Key Points
-
Clinical trials in interventional oncology have been successful in demonstrating clinical efficacy, but limited data are available on patient-reported outcomes and cost-effectiveness
-
Investigators have met several barriers in conducting phase III trials in interventional oncology, particularly relating to timely recruitment
-
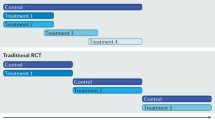
Important methodological challenges should be considered when planning trials in interventional oncology, particularly those related to selection of trial end points
-
Multicentre trials require careful quality assurance to ensure interventions are consistent across participating centres
-
Innovative and pragmatic approaches to generating robust clinical effectiveness data are being implemented in contemporary trial designs, including the use of registries in evaluating a treatment during commissioning
Abstract
Interventional oncology is a rapidly growing sub-speciality that aims to develop new disease-modifying treatment options beyond conventional surgical and oncological therapies in several disease settings. The evidence for interventional oncology success is dominated by single-arm studies reporting technical success or clinical efficacy. These studies have successfully resulted in the development of new techniques, but are not sufficient to change clinical practice uniformly across health-care systems. This Review discusses why clinical investigators must incorporate measures of cost-effectiveness and patient-reported outcomes into large-scale studies currently being designed to provide robust evidence for changing clinical practice. In particular, interventional oncology trials could be designed to show that certain treatments may be as effective as the current standard of care, but with significantly less morbidity and with better outcomes for patients with cancer. Innovative trial design and awareness of the challenges from interventional studies in other fields of medicine and surgery are also discussed to demonstrate how this new speciality can make progress. Registry-based models are emerging as an alternative means of deriving cohort data and can be used in parallel with local or national commissioning of new services.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Clinical Oncology Faculty Board & Clinical Radiology Faculty Board. Interventional oncology: guidance for service delivery. The Royal College of Radiologists [online], (2013).
OCEBM Levels of Evidence Working Group. The Oxford Levels of Evidence 2. Oxford Centre for Evidence-Based Medicine [online], (2011)
Loveman, E. et al. The clinical effectiveness and cost-effectiveness of ablative therapies in the management of liver metastases: systematic review and economic evaluation. Health Technol. Assess. 18, 1–284 (2014).
Guyatt, G. H. et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336, 924–926 (2008).
Lencioni, R. et al. Response to radiofrequency ablation of pulmonary tumours: a prospective, intention-to-treat, multicentre clinical trial (the RAPTURE study). Lancet Oncol. 9, 621–628 (2008).
On Behalf of the PRECISION V Investigators, Lammer, J. et al. Prospective Randomized Study of Doxorubicin-Eluting-Bead Embolization in the Treatment of Hepatocellular Carcinoma: Results of the PRECISION V Study. Cardiovasc. Intervent. Radiol. 33, 41–52 (2009).
Yamakado, K. et al. Radiofrequency ablation for the treatment of unresectable lung metastases in patients with colorectal cancer: a multicenter study in Japan. J. Vasc. Interv. Radiol. 3, 393–398 (2007).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Bruix, J. et al. Transarterial embolization versus symptomatic treatment in patients with advanced hepatocellular carcinoma: results of a randomized, controlled trial in a single institution. Hepatology 27, 1578–1583 (1998).
Llovet, J. M. et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 359, 378–390 (2008).
Ricke, J. & Malfertheiner, P. Evaluation of sorafenib in combination with local micro-therapy guided by Gd-EOB-DTPA enhanced MRI in patients with inoperable hepatocellular carcinoma. Clinical Trials in HCC [online], (2013).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Sharma, R. & Wasan, H. Foxfire Update. Oncology Clinical Trials Office [online], (2014).
The Sirflox Study Group. The Sirflox Study [online], (2014).
Dutton, S. J. et al. FOXFIRE protocol: an open-label, randomised, phase III trial of 5-fluorouracil, oxaliplatin and folinic acid (OxMdG) with or without interventional Selective Internal Radiation Therapy (SIRT) as first-line treatment for patients with unresectable liver-only or liver-dominant metastatic colorectal cancer. BMC Cancer 14, 497 (2014).
Poston, G. J. in Textbook of Surgical Oncology 1st edn Ch. 1. (eds Poston, G. J., Beauchamp, R. D. & Ruers, T.) 1–4 (Informa Healthcare, 2007).
Solomon, M. J. & McLeod, R. S. Clinical studies in surgical journals— have we improved? Dis. Colon. Rectum 36, 43–48 (1993).
Ergina, P. L. et al. Surgical Innovation and Evaluation 2: Challenges in evaluating surgical innovation. Lancet 26, 1097–1104 (2009).
[No authors listed]. Surgical research: the reality and the IDEAL. Lancet 374, 1037 (2009).
McCulloch, P. et al. Surgical Innovation and Evaluation 3: No surgical innovation without evaluation: the IDEAL recommendations. Lancet 374, 1105–1112 (2009).
Barkun, J. S. et al. Surgical Innovation and Evaluation 1: Evaluation and stages of surgical innovations. Lancet 374, 1089–1096 (2009).
Nordlinger, B. et al. Perioperative chemotherapy with FOLFOX4 and surgery for resectable liver metastases from colorectal cancer. Lancet 371, 1007–1016 (2008).
Yip, V. S. et al. Optimal imaging sequence for staging in colorectal liver metastases: Analysis of three hypothetical imaging strategies. Eur. J. Cancer 50, 937–943 (2014).
Ruers, T. et al. Radiofrequency ablation combined with systemic treatment versus systemic treatment alone in patients with non-resectable colorectal liver metastases: a randomized EORTC Intergroup phase II study (EORTC 40004). Ann Oncol. 23, 2619–2626 (2012).
Medical Research Council Clinical Trials Unit. FOCUS 3 [online], (2010).
Medical Research Council Clinical Trials Unit. FOCUS 4 [online], (2014).
Songun, I., Putter, H., Kranenbarg, E. M., Sasako M. & van de Velde, C. J. Surgical treatment of gastric cancer: 15-year follow-up results of the randomised nationwide Dutch D1D2 trial. Lancet Oncol. 11, 439–449 (2010).
van Gijn, W. et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 12, 575–582 (2011).
Primrose, J. et al. Systemic chemotherapy with or without cetuximab in patients with resectable colorectal liver metastasis: the New EPOC randomised controlled trial. Lancet Oncol. 15, 601–611 (2014).
Weng, M. et al. Radiofrequency ablation versus resection for colorectal cancer liver metastases: a meta-analysis. PLoS ONE 7, e45493 (2012).
Jones, B., Jarvis, P., Lewis, J. A. & Ebbutt, E. Trials to assess equivalence: the importance of rigorous methods. BMJ 313, 36–39 (1996).
Cirocchi, R. et al. Radiofrequency ablation in the treatment of liver metastases from colorectal cancer. Cochrane Database of Systematic Reviews. Issue 6. Art. No.: CD006317 http://dx.doi.org/10.1002/14651858.CD006317.pub3.
Weis, S., Franke, A., Mössner, J., Jakobsen, J. C. & Schoppmeyer, K. Radiofrequency (thermal) ablation versus no intervention or other interventions for hepatocellular carcinoma. Cochrane Database of Systematic Reviews. Issue 12. Art. No.: CD003046. http://dx.doi.org/10.1002/14651858.CD003046.pub3.
Sharma, R. A., Anthony, S. & Anderson, E. M. Clinical benefit and potential pitfalls in combining thermal ablation and radiation therapy to treat liver metastases. J. Clin. Oncol. 31, e404–e406 (2013).
Ibrahim, S. et al. Radiologic findings following Y90 radioembolization. Abdom. Imaging 34, 566–581 (2008).
Bassel Atassi, B. et al. Multimodality imaging following 90Y radioembolization: A comprehensive review and pictorial essay. Radiographics 28, 81–99 (2008).
Van Hazel, G. et al. Randomised phase 2 trial of SIR-Spheres plus fluorouracil/leucovorin chemotherapy versus fluorouracil/leucovorin chemotherapy alone in advanced colorectal cancer. J. Surg. Oncol. 88, 78–85 (2004).
Gray, B. et al. Randomised trial of SIR-Spheres® plus chemotherapy vs. chemotherapy alone for treating patients with liver metastases from primary large bowel cancer. Ann. Oncol. 12, 1711–1720 (2001).
Donnelly, B. J. et al. A randomized trial of external beam radiotherapy versus cryoablation in patients with localized prostate cancer. Cancer 116, 323–330 (2010).
Caplan, R., Pajak, T. & Cox, M. Analysis of the probability and risk of cause specific failure. Int. J. Radiat. Oncol. Biol. Phys. 29, 1183–1186 (1994).
Tai, B., Machin, D., White, I. & Gebski, V. Competing risks analysis of patients with osteosarcoma: a comparison of four different approaches. Stat. Med. 20, 661–684 (2001).
Saad, E. D., Katz, A., Hoff, P. M. & Buyse, M. Progression-free survival as surrogate and as true end point: insights from the breast and colorectal cancer literature. Ann. Oncol. 21, 7–12 (2009).
Homs, M. Y. V. et al. Single-dose brachytherapy versus metal stent placement for the palliation of dysphagia from oesophageal cancer: multicentre randomised trial. Lancet 364, 1497–1504 (2004).
The HTA commissioning team. HTA commissioned funding opportunities. The National Institute for Health Research (UK) [online], (2014).
Buchbinder, R. et al. A randomized trial of vertebroplasty for painful osteoporotic vertebral fractures. N. Engl. J. Med. 361, 557–568 (2009).
Kornblith, A. B. et al. Quality of life of patients with endometrial cancer undergoing laparoscopic international federation of gynecology and obstetrics staging compared with laparotomy: a Gynecologic Oncology Group study. J. Clin. Oncol. 27, 5337–5342 (2009).
Janda, M. et al. Quality of life after total laparoscopic hysterectomy versus total abdominal hysterectomy for stage I endometrial cancer (LACE): a randomised trial. Lancet Oncol. 11, 272–280 (2010).
Hasan, A., Pozzi, M. & Hamilton, J. R. New surgical procedures: can we minimise the learning curve? BMJ 320, 171–173 (2000).
Mirnezami, R. et al. Short- and long-term outcomes after laparoscopic and open hepatic resection: systematic review and meta-analysis. HPB (Oxford) 13, 295–308 (2011).
National Cancer Research Institute (UK). CTRad: Clinical and Translational Radiotherapy Research Working Group [online], (2013).
Collinson, F. J. et al. An international, multicentre, prospective, randomised, controlled, unblinded, parallel-group trial of robotic-assisted versus standard laparoscopic surgery for the curative treatment of rectal cancer. Int. J. Colorectal Dis. 27, 233–241 (2011).
Bach, S. Transanal Endoscopic Microsurgery (TEM) and Radiotherapy in Early Rectal Cancer (TREC). UK Clinical Research Network Portfolio Database [online], (2012).
O'Quigley, J. O., Pepe, M. & Fisher, L. Continual reassessment method: a practical design for phase I clinical trials in cancer. Biometrics 46, 33–48 (1990).
Iasonos, A., Wilton, A. S., Riedel, E. R., Seshan, V. E. & Spriggs, D. R. A comprehensive comparison of the continual reassessment method to the standard 3 + 3 dose escalation scheme in phase I dose-finding studies. Clinical Trials 5, 465–477 (2008).
Garrett-Mayer, E. Understanding the Continual Reassessment Method for Dose Finding Studies: An Overview for Non-Statisticians, Johns Hopkins University, Department of Biostatistics Working Papers. Working Paper 74. Berkeley Electronic Press [online], (2005).
Orloff, J. et al. The future of drug development: advancing clinical trial design. Nat. Rev. Drug Discov. 8, 949–957 (2009).
Brady, M. & Gebski, V. in Controversies in the Management of Gynaecological Cancers 1st edn Ch. 25 (eds Ledermann, J., Creutzberg, C. & Quinn, M.) 263–284 (Springer, 2014).
Shibata, T., Niinobu, T., Ogata, N. & Takami, M. Microwave coagulation therapy for multiple hepatic metastases from colorectal carcinoma. Cancer 89, 276–284 (2000).
Solbiati, L. et al. Percutaneous radio-frequency ablation of hepatic metastases from colorectal cancer: long-term results in 117 patients. Radiology 221, 159–166 (2001).
Mack, M. G. et al. Percutaneous MR imaging-guided laser-induced thermotherapy of hepatic metastases. Abdom. Imaging 26, 369–374 (2001).
Berber, E., Pelley, R. & Siperstein, A. E. Predictors of survival after radiofrequency thermal ablation of colorectal cancer metastases to the liver: a prospective study. J. Clin. Oncol. 23, 1358–1364 (2005).
Siperstein, A. E., Berber, E., Ballem, N. & Parikh, R. T. Survival after radiofrequency ablation of colorectal liver metastases: 10-year experience. Ann. Surg. 246, 559–565 (2007).
Sorensen, S. M., Mortensen, F. V. & Nielsen, D. T. Radiofrequency ablation of colorectal liver metastases: long-term survival. Acta Radiol. 48, 253–258 (2007).
Gillams, A. R. & Lees, W. R. Five-year survival in 309 patients with colorectal liver metastases treated with radiofrequency ablation. Eur. Radiol. 19, 1206–1213 (2009).
Kim, K. H. et al. Comparative analysis of radiofrequency ablation and surgical resection for colorectal liver metastases. J. Korean Surg. Soc. 81, 25–34 (2011).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Wahl, R. L., Jacene, H., Kasamon, Y. & Lodge, M. A. From RECIST to PERCIST: Evolving Considerations for PET Response Criteria in Solid Tumors. J. Nucl. Med. 50, 122S–150S (2009).
Acknowledgements
R.A.S. is supported by the NIHR Biomedical Research Centre Oxford, the Bobby Moore Fund of Cancer Research UK and the UK Medical Research Council.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided substantial contributions to discussions of its content, wrote the article and undertook review and/or editing of the manuscript before submission and following peer review.
Corresponding author
Ethics declarations
Competing interests
R.A.S. declares that he has been the principal investigator for unrestricted educational grants from Sirtex. The other authors declare no competing interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Franklin, J., Gebski, V., Poston, G. et al. Clinical trials of interventional oncology—moving from efficacy to outcomes. Nat Rev Clin Oncol 12, 93–104 (2015). https://doi.org/10.1038/nrclinonc.2014.199
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2014.199
This article is cited by
-
Local Tumour Control Following Microwave Ablation: Protocol for the Prospective Observational CIEMAR Study
CardioVascular and Interventional Radiology (2024)
-
Large-scale data from real-life practice of percutaneous liver thermal ablation from an international registry: unconditionally trustful Atlas or colossus with feet of clay?
European Radiology (2023)
-
The need for multidisciplinarity in specialist training to optimize future patient care
Nature Reviews Clinical Oncology (2017)