterology 136, 551–563 (2009).
Johansson, M. E. et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl Acad. Sci. USA 105, 15064–15069 (2008).
Velcich, A. et al. Colorectal cancer in mice genetically deficient in the mucin Muc2. Science 295, 1726–1729 (2002).
Van der Sluis, M. et al. Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 131, 117–129 (2006).
Heazlewood, C. K. et al. Aberrant mucin assembly in mice causes endoplasmic reticulum stress and spontaneous inflammation resembling ulcerative colitis. PLoS Med. 5, e54 (2008).
Abreu, M. T. Toll-like receptor signalling in the intestinal epithelium: how bacterial recognition shapes intestinal function. Nature Rev. Immunol. 10, 131–144 (2010).
Salzman, N. H. et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nature Immunol. 11, 76–83 (2010).
Grivennikov, S. I., Greten, F. R. & Karin, M. Immunity, inflammation, and cancer. Cell 140, 883–899 (2010). A comprehensive and up-to-date review of many of the molecular mechanisms involved in the interactions between inflammation and cancer.
Nenci, A. et al. Epithelial NEMO links innate immunity to chronic intestinal inflammation. Nature 446, 557–561 (2007).
Steinbrecher, K. A., Harmel-Laws, E., Sitcheran, R. & Baldwin, A. S. Loss of epithelial RelA results in deregulated intestinal proliferative/apoptotic homeostasis and susceptibility to inflammation. J. Immunol. 180, 2588–2599 (2008).
Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661–678 (2007).
Vereecke, L. et al. Enterocyte-specific A20 deficiency sensitizes to tumor necrosis factor-induced toxicity and experimental colitis. J. Exp. Med. 207, 1513–1523 (2010).
Zeissig, S. et al. Downregulation of epithelial apoptosis and barrier repair in active Crohn's disease by tumour necrosis factor α antibody treatment. Gut 53, 1295–1302 (2004).
van den Brande, J. M. et al. Prediction of antitumour necrosis factor clinical efficacy by real-time visualisation of apoptosis in patients with Crohn's disease. Gut 56, 509–517 (2007).
Greten, F. R. et al. IKKβ links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118, 285–296 (2004). A seminal paper showing the role in carcinogenesis of NF-κB expression in IECs versus haematopoietic cells.
Medzhitov, R. Origin and physiological roles of inflammation. Nature 454, 428–435 (2008).
Liew, F. Y., Xu, D., Brint, E. K. & O'Neill, L. A. Negative regulation of Toll-like receptor-mediated immune responses. Nature Rev. Immunol. 5, 446–458 (2005).
Lebeer, S., Vanderleyden, J. & de Keersmaecker, S. C. Host interactions of probiotic bacterial surface molecules: comparison with commensals and pathogens. Nature Rev. Microbiol. 8, 171–184 (2010).
Hooper, L. V. & Macpherson, A. J. Immune adaptations that maintain homeostasis with the intestinal microbiota. Nature Rev. Immunol. 10, 159–169 (2010).
Xiao, H. et al. The Toll–interleukin-1 receptor member SIGIRR regulates colonic epithelial homeostasis, inflammation, and tumorigenesis. Immunity 26, 461–475 (2007). A demonstration of the inflammatory and carcinogenic role of enhanced innate immune responses in the absence of SIGIRR.
Rakoff-Nahoum, S., Paglino, J., Eslami-Varzaneh, F., Edberg, S. & Medzhitov, R. Recognition of commensal microflora by Toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 (2004).
Salcedo, R. et al. MyD88-mediated signaling prevents development of adenocarcinomas of the colon: role of interleukin 18. J. Exp. Med. 207, 1625–1636 (2010). A demonstration of the protective role of MYD88 and IL-18 in colitis-associated colorectal cancer.
Clevers, H. At the crossroads of inflammation and cancer. Cell 118, 671–674 (2004).
Lee, J. et al. Maintenance of colonic homeostasis by distinctive apical TLR9 signalling in intestinal epithelial cells. Nature Cell Biol. 8, 1327–1336 (2006).
Katakura, K. et al. Toll-like receptor 9-induced type I IFN protects mice from experimental colitis. J. Clin. Invest. 115, 695–702 (2005).
Cho, J. H. The genetics and immunopathogenesis of inflammatory bowel disease. Nature Rev. Immunol. 8, 458–466 (2008).
Coulombe, F. et al. Increased NOD2-mediated recognition of N-glycolyl muramyl dipeptide. J. Exp. Med. 206, 1709–1716 (2009).
Watanabe, T., Kitani, A., Murray, P. J. & Strober, W. NOD2 is a negative regulator of Toll-like receptor 2-mediated T helper type 1 responses. Nature Immunol. 5, 800–808 (2004).
Watanabe, T. et al. Muramyl dipeptide activation of nucleotide-binding oligomerization domain 2 protects mice from experimental colitis. J. Clin. Invest. 118, 545–559 (2008).
Magalhaes, J. G. et al. Nod2-dependent Th2 polarization of antigen-specific immunity. J. Immunol. 181, 7925–7935 (2008).
Biswas, A. et al. Induction and rescue of Nod2-dependent Th1-driven granulomatous inflammation of the ileum. Proc. Natl Acad. Sci. USA 107, 14739–14744 (2010).
Wehkamp, J. et al. Reduced Paneth cell α-defensins in ileal Crohn's disease. Proc. Natl Acad. Sci. USA 102, 18129–18134 (2005).
Smith, A. M. et al. Disordered macrophage cytokine secretion underlies impaired acute inflammation and bacterial clearance in Crohn's disease. J. Exp. Med. 206, 1883–1897 (2009).
Noguchi, E., Homma, Y., Kang, X., Netea, M. G. & Ma, X. A Crohn's disease-associated NOD2 mutation suppresses transcription of human IL10 by inhibiting activity of the nuclear ribonucleoprotein hnRNP-A1. Nature Immunol. 10, 471–479 (2009).
Munz, C. Enhancing immunity through autophagy. Annual Rev. Immunol. 27, 423–449 (2009).
Virgin, H. W. & Levine, B. Autophagy genes in immunity. Nature Immunol. 10, 461–470 (2009).
Rioux, J. D. et al. Genome-wide association study identifies new susceptibility loci for Crohn disease and implicates autophagy in disease pathogenesis. Nature Genet. 39, 596–604 (2007).
Hampe, J. et al. A genome-wide association scan of nonsynonymous SNPs identifies a susceptibility variant for Crohn disease in ATG16L1. Nature Genet. 39, 207–211 (2007).
McCarroll, S. A. et al. Deletion polymorphism upstream of IRGM associated with altered IRGM expression and Crohn's disease. Nature Genet. 40, 1107–1112 (2008).
Cooney, R. et al. NOD2 stimulation induces autophagy in dendritic cells influencing bacterial handling and antigen presentation. Nature Med. 16, 90–97 (2010).
Travassos, L. H. et al. Nod1 and Nod2 direct autophagy by recruiting ATG16L1 to the plasma membrane at the site of bacterial entry. Nature Immunol. 11, 55–62 (2009).
Cadwell, K. et al. A key role for autophagy and the autophagy gene Atg16l1 in mouse and human intestinal Paneth cells. Nature 456, 259–263 (2008).
Cadwell, K. et al. Virus-plus-susceptibility gene interaction determines Crohn's disease gene Atg16L1 phenotypes in intestine. Cell 141, 1135–1145 (2010). An important demonstration that interaction between viruses and commensal flora affects intestinal pathophysiology.
Saitoh, T. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature 456, 264–268 (2008).
Le Bourhis, L., Benko, S. & Girardin, S. E. Nod1 and Nod2 in innate immunity and human inflammatory disorders. Biochem. Soc. Trans. 35, 1479–1484 (2007).
Petrilli, V., Dostert, C., Muruve, D. A. & Tschopp, J. The inflammasome: a danger sensing complex triggering innate immunity. Curr. Opin. Immunol. 19, 615–622 (2007).
Cookson, B. T. & Brennan, M. A. Pro-inflammatory programmed cell death. Trends Microbiol. 9, 113–114 (2001).
Labbé, K. & Saleh, M. Cell death in the host response to infection. Cell Death Differ. 15, 1339–1349 (2008).
Saleh, M. et al. Differential modulation of endotoxin responsiveness by human caspase-12 polymorphisms. Nature 429, 75–79 (2004).
Saleh, M. et al. Enhanced bacterial clearance and sepsis resistance in caspase-12-deficient mice. Nature 440, 1064–1068 (2006). A demonstration of the role of caspase 12 in the control of the inflammatory response to bacterial infections.
Yeretssian, G., Labbé, K. & Saleh, M. Molecular regulation of inflammation and cell death. Cytokine 43, 380–390 (2008).
Villani, A. C. et al. Common variants in the NLRP3 region contribute to Crohn's disease susceptibility. Nature Genet. 41, 71–76 (2009).
Pizarro, T. T. et al. IL-18, a novel immunoregulatory cytokine, is up-regulated in Crohn's disease: expression and localization in intestinal mucosal cells. J. Immunol. 162, 6829–6835 (1999).
Zhernakova, A. et al. Genetic analysis of innate immunity in Crohn's disease and ulcerative colitis identifies two susceptibility loci harboring CARD9 and IL18RAP. Am. J. Hum. Genet. 82, 1202–1210 (2008).
Rodriguez-Bores, L., Fonseca, G. C., Villeda, M. A. & Yamamoto-Furusho, J. K. Novel genetic markers in inflammatory bowel disease. World J. Gastroenterol. 13, 5560–5570 (2007).
Dinarello, C. A. Interleukin-18 and the pathogenesis of inflammatory diseases. Semin. Nephrol. 27, 98–114 (2007).
Sivakumar, P. V. et al. Interleukin 18 is a primary mediator of the inflammation associated with dextran sulphate sodium induced colitis: blocking interleukin 18 attenuates intestinal damage. Gut 50, 812–820 (2002).
Reuter, B. K. & Pizarro, T. T. Commentary: the role of the IL-18 system and other members of the IL-1R/TLR superfamily in innate mucosal immunity and the pathogenesis of inflammatory bowel disease: friend or foe? Eur. J. Immunol. 34, 2347–2355 (2004).
Takagi, H. et al. Contrasting action of IL-12 and IL-18 in the development of dextran sodium sulphate colitis in mice. Scand. J. Gastroenterol. 38, 837–844 (2003).
Kampfer, H. et al. Lack of interferon-γ production despite the presence of interleukin-18 during cutaneous wound healing. Mol. Med. 6, 1016–1027 (2000).
Siegmund, B. Interleukin-18 in intestinal inflammation: friend and foe? Immunity 32, 300–302 (2010).
Dupaul-Chicoine, J. et al. Control of intestinal homeostasis, colitis, and colitis-associated colorectal cancer by the inflammatory caspases. Immunity 32, 367–378 (2010). A demonstration of the role of ASC and caspase 1 in DSS-induced colitis and of caspase 12 in colitis and colitis-associated colorectal cancer.
Zaki, M. H. et al. The NLRP3 inflammasome protects against loss of epithelial integrity and mortality during experimental colitis. Immunity 32, 379–391 (2010). A demonstration of the role of NLRP3 in colitis.
Allen, I. C. et al. The NLRP3 inflammasome functions as a negative regulator of tumorigenesis during colitis-associated cancer. J. Exp. Med. 207, 1045–1056 (2010). A demonstration of the role of caspase 1, ASC and NLRP3 in colitis-associated colorectal cancer.
Terzic, J., Grivennikov, S., Karin, E. & Karin, M. Inflammation and colon cancer. Gastroenterology 138, 2101–2114 (2010).
Bollrath, J. et al. gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell 15, 91–102 (2009).
Grivennikov, S. et al. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell 15, 103–113 (2009). A characterization of the role of the IL-6–STAT3 axis in colon homeostasis and carcinogenesis.
Neufert, C. et al. Activation of epithelial STAT3 regulates intestinal homeostasis. Cell Cycle 9, 652–655 (2010).
Sears, C. L. Enterotoxigenic Bacteroides fragilis: a rogue among symbiotes. Clin. Microbiol. Rev. 22, 349–369 (2009).
Maddocks, O. D., Short, A. J., Donnenberg, M. S., Bader, S. & Harrison, D. J. Attaching and effacing Escherichia coli downregulate DNA mismatch repair protein in vitro and are associated with colorectal adenocarcinomas in humans. PLoS ONE 4, e5517 (2009).
Uronis, J. M. et al. Modulation of the intestinal microbiota alters colitis-associated colorectal cancer susceptibility. PLoS ONE 4, e6026 (2009).
Vannucci, L. et al. Colorectal carcinogenesis in germ-free and conventionally reared rats: different intestinal environments affect the systemic immunity. Int. J. Oncol. 32, 609–617 (2008).
Yang, L. & Pei, Z. Bacteria, inflammation, and colon cancer. World J. Gastroenterol. 12, 6741–6746 (2006).
Rao, V. P. et al. Innate immune inflammatory response against enteric bacteria Helicobacter hepaticus induces mammary adenocarcinoma in mice. Cancer Res. 66, 7395–7400 (2006). A demonstration of the systemic effect of intestinal inflammation on carcinogenesis in distant organs.
Westbrook, A. M., Wei, B., Braun, J. & Schiestl, R. H. Intestinal mucosal inflammation leads to systemic genotoxicity in mice. Cancer Res. 69, 4827–4834 (2009).
Lee, S. H. et al. ERK activation drives intestinal tumorigenesis in Apcmin/+ mice. Nature Med. 16, 665–670 (2010).
Rakoff-Nahoum, S. & Medzhitov, R. Regulation of spontaneous intestinal tumorigenesis through the adaptor protein MyD88. Science 317, 124–127 (2007). This article describes a role for MYD88 and TLRs in the regulation of intestinal homeostasis and carcinogenesis.
Swann, J. B. et al. Demonstration of inflammation-induced cancer and cancer immunoediting during primary tumorigenesis. Proc. Natl Acad. Sci. USA 105, 652–656 (2008).
Naugler, W. E. et al. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science 317, 121–124 (2007).
Lowe, E. L. et al. Toll-like receptor 2 signaling protects mice from tumor development in a mouse model of colitis-induced cancer. PLoS ONE 5, e13027 (2010).
Fukata, M. et al. Toll-like receptor-4 promotes the development of colitis-associated colorectal tumors. Gastroenterology 133, 1869–1881 (2007).
Turovskaya, O. et al. RAGE, carboxylated glycans and S100A8/A9 play essential roles in colitis-associated carcinogenesis. Carcinogenesis 29, 2035–2043 (2008).
Maeda, S. et al. Essential roles of high-mobility group box 1 in the development of murine colitis and colitis-associated cancer. Biochem. Biophys. Res. Commun. 360, 394–400 (2007).
Ranzato, E., Patrone, M., Pedrazzi, M. & Burlando, B. Hmgb1 promotes wound healing of 3T3 mouse fibroblasts via RAGE-dependent ERK1/2 activation. Cell Biochem. Biophys. 57, 9–17 (2010).
Rodier, F. et al. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nature Cell Biol. 11, 973–979 (2009).
Dunn, G. P., Koebel, C. M. & Schreiber, R. D. Interferons, immunity and cancer immunoediting. Nature Rev. Immunol. 6, 836–848 (2006).
Popivanova, B. K. et al. Blocking TNF-α in mice reduces colorectal carcinogenesis associated with chronic colitis. J. Clin. Invest. 118, 560–570 (2008).
Kim, J. J. et al. Helicobacter pylori impairs DNA mismatch repair in gastric epithelial cells. Gastroenterology 123, 542–553 (2002).
Hanada, T. et al. IFNγ-dependent, spontaneous development of colorectal carcinomas in SOCS1-deficient mice. J. Exp. Med. 203, 1391–1397 (2006).
Garlanda, C. et al. Increased susceptibility to colitis-associated cancer of mice lacking TIR8, an inhibitory member of the interleukin-1 receptor family. Cancer Res. 67, 6017–6021 (2007). References 116 and 117 demonstrate the role of SIGIRR in colon carcinogenesis.
Xiao, H. et al. Loss of single immunoglobulin interlukin-1 receptor-related molecule leads to enhanced colonic polyposis in Apcmin mice. Gastroenterology 139, 574–585 (2010).
Vaishnava, S., Behrendt, C. L., Ismail, A. S., Eckmann, L. & Hooper, L. V. Paneth cells directly sense gut commensals and maintain homeostasis at the intestinal host-microbial interface. Proc. Natl Acad. Sci. USA 105, 20858–20863 (2008).
Penders, J. et al. Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics 118, 511–521 (2006).
Turnbaugh, P. J., Backhed, F., Fulton, L. & Gordon, J. I. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe 3, 213–223 (2008).
Backhed, F. et al. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl Acad. Sci. USA 101, 15718–15723 (2004).
Backhed, F., Ley, R. E., Sonnenburg, J. L., Peterson, D. A. & Gordon, J. I. Host-bacterial mutualism in the human intestine. Science 307, 1915–1920 (2005).
Boivin, G. P. et al. Pathology of mouse models of intestinal cancer: consensus report and recommendations. Gastroenterology 124, 762–777 (2003).
Rosenberg, D. W., Giardina, C. & Tanaka, T. Mouse models for the study of colon carcinogenesis. Carcinogenesis 30, 183–196 (2009).
Chen, J. & Huang, X. F. The signal pathways in azoxymethane-induced colon cancer and preventive implications. Cancer Biol. Ther. 8, 1313–1317 (2009).
Kohno, H., Suzuki, R., Sugie, S. & Tanaka, T. β-Catenin mutations in a mouse model of inflammation-related colon carcinogenesis induced by 1,2-dimethylhydrazine and dextran sodium sulfate. Cancer Sci. 96, 69–76 (2005).
Tanaka, T. et al. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 94, 965–973 (2003).
Moser, A. R. et al. ApcMin, a mutation in the murine Apc gene, predisposes to mammary carcinomas and focal alveolar hyperplasias. Proc. Natl Acad. Sci. USA 90, 8977–8981 (1993).
Suzui, M., Okuno, M., Tanaka, T., Nakagama, H. & Moriwaki, H. Enhanced colon carcinogenesis induced by azoxymethane in min mice occurs via a mechanism independent of β-catenin mutation. Cancer Lett. 183, 31–41 (2002).
Acknowledgements
M.S. thanks the students and colleagues in her laboratory for reading and commenting on this manuscript. Work in M.S.'s laboratory is supported by the Canadian Institutes for Health Research (MOP-79410, MOP-82801, MOP-86546, CTP-87520) and the Burroughs Wellcome Fund. Work in G.T.'s laboratory is supported by the Intramural Research Program of the US National Institutes of Health, National Cancer Institute, Center for Cancer Research, USA.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Helminth therapy
-
Helminth therapy is a form of immunotherapy aimed at modulating the T helper 1 (TH1)/TH2 immune balance. It involves the deliberate inoculation of patients suffering from immune-mediated inflammatory diseases with helminth (parasitic intestinal nematodes) or helminth larvae. The currently studied regimens in humans use Trichuris suis, which does not cross the intestinal barrier or cause an invasive infection.
- Autophagy
-
A tightly regulated catabolic process that involves degradation of the cell's own intracellular components through the lysosomal machinery. It is a normal part of cell growth, development and homeostasis, and recently it has emerged as an efficient innate immunity mechanism.
- Villi
-
Villi are projections into the intestinal lumen. Their outer epithelial layer consists mainly of mature, absorptive enterocytes but also contains mucus-secreting goblet cells.
- Crypts
-
Crypts are tubular invaginations of the intestinal epithelium. At the base of the crypts are Paneth cells, which produce bactericidal defensins, and stem cells, which continuously divide and are the source of all intestinal epithelial cells.
- Tight junction
-
A ring of proteins that seals apical epithelium; these proteins include the integral membrane proteins occludin and claudin, in association with cytoplasmic zonula occludins proteins.
- Lamina propria
-
The connective tissue that underlies the mucosal epithelium and contains various myeloid and lymphoid cells, including macrophages, dendritic cells, B cells and T cells.
- Wnt pathway
-
A signalling pathway that regulates cell fate determination, proliferation, adhesion, migration and polarity during development. In addition to the crucial role of this pathway in embryogenesis, Wnt ligands and their downstream signalling molecules have been implicated in tumorigenesis and have causative roles in human colon cancers.
- Adenomatous polyposis coli
-
(APC). A scaffold protein that sequesters β-catenin in the cytoplasm of resting cells. APC mutations, which are found in 90% of intestinal cancers, lead to constitutively active β-catenin.
- Inflammasome
-
A large multiprotein complex formed by a NOD- and LRR-containing (NLR) protein, the adaptor protein apoptosis-associated speck-like protein containing a CARD (ASC; also known as PYCARD) and pro-caspase 1. The assembly of the inflammasome leads to the activation of caspase 1, which cleaves pro-interleukin-1 β (pro-IL-1β) and pro-IL-18 to generate the active pro-inflammatory cytokines.
- Pyroptosis
-
A form of cell death that is distinct from immunologically silent apoptosis. It is triggered concomitantly with the activation of the inflammasome and requires caspase 1 activity. During pyroptotic cell death, an inflammasome complex forms to activate caspase 1.
- ApcMin/+ mice
-
A mouse strain that carries a point mutation in one adenomatous polyposis coli (Apc) allele and spontaneously develops intestinal adenomas. It is used as a model for human familial adenomatous polyposis and for human sporadic colorectal cancer.
- Tumour immunosurveillance
-
Tumour immunosurveillance or attack refers to the identification and elimination of cancerous or pre-cancerous cells by the immune system. However, as tumours do still develop despite a functioning immune system, the concept of 'immune editing' has taken over.
Rights and permissions
About this article
Cite this article
Saleh, M., Trinchieri, G. Innate immune mechanisms of colitis and colitis-associated colorectal cancer. Nat Rev Immunol 11, 9–20 (2011). https://doi.org/10.1038/nri2891
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri2891
This article is cited by
-
Intratumoral microbiota: roles in cancer initiation, development and therapeutic efficacy
Signal Transduction and Targeted Therapy (2023)
-
A Potential Role for Gut Microbes in Mediating Effects of Omega-3 Fatty Acids in Inflammatory Bowel Diseases: A Comprehensive Review
Current Microbiology (2023)
-
PD-1 signaling facilitates activation of lymphoid tissue inducer cells by restraining fatty acid oxidation
Nature Metabolism (2022)
-
Blockade of Myd88 signaling by a novel MyD88 inhibitor prevents colitis-associated colorectal cancer development by impairing myeloid-derived suppressor cells
Investigational New Drugs (2022)
-
Using BioPAX-Parser (BiP) to enrich lists of genes or proteins with pathway data
BMC Bioinformatics (2021)