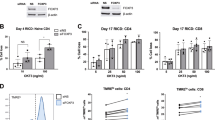
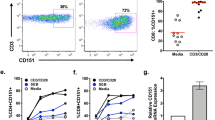
Abstract
The molecular properties that characterize CD4+CD25+ regulatory T cells (TR cells) remain elusive. Absence of the transcription factor Scurfin (also known as forkhead box P3 and encoded by Foxp3) causes a rapidly fatal lymphoproliferative disease, similar to that seen in mice lacking cytolytic T lymphocyte–associated antigen 4 (CTLA-4). Here we show that Foxp3 is highly expressed by TR cells and is associated with TR cell activity and phenotype. Scurfin-deficient mice lack TR cells, whereas mice that overexpress Foxp3 possess more TR cells. In Foxp3-overexpressing mice, both CD4+CD25− and CD4−CD8+ T cells show suppressive activity and CD4+CD25− cells express glucocorticoid-induced tumor-necrosis factor receptor–related (GITR) protein. The forced expression of Foxp3 also delays disease in CTLA-4−/− mice, indicating that the Scurfin and CTLA-4 pathways may intersect and providing further insight into the TR cell lineage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Shevach, E.M. Regulatory T cells in autoimmmunity. Annu. Rev. Immunol. 18, 423–449 (2000).
Roncarolo, M.G. & Levings, M.K. The role of different subsets of T regulatory cells in controlling autoimmunity. Curr. Opin. Immunol. 12, 676–683 (2000).
Sakaguchi, S. Regulatory T cells: key controllers of immunologic self-tolerance. Cell 101, 455–458 (2000).
McHugh, R.S. et al. CD4+CD25+ immunoregulatory T cells: gene expression analysis reveals a functional role for the glucocorticoid-induced TNF receptor. Immunity 16, 311–323 (2002).
Shimizu, J., Yamazaki, S., Takahashi, T., Ishida, Y. & Sakaguchi, S. Stimulation of CD25+CD4+ regulatory T cells through GITR breaks immunological self-tolerance. Nat. Immunol. 3, 135–142 (2002).
Takahashi, T. et al. Immunologic self-tolerance maintained by CD25+CD4+ regulatory T cells constitutively expressing cytotoxic T lymphocyte-associated antigen 4. J. Exp. Med. 192, 303–310 (2000).
Nakamura, K., Kitani, A. & Strober, W. Cell contact–dependent immunosuppression by CD4+CD25+ regulatory T cells is mediated by cell surface-bound transforming growth factor β. J. Exp. Med. 194, 629–644 (2001).
Asano, M., Toda, M., Sakaguchi, N. & Sakaguchi, S. Autoimmune disease as a consequence of developmental abnormality of a T cell subpopulation. J. Exp. Med. 184, 387–396 (1996).
Shull, M.M. et al. Targeted disruption of the mouse transforming growth factor-β1 gene results in multifocal inflammatory disease. Nature 359, 693–699 (1992).
Kulkarni, A.B. et al. Transforming growth factor β1 null mutation in mice causes excessive inflammatory response and early death. Proc. Natl. Acad. Sci. USA 90, 770–774 (1993).
Tivol, E.A. et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity 3, 541–547 (1995).
Waterhouse, P. et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science 270, 985–988 (1995).
Piccirillo, C.A. et al. CD4+CD25+ Regulatory T cells can mediate suppressor function in the absence of transforming growth factor β1 production and responsiveness. J. Exp. Med. 196, 237–246 (2002).
Lyon, M.F., Peters, J., Glenister, P.H., Ball, S. & Wright, E. The scurfy mouse mutant has previously unrecognized hematological abnormalities and resembles Wiskott-Aldrich syndrome. Proc. Natl. Acad. Sci. USA 87, 2433–2437 (1990).
Godfrey, V.L., Wilkinson, J.E. & Russell, L.B. X-linked lymphoreticular disease in the scurfy (sf) mutant mouse. Am. J. Pathol. 138, 1379–1387 (1991).
Godfrey, V., Wilkinson, J., Rinchik, E. & Russell, L. Fatal lymphoreticular disease in the scurfy (sf) mouse requires T cells that mature in a sf thymic environment: potential model for thymic education. Proc. Natl. Acad. Sci. USA 88, 5528–5532 (1991).
Godfrey, V.L., Rouse, B.T. & Wilkinson, J.E. Transplantation of T cell-mediated, lymphoreticular disease from the scurfy (sf) mouse. Am. J. Pathol. 145, 281–286 (1994).
Kanangat, S. et al. Disease in the scurfy (sf) mouse is associated with overexpression of cytokine genes. Eur. J. Immunol. 26, 161–165 (1996).
Blair, P.J. et al. CD4+CD8− T cells are the effector cells in disease pathogenesis in the scurfy (sf) mouse. J. Immunol. 153, 3764–3774 (1994).
Brunkow, M.E. et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat. Genet. 27, 68–73 (2001).
Chatila, T.A. et al. JM2, encoding a fork head-related protein, is mutated in X-linked autoimmunity-allergic disregulation syndrome. J. Clin. Invest. 106, R75–R81 (2000).
Wildin, R.S. et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat. Genet. 27, 18–20 (2001).
Bennett, C.L. et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 27, 20–21 (2001).
Khattri, R. et al. The amount of scurfin protein determines peripheral T cell number and responsiveness. J. Immunol. 167, 6312–6320 (2001).
Clark, L.B. et al. Cellular and molecular characterization of the scurfy mouse mutant. J. Immunol. 162, 2546–2554 (1999).
Chambers, C.A., Cado, D., Truong, T. & Allison, J.P. Thymocyte development is normal in CTLA-4-deficient mice. Proc. Natl. Acad. Sci. USA 94, 9296–9301 (1997).
Chambers, C.A. et al. The lymphoproliferative defect in CTLA-4-deficient mice is ameliorated by an inhibitory NK cell receptor. Blood 99, 4509–4516 (2002).
Schubert, L.A., Jeffery, E., Zhang, Y., Ramsdell, F. & Ziegler, S.F. Scurfin (foxp3) acts as a repressor of transcription and regulates T cell activation. J. Biol. Chem. 276, 37672–37679 (2001).
Itoh, M. et al. Thymus and autoimmunity: production of CD25+CD4+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologic self-tolerance. J. Immunol. 162, 5317–5326 (1999).
Bensinger, S.J., Bandeira, A., Jordan, M.S., Caton, A.J. & Laufer, T.M. Major histocompatibility complex class II-positive cortical epithelium mediates the selection of CD4+25+ immunoregulatory T cells. J. Exp. Med. 194, 427–438 (2001).
Jordan, M.S. et al. Thymic selection of CD4+CD25+ regulatory T cells induced by an agonist self-peptide. Nat. Immunol. 2, 301–306 (2001).
Gavin, M.A., Clarke, S.R., Negrou, E., Gallegos, A. & Rudensky, A. Homeostasis and anergy of CD4+CD25+ suppressor T cells in vivo. Nat. Immunol. 3, 33–41 (2002).
Hong, N.A. et al. A targeted mutation at the T-cell receptor α/δ locus impairs T-cell development and reveals the presence of the nearby antiapoptosis gene Dad1. Mol. Cell. Biol. 17, 2151–2157 (1997).
Acknowledgements
We thank D. Walker for cell sorting and analysis; S. Rudensky, M. Gavin and J. Fontenot for discussions; and J. Allison for the CTLA-4−/− mice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors' primary employment is by a for-profit corporation (Celltech Group).
Supplementary information
Supplementary Fig. 1.
CD4+CD25+ TR cells from OT-II Foxp3 transgenic mice express more Foxp3 and show an increased regulatory activity. (a) Foxp3 expression was determined in cDNA samples from CD4+CD25+ isolated either from OT-II or OT-II Foxp3 transgenic mice using a real-time RT-PCR with Dad1 as an endogenous reference gene. (b) 2.5 x 104 OT-II CD4+CD25- T cells were stimulated either with different concentrations of Ova323-339 peptide or anti-CD3 (1µg/ml) and equal numbers of mitomycin C treated Thy-1- APC. CD4+CD25+ T cells from OT-II (open symbols) or OT-II Foxp3 TG (filled symbols, dashed lines) animals were added at various ratios. The cells were cultured for 72 h and pulsed with [3H]thymidine for final 8 h of the culture. Data is expressed as percentage inhibition calculated as 100 x [1 - (counts with suppressor/counts without suppressor)]. Data is mean of triplicates and is representative of three independent experiments. The proliferation of responders in OT-II CD4+CD25+ assay were 43,661 cpm at 0.25 µM peptide, 38,790 cpm at 0.125 µM peptide and 59,486 cpm with anti-CD3; in OT-II Foxp3 transgenic CD4+CD25+ assay were 36,706 cpm at 0.25 µM peptide, 36,496 cpm at 0.125 µM peptide and 54,800 cpm with anti-CD3. (PDF 13 kb)
Supplementary Fig. 2.
IL-10 mRNA levels do not correlate with Foxp3 mRNA expression levels. IL-10 mRNA was determined in cDNA samples using a real-time RT-PCR method in which Dad1 served as an endogenous reference gene. Samples are from freshly isolated cell or 72 h preactivated subsets from wild type (WT), scurfy (SF) or Foxp3 transgenic (TG) mice. Normalized IL-10 values were derived from the ratio of IL-10 expression to Dad1 expression. Data is representative of analysis with 3 independent experiments. (PDF 7 kb)
Supplementary Fig. 3.
Scurfy CD4+ T cells are inhibited by TR cells but not by TGF-ß. (a) 5 x 104 wild-type (WT) or scurfy (SF) CD4+ T cells were stimulated with anti-CD3 (1µg/ml) and equal numbers of mitomycin C treated Thy-1- APC. CD4+CD25+ TR cells were added at various ratios to the assay. Specific responder:suppressor ratios are defined in the chart legend. The cells were cultured for 72 h and pulsed with [3H]thymidine for final 8 h of the culture. (b) Wild-type (WT) or scurfy (SF) CD4+ T cells were stimulated either with immobilized anti-CD3 (4 µg/ml) or anti-CD3 (0.25 µg/ml) and anti-CD28 (1 µg/ml). TGF-ß (2.5 ng/ml) was added at the beginning of the assay. The cells were cultured for 48 h and pulsed with [3H]thymidine for final 8 h of the culture. Data in is mean of triplicates and is representative of at least three independent experiments. (PDF 23 kb)
Supplementary Fig. 4.
CD4+ T cells from Ctla-4-/-Foxp3 transgenic mice express increased CD25. Lymph node CD4+ T cells from wild-type (top panel), Foxp3 transgenic (middle panel) and Ctla-4-/-Foxp3 transgenic (bottom panel) mice were analyzed for the expression of CD25. Numbers represent percent positive above an isotype control antibody. Analyses are from samples gated for CD4+ cells and represent individual mice and are representative of four animals examined from each group. (PDF 11 kb)
Rights and permissions
About this article
Cite this article
Khattri, R., Cox, T., Yasayko, SA. et al. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat Immunol 4, 337–342 (2003). https://doi.org/10.1038/ni909
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni909
This article is cited by
-
A promising target for breast cancer: B7-H3
BMC Cancer (2024)
-
T cell effects and mechanisms in immunotherapy of head and neck tumors
Cell Communication and Signaling (2023)
-
Exploiting E3 ubiquitin ligases to reeducate the tumor microenvironment for cancer therapy
Experimental Hematology & Oncology (2023)
-
Regulatory T cells in autoimmune kidney diseases and transplantation
Nature Reviews Nephrology (2023)
-
Murine regulatory T cells utilize granzyme B to promote tumor metastasis
Cancer Immunology, Immunotherapy (2023)