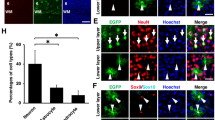
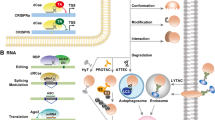
Abstract
This protocol describes a method for directing the expression of genes of interest into postmitotic neocortical neurons in vivo. Microinjection of a DNA plasmid-amphiphilic molecule mix into the neocortex followed by delivery of an ad hoc electric pulse protocol during the first few days of life in mice allows rapid, focal and efficient expression of genes in postmitotic neurons. Compared with other gene delivery techniques such as in utero electroporation and viral infection, this method allows rapid (12 h), focal (50–200 μm), mosaic-like (50 to several hundred neurons) targeting of postmitotic neurons within existing circuits. This 'iontoporation' protocol, which can be completed within ∼20 min per mouse, allows straightforward assessment of genetic constructs in postmitotic cortical neurons and subsequent genetic, histological and physiological investigations of gene function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
De la Rossa, A. et al. In vivo reprogramming of circuit connectivity in postmitotic neocortical neurons. Nat. Neurosci. 16, 193–200 (2013).
Chang, D.C. & Reese, T.S. Changes in membrane structure induced by electroporation as revealed by rapid-freezing electron microscopy. Biophys. J. 58, 1–12 (1990).
Hovis, K.R., Padmanabhan, K. & Urban, N.N. A simple method of in vitro electroporation allows visualization, recording, and calcium imaging of local neuronal circuits. J. Neurosci. Methods 191, 1–10 (2010).
Capecchi, M.R. High-efficiency transformation by direct microinjection of DNA into cultured mammalian cells. Cell 22, 479–488 (1980).
Barnabé-Heider, F. et al. Genetic manipulation of adult mouse neurogenic niches by in vivo electroporation. Nat. Methods 5, 189–196 (2008).
Saito, T. In vivo electroporation in the embryonic mouse central nervous system. Nat. Protoc. 1, 1552–1558 (2006).
Washbourne, P. & McAllister, A.K. Techniques for gene transfer into neurons. Curr. Opin. Neurobiol. 12, 566–573 (2002).
De Vry, J. et al. In vivo electroporation of the central nervous system: a non-viral approach for targeted gene delivery. Prog. Neurobiol. 92, 227–244 (2010).
Rouaux, C. & Arlotta, P. Direct lineage reprogramming of post-mitotic callosal neurons into corticofugal neurons in vivo. Nat. Cell Biol. 15, 214–221 (2013).
LoTurco, J., Manent, J.-B. & Sidiqi, F. New and improved tools for in utero electroporation studies of developing cerebral cortex. Cereb. Cortex 19 (suppl. 1), i120–i125 (2009).
Judkewitz, B., Rizzi, M., Kitamura, K. & Häusser, M. Targeted single-cell electroporation of mammalian neurons in vivo. Nat. Protoc. 4, 862–869 (2009).
Matsui, A., Yoshida, A.C., Kubota, M., Ogawa, M. & Shimogori, T. Mouse in utero electroporation: controlled spatiotemporal gene transfection. J. Vis. Exp. 54, e3024 (2011).
Kitamura, K., Judkewitz, B., Kano, M., Denk, W. & Häusser, M. Targeted patch-clamp recordings and single-cell electroporation of unlabeled neurons in vivo. Nat. Methods 5, 61–67 (2008).
dal Maschio, M. et al. High-performance and site-directed in utero electroporation by a triple-electrode probe. Nat. Commun. 3, 960 (2012).
Taniguchi, H., Lu, J. & Huang, Z.J. The spatial and temporal origin of chandelier cells in mouse neocortex. Science 339, 70–74 (2013).
Mizuno, H., Hirano, T. & Tagawa, Y. Pre-synaptic and post-synaptic neuronal activity supports the axon development of callosal projection neurons during different post-natal periods in the mouse cerebral cortex. Eur. J. Neurosci. 31, 410–424 (2010).
Molotkov, D.A., Yukin, A.Y., Afzalov, R.A. & Khiroug, L.S. Gene delivery to postnatal rat brain by non-ventricular plasmid injection and electroporation. J. Vis. Exp. 43, e2244 (2010).
Ohmura, N., Kawasaki, K., Satoh, T. & Hata, Y. In vivo electroporation to physiologically identified deep brain regions in postnatal mammals. Brain Struct. Funct. 10.1007/s00429-014-0724-x (2014).
Inoue, M., Mishina, M. & Ueda, H. Locus-specific rescue of GluRepsilon1 NMDA receptors in mutant mice identifies the brain regions important for morphine tolerance and dependence. J. Neurosci. 23, 6529–6536 (2003).
Wei, F. et al. Calmodulin regulates synaptic plasticity in the anterior cingulate cortex and behavioral responses: a microelectroporation study in adult rodents. J. Neurosci. 23, 8402–8409 (2003).
Konishi, Y., Stegmüller, J., Matsuda, T., Bonni, S. & Bonni, A. Cdh1-APC controls axonal growth and patterning in the mammalian brain. Science 303, 1026–1030 (2004).
Akaneya, Y., Jiang, B. & Tsumoto, T. RNAi-induced gene silencing by local electroporation in targeting brain region. J. Neurophysiol. 93, 594–602 (2005).
Guo, W. et al. Supraspinal brain-derived neurotrophic factor signaling: a novel mechanism for descending pain facilitation. J. Neurosci. 26, 126–137 (2006).
Nagayama, S. et al. In vivo simultaneous tracing and Ca2+ imaging of local neuronal circuits. Neuron 53, 789–803 (2007).
Nevian, T. & Helmchen, F. Calcium indicator loading of neurons using single-cell electroporation. Pflugers Arch. 454, 675–688 (2007).
Wang, H., Ko, C.H., Koletar, M.M., Ralph, M.R. & Yeomans, J. Casein kinase I epsilon gene transfer into the suprachiasmatic nucleus via electroporation lengthens circadian periods of tau mutant hamsters. Eur. J. Neurosci. 25, 3359–3366 (2007).
Fan, J. et al. Up-regulation of anterior cingulate cortex NR2B receptors contributes to visceral pain responses in rats. Gastroenterology 136, 1732–1740 (2009).
De Vry, J. et al. Low current-driven micro-electroporation allows efficient in vivo delivery of nonviralDNA into the adult mouse brain. Mol. Ther. 18, 1183–1191 (2009).
Papale, A., Cerovic, M. & Brambilla, R. Viral vector approaches to modify gene expression in the brain. J. Neurosci. Methods 185, 1–14 (2009).
Lentz, T.B., Gray, S.J. & Samulski, R.J. Viral vectors for gene delivery to the central nervous system. Neurobiol. Dis. 48, 179–188 (2012).
Beier, K. & Cepko, C. Viral tracing of genetically defined neural circuitry. J. Vis. Exp. 68, e4253 (2012).
Osakada, F. & Callaway, E.M. Design and generation of recombinant rabies virus vectors. Nat. Protoc. 8, 1583–1601 (2013).
Jager, L. et al. A rapid protocol for construction and production of high-capacity adenoviral vectors. Nat. Protoc. 4, 547–564 (2009).
Liashkovich, I., Meyring, A., Kramer, A. & Shahin, V. Exceptional structural and mechanical flexibility of the nuclear pore complex. J. Cell. Physiol. 226, 675–682 (2011).
Vandenbroucke, R.E., Lucas, B., Demeester, J., De Smedt, S.C. & Sanders, N.N. Nuclear accumulation of plasmid DNA can be enhanced by non-selective gating of the nuclear pore. Nucleic Acids Res. 35, e86 (2007).
De Marco García, N.V., Karayannis, T. & Fishell, G. Neuronal activity is required for the development of specific cortical interneuron subtypes. Nature 472, 351–355 (2011).
Matsuda, T. & Cepko, C.L. Controlled expression of transgenes introduced by in vivo electroporation. Proc. Natl. Acad. Sci. USA 104, 1027–1032 (2007).
Cohen, S.N., Chang, A.C., Boyer, H.W. & Helling, R.B. Construction of biologically functional bacterial plasmids in vitro. Proc. Natl. Acad. Sci. USA 70, 3240 (1973).
Ausubel, F.M. et al. Short Protocols in Molecular Biology (John Wiley & Sons, 1995).
Cetin, A., Komai, S., Eliava, M., Seeburg, P.H. & Osten, P. Stereotaxic gene delivery in the rodent brain. Nat. Protoc. 1, 3166–3173 (2007).
Paxinos, G. & Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates (Gulf Professional Publishing, 2004).
Acknowledgements
We thank the members of our laboratory for helpful discussions. We also thank A. Benoit and F. Smets for technical assistance, L. Frangeul for help with the manuscript and L. Telley for photomicrograph image acquisition. Work in the Jabaudon laboratory is supported by the Swiss National Science Foundation, the Leenaards Foundation, the Brain and Behavior Foundation and the Synapsy Foundation.
Author information
Authors and Affiliations
Contributions
A.D.l.R. and D.J. developed the protocol and wrote the manuscript; A.D.l.R. performed the experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
De la Rossa, A., Jabaudon, D. In vivo rapid gene delivery into postmitotic neocortical neurons using iontoporation. Nat Protoc 10, 25–32 (2015). https://doi.org/10.1038/nprot.2015.001
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2015.001
This article is cited by
-
Targeted in vivo genetic manipulation of the mouse or rat brain by in utero electroporation with a triple-electrode probe
Nature Protocols (2016)
-
A cross-modal genetic framework for the development and plasticity of sensory pathways
Nature (2016)
-
Astroglial β-Arrestin1-mediated Nuclear Signaling Regulates the Expansion of Neural Precursor Cells in Adult Hippocampus
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.