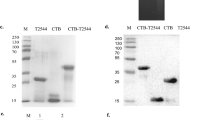
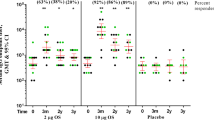
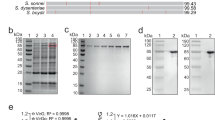
Abstract
Control of pandemic infection of human immunodeficiency virus type 1 (HIV-1) requires some means of developing mucosal immunity against HIV-1 because sexual transmission of the virus occurs mainly through the mucosal tissues. However, there is no evidence as yet that the secretory immunoglobulin A (IgA) antibody induced by immunization with antigens in experimental animals can neutralize HIV-1. We demonstrate here that oral immunization with a new macromolecular peptide antigen and cholera toxin (CT) induces a high titre (1:211) of gut-associated and secretory IgA antibody to HIV-1. Using three different neutralizing assays, we clearly demonstrate that this secretory IgA antibody is able to neutralize HIV-1 IIIB, HIV-1SF2 and HIV-1MN. Our new approach may prove to be important in the development of a mucosal vaccine that will provide protection of mucosal surfaces against HIV-1.
Similar content being viewed by others
Article PDF
References
Lehner, T. et al. Induction of mucosal and systemic immunity to a recombinant simian immunodeficiency viral protein. Science 258, 1365–1369 (1992).
Lehner, T. et al. T- and B-cell functions and epitope expression in nonhuman primates immunized with simian immunodeficiency virus antigen by the rectal route. Proc. natn. Acad. Sci. U.SA. 90, 8638–8642 (1993).
Lehner, T. et al. Mucosal model of genital immunization in male rhesus macaques with a recombinant simian immunodeficiency virus p27 antigen. J. Virol 68, 1624–1632 (1994).
Burnett, P.R., VanCott, T.C., Polonis, V.R., Redfield, R.R. & Birx, D.L., IgA-mediated neutralization of HIV type 1. J. Immun. 152, 4642–4648 (1994).
Okuda, K. et al. Strong immunogenicity of a multicomponent peptide vaccine developed with the branched lysine oligopeptide method for human immunodeficiency virus infection. J. Molec. Recognition 6, 101–109 (1993).
Okuda, K. et al. Strong synergistic effects of multicomponent vaccine for human immunodeficiency virus infection. J. clin. lab. Immun. 40, 97–113 (1993).
Rockson, R.J. et al. Optimizing oral vaccines: Induction of systemic and mucosal B-cell and antibody responses to tetanus toxoid by use of cholera toxin as an adjuvant. Infect Immun. 61, 4272–4279 (1993).
Xu-Amano, J. et al. Helper T cell subsets for immunoglobulin A responses: Oral immunization with tetanus toxoid and cholera toxin as adjuvant selectively induces Th2 cells in mucosa associated tissues. J. exp. Med. 178, 1309–1320 (1993).
Lycke, N. & Holmgren, J. Strong adjuvant properties of cholera toxin on gut mucosal immune responses to orally presented antigens. Immunology 59, 301–308 (1986).
Inami, S. et al. Serum antibody directed against synthetic peptides derived from HIV-1 protein sequence obtained from 26 Japanese HIV-1-infected individuals. AIDS 5, 1140–1141 (1991).
Okuda, K. et al. A serogic analysis and the amino acid sequence of the V3 region of human immunodeficiency virus from carriers in Bangkok. J. infect. Dis. 169, 227–228 (1994).
Ubolyam, S., Ruxrungtham SirMchayakul, S., Okuda, K. & Phanuphak, P. Evidence of three HIV-1 subtypes in subgroups of individuals in Thailand. Lancet 344, 485–486 (1994).
Bukawa, H. et al. Antibody responses raised against a conformational V3 loop peptide of HIV-1. Micorbiol Immun. (in the press).
McKeating, J.A., Cordell, J., Dean, C.J. & Balfe, P. Synergistic interaction between ligands binding to the CD4 binding site and V3 domain of human immunodeficiency virus type 1 gp120. Virology 191, 732–742 (1992).
Achour, A. et al. HGP-30, a synthetic analogue of human immunodeficiency virus (HIV) p17, is a target for cytotoxic lymphocytes in HIV-infected individuals. Proc. natn. Acad. Sci. U.S.A. 87, 7045–049 (1990).
Boucher, C.A.B. et al. Immune response and epitope mapping of a candidate HIV-1 pl7 vaccine HGP30. J. clin. Lab. Anal. 4, 43–47 (1990).
Nardelli, B. et al. A chemically defined synthetic vaccine model for HIV-1. J. Immun. 148, 914–920 (1992).
Defoort, J.-P., Nardelli, B., Huang, W., Ho, D.D. & Tam, J.P. Macromolecular assemblage in the design of a synthetic AIDS vaccine. Proc. natn. Acad. Sci. U.S.A. 89, 3879–3883 (1992).
Korn, A.H., Feairheller, S.H. & Filachione, E.M., Nature of the reagent. J. molec. Biol 65, 525–529 (1972).
Reichlin, M. Use of glutaraldehyde as a coupling agent for proteins and peptides. Meth. Enzym. 70, 159–165 (1980).
Staats, H.F. et al. Mucosal immunity to infection with implications for vaccine development. Curr. Opin. Immun. 6, 572–583 (1994).
Bourguin, I., Chardes, T. & Bout, D. Oral immunization with Toxoplasma gondiiantigens in association with cholera toxin induces enhanced protective and cell-mediated immunity in C57/BL/6 mice. Infect. Immun. 61, 2082–2088 (1993).
Nara, P.L. et al. Simple, rapid, quantitative, syncytium-forming microassay for the detection of human immunodeficiency virus neutralizing antibody. AIDS Res. hum. Retrovir. 3, 283–302 (1987).
Putney, S.D. et al. HTLV-III/LAV-neutralizing antibodies to an E. coli-produced fragment of the virus envelope. Science 234, 1392–1395 (1986).
Pauwels, R. et al. Rapid and automated tetrazolium-based colorimetric assay for the detection of anti-HIV compounds. J. virol. Meth. 20, 309–321 (1988).
Nakashima, H. Development of anti-human immunodeficiency virus (HIV) agents. Bull. Yamaguchi Med. Sch. 37, 169–180 (1990).
Lai, P.K. et al. Modification of human immunodeficiency viral replication by pine cone extracts. AIDS Res. hum. Retrovir. 6, 205–217 (1990).
Goudsmit, J. et al. Expression of human immunodeficiency virus antigen (HIV-Ag) in serum and cerebrospinal fluid during acute and chronic infection. Lancet 2, 177–180(1986).
Okuda, K., Minami, M., Ju, S.-T. & Dorf, M.E. Functional association of idio-typic and I-J determinants on the antigen receptor of suppressor T cells. Proc. natn. Acad. Sci. U.SA. 78, 4557–4561 (1981).
Lehner, T. et al. A comparison of the immune responses following oral, vaginal, or rectal route of immunization with SIV antigens in nonhuman primates. Vaccine Res. 1, 319–330 (1992).
Kaartinen, M., Imir, T., Klockars, M., Sandholm, M. & Mäkelä, O. IgA in blood and thoracic duct lymph: Concentration and degree of polymerization. Scand. J. Immun. 7, 229–232 (1978).
Olson, C.A., Williams, L.C., McLaughlin-Taylor, E. & McMillan, M. Creation of H-2 class I epitopes using synthetic peptides: Recognition by alloreactive cytotoxic T lymphocytes. Proc. natn. Acad. Sci. U.SA. 86, 1031–1035 (1989).
deVos, T. & Dick, T.A. A rapid method to determine the isotype and specificity of coproantibodies in mice infected with Trichinella or fed cholera toxin. J. immunoh Meth. 141, 285–288 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bukawa, H., Sekigawa, KI., Hamajima, K. et al. Neutralization of HIV-1 by secretory IgA induced by oral immunization with a new macromolecular multicomponent peptide vaccine candidate. Nat Med 1, 681–685 (1995). https://doi.org/10.1038/nm0795-681
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0795-681
This article is cited by
-
Enhanced antitumor efficacy by combination treatment with a human umbilical vein endothelial cell vaccine and a tumor cell lysate-based vaccine
Tumor Biology (2013)
-
Prime-boost vaccination with plasmid DNA and a chimeric adenovirus type 5 vector with type 35 fiber induces protective immunity against HIV
Gene Therapy (2005)
-
8 Br-cAMP enhances both humoral and cell-mediated immune responses induced by an HIV-1 DNA vaccine
Gene Therapy (2000)
-
HIV-specific mucosal and cellular immunity in HIV-seronegative partners of HIV-seropositive individuals
Nature Medicine (1997)
-
Dimers are a girl's best friend
Nature Medicine (1997)