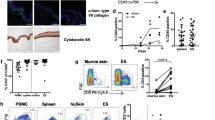
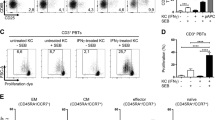
Abstract
It remains largely unclear how antigen-presenting cells (APCs) encounter effector or memory T cells efficiently in the periphery. Here we used a mouse contact hypersensitivity (CHS) model to show that upon epicutaneous antigen challenge, dendritic cells (DCs) formed clusters with effector T cells in dermal perivascular areas to promote in situ proliferation and activation of skin T cells in a manner dependent on antigen and the integrin LFA-1. We found that DCs accumulated in perivascular areas and that DC clustering was abrogated by depletion of macrophages. Treatment with interleukin 1α (IL-1α) induced production of the chemokine CXCL2 by dermal macrophages, and DC clustering was suppressed by blockade of either the receptor for IL-1 (IL-1R) or the receptor for CXCL2 (CXCR2). Our findings suggest that the dermal leukocyte cluster is an essential structure for elicitating acquired cutaneous immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others

Accession codes
References
von Andrian, U.H. & Mempel, T.R. Homing and cellular traffic in lymph nodes. Nat. Rev. Immunol. 3, 867–878 (2003).
Clark, R.A. et al. The vast majority of CLA+ T cells are resident in normal skin. J. Immunol. 176, 4431–4439 (2006).
Wang, L. et al. Langerin expressing cells promote skin immune responses under defined conditions. J. Immunol. 180, 4722–4727 (2008).
Tuckermann, J.P. et al. Macrophages and neutrophils are the targets for immune suppression by glucocorticoids in contact allergy. J. Clin. Invest. 117, 1381–1390 (2007).
Sims, J.E. & Smith, D.E. The IL-1 family: regulators of immunity. Nat. Rev. Immunol. 10, 89–102 (2010).
Murphy, J.E., Robert, C. & Kupper, T.S. Interleukin-1 and cutaneous inflammation: a crucial link between innate and acquired immunity. J. Invest. Dermatol. 114, 602–608 (2000).
Nakae, S. et al. IL-1-induced tumor necrosis factor-α elicits inflammatory cell infiltration in the skin by inducing IFN-γ-inducible protein 10 in the elicitation phase of the contact hypersensitivity response. Int. Immunol. 15, 251–260 (2003).
Thyssen, J.P., Linneberg, A., Menne, T., Nielsen, N.H. & Johansen, J.D. Contact allergy to allergens of the TRUE-test (panels 1 and 2) has decreased modestly in the general population. Br. J. Dermatol. 161, 1124–1129 (2009).
Ng, L.G. et al. Migratory dermal dendritic cells act as rapid sensors of protozoan parasites. Plos Pathog 4, e1000222 (2008).
Springer, T.A. & Dustin, M.L. Integrin inside-out signaling and the immunological synapse. Curr. Opin. Cell Biol. 24, 107–115 (2012).
Egawa, G. et al. In vivo imaging of T-cell motility in the elicitation phase of contact hypersensitivity using two-photon microscopy. J. Invest. Dermatol. 131, 977–979 (2011).
Miyawaki, S. et al. A new mutation, aly, that induces a generalized lack of lymph nodes accompanied by immunodeficiency in mice. Eur. J. Immunol. 24, 429–434 (1994).
Sawaguchi, M. et al. Role of mast cells and basophils in IgE responses and in allergic airway hyperresponsiveness. J. Immunol. 188, 1809–1818 (2012).
Otsuka, A. et al. Requirement of interaction between mast cells and skin dendritic cells to establish contact hypersensitivity. PLoS ONE 6, e25538 (2011).
Enk, A.H. & Katz, S.I. Early molecular events in the induction phase of contact sensitivity. Proc. Natl. Acad. Sci. USA 89, 1398–1402 (1992).
Weisser, S.B., McLarren, K.W., Kuroda, E. & Sly, L.M. Generation and characterization of murine alternatively activated macrophages. Methods Mol. Biol. 946, 225–239 (2013).
Liao, L. et al. CXCR2 blockade reduces radical formation in hyperoxia-exposed newborn rat lung. Pediatr. Res. 60, 299–303 (2006).
Honda, T., Egawa, G., Grabbe, S. & Kabashima, K. Update of immune events in the murine contact hypersensitivity model: toward the understanding of allergic contact dermatitis. J. Invest. Dermatol. 133, 303–315 (2013).
Kaplan, D.H., Igyarto, B.Z. & Gaspari, A.A. Early immune events in the induction of allergic contact dermatitis. Nat. Rev. Immunol. 12, 114–124 (2012).
Lee, R.T. et al. Mechanical deformation promotes secretion of IL-1 alpha and IL-1 receptor antagonist. J. Immunol. 159, 5084–5088 (1997).
Sackstein, R., Falanga, V., Streilein, J.W. & Chin, Y.H. Lymphocyte adhesion to psoriatic dermal endothelium is mediated by a tissue-specific receptor/ligand interaction. J. Invest. Dermatol. 91, 423–428 (1988).
Kish, D.D., Gorbachev, A.V. & Fairchild, R.L. IL-1 receptor signaling is required at multiple stages of sensitization and elicitation of the contact hypersensitivity response. J. Immunol. 188, 1761–1771 (2012).
Kondo, S. et al. Interleukin-1 receptor antagonist suppresses contact hypersensitivity. J. Invest. Dermatol. 105, 334–338 (1995).
Cattani, F. et al. The role of CXCR2 activity in the contact hypersensitivity response in mice. Eur. Cytokine Netw. 17, 42–48 (2006).
Brandtzaeg, P., Kiyono, H., Pabst, R. & Russell, M.W. Terminology: nomenclature of mucosa-associated lymphoid tissue. Mucosal Immunol. 1, 31–37 (2008).
Streilein, J.W. Skin-associated lymphoid tissues (SALT): origins and functions. J. Invest. Dermatol. 80 (suppl.), 12s–16s (1983).
Egawa, G. & Kabashima, K. Skin as a peripheral lymphoid organ: revisiting the concept of skin-associated lymphoid tissues. J. Invest. Dermatol. 131, 2178–2185 (2011).
Moyron-Quiroz, J.E. et al. Role of inducible bronchus associated lymphoid tissue (iBALT) in respiratory immunity. Nat. Med. 10, 927–934 (2004).
Kissenpfennig, A. et al. Dynamics and function of Langerhans cells in vivo: dermal dendritic cells colonize lymph node areas distinct from slower migrating Langerhans cells. Immunity 22, 643–654 (2005).
Jung, S. et al. In vivo depletion of CD11c+ dendritic cells abrogates priming of CD8+ T cells by exogenous cell-associated antigens. Immunity 17, 211–220 (2002).
Lindquist, R.L. et al. Visualizing dendritic cell networks in vivo. Nat. Immunol. 5, 1243–1250 (2004).
Miyake, Y. et al. Protective role of macrophages in noninflammatory lung injury caused by selective ablation of alveolar epithelial type II Cells. J. Immunol. 178, 5001–5009 (2007).
Hao, Z. & Rajewsky, K. Homeostasis of peripheral B cells in the absence of B cell influx from the bone marrow. J. Exp. Med. 194, 1151–1164 (2001).
Horai, R. et al. Production of mice deficient in genes for interleukin (IL)-1α, IL-1β, IL-1α/β, and IL-1 receptor antagonist shows that IL-1β is crucial in turpentine-induced fever development and glucocorticoid secretion. J. Exp. Med. 187, 1463–1475 (1998).
Coban, C. et al. Immunogenicity of whole-parasite vaccines against Plasmodium falciparum involves malarial hemozoin and host TLR9. Cell Host Microbe 7, 50–61 (2010).
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Koedel, U. et al. Role of caspase-1 in experimental pneumococcal meningitis: evidence from pharmacologic caspase inhibition and caspase-1-deficient mice. Ann. Neurol. 51, 319–329 (2002).
Tomura, M. et al. Activated regulatory T cells are the major T cell type emigrating from the skin during a cutaneous immune response in mice. J. Clin. Invest. 120, 883–893 (2010).
Acknowledgements
We thank H. Yagita (Juntendo University) for the KBA neutralizing antibody to LFA-1; P. Bergstresser and J. Cyster for critical reading of our manuscript. Supported by grants-in-aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Contributions
Y.N., G.E. and K.K. designed this study and wrote the manuscript; Y.N., G.E., S. Nakamizo, S.O., S.H., N.K., A.O., A.K., T. Honda and S. Nakajima performed the experiments and analyzed data; S.T. and Y.S. did experiments related to microarray analysis; K.J.I., H.T., H.Y., Y.I., M.K. and L.g.N. developed experimental reagents and gene-targeted mice; J.F. and E.G.-Y. did experiments related to immunohistochemistry of human samples; T.O., T. Hashimoto, Y.M. and K.K. directed the project and edited the manuscript; and all authors reviewed and discussed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 DC motility in the elicitation phase of CHS.
(a) Superimposed 30-min tracks of 30 randomly selected dermal DCs in the x–y plane, setting the starting coordinates to the origin. Tracks of a steady state, 6, 12, and 24 h after the elicitation with DNFB are shown. (b and c) Velocity (b) and displacement (c) of dDCs at each time point (n=30). Each bar represents the mean + SD. *, P < 0.05.
Supplementary Figure 2 Subset-specific depletion of cutaneous DCs.
(a) A schematic representation of our strategy to generate subset-specific cutaneous DC depletion models. To deplete all cutaneous DC subsets, Langerin-DTR mice were transferred with BM cells from CD11c-DTR mice, and DT was injected. To selectively deplete LCs, Langerin-DTR mice were transferred with BM cells from C57BL/6 mice, and DT was injected. To selectively deplete dDCs, C57BL/6 mice were transferred with BM cells from CD11c-DTR mice, and DT was injected. BMT; BM transplantation. (b) FACS plots of each group of mice after DT treatment. In dermis, the percentages in CD45+ cells were indicated. (c) Histological findings of the ear skin after CHS. HE staining of the ears of mice 24 h after challenge with DNFB. Mice were pretreated with or without sensitization, depleted of LCs and/or dDCs, and challenged with DFNB. Scale bar = 100 μm. (d) CHS response was induced on the ear skin, and skin-infiltrating cells were stained and analyzed with TCR beta, CD44, and CD62L antibodies by flow cytometry.
Supplementary Figure 3 dDC clusters are formed in response to various stimuli.
(a) The scoring criteria for DC clusters by numbers and diameters of clusters. (b) DC (green) cluster formation 24 h after topical application without (NT) or with acetone, olive oil, 3% TNCB, 2% DNTB, 0.3% DNFB, or Mycobacterium bovis BCG-inoculation (n=4, each). (c) Scores of DC cluster numbers of each group 24 h after each stimuli. (d) DC (green) cluster formation 24 h after topical application without (NT) or with 0.5% DNFB on the back skin and footpad. Scale bar = 100 μm. (e) Mobility of DCs and T cells of the cluster by treatment with anti-LFA-1 treatment. Anti-LFA-1 neutralizing antibody, KBA, was injected intravenously 14 h after elicitation. T cell (red) clustering was dissolved but DC (green) clustering persisted 10 h after KBA-treatment. Scale bar = 100 μm. (f) Score of DC cluster number 24 h after DNFB application with KBA (red) or control IgG (black) treatment (n=5, each).
Supplementary Figure 4 Analysis of M1 and M2 macrophage markers.
(a) TRITC-conjugated dextran was injected and dermal suspension was prepared 24 h later. CD45+ and TRITC+ cells were further analyzed with CD11b and F4/80 antibodies by flow cytometry. (b) M1 macrophage markers, such as TNF-a, Nos2, and IL-12a, and M2 macrophage markers, such as arginase (Arg)-1, Retnla, and Chi313, were examined in BM-derived M1 and M2 macrophages. Each bar represents the mean + SD (n=3). A.U., arbitrary units. *, P < 0.05. ***P < 0.0001.
Supplementary Figure 5 Neutrophils are not essential for CXCL2 expression in DNFB-painted skin.
(a) Relative amount of Il1r1 24 h after with or without DNFB-sensitization (n=5). (b) Relative amount of Cxcl2 in DNFB-painted skin in 1A8- or control IgG-treated mice (n=5, each). (c) FACS plot of DNFB-painted skin prepared from 1A8- or control IgG-treated mice. CD11b+ Gr-1+ neutrophils were significantly depleted with 1A8-treatment. (d) Relative amount of Il1r1 from dermal macrophages cultured with or without IL-1α (n=4, each). (e) RT-PCR analysis of chemokine receptor mRNA expression in BM-derived DCs.
Supplementary Figure 6 A schema of immunological events in CHS response.
Sensitization phase. Epidermal contact with antigens triggers release of IL-1 in the skin, which activates macrophages that subsequently attract dDCs to perivascular area via CXCL2 to form clusters. In the absence of antigen-specific effector/memory T cells, DC clustering is a transient event, and hapten-carrying DCs migrate into draining LNs to establish sensitization. (b) Elicitation phase. In the presence of antigen-specific effector/memory T cells, the antigen is recognized efficiently in the DC clusters by antigen-specific effector T cells to form clusters, and inflammation is induced promptly via activation and proliferation of antigen-specific effector T cells.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1–2 (PDF 2752 kb)
Leukocyte cluster formation in elicitation phase of DNFB-induced CHS response
CMTMR-labeled DNFB-sensitized T cells were transferred into CD11c-YFP mice and then challenged with DNFB to the ear. CD11c+ dermal DCs (green) and T cells (red) formed clusters approximately 6 h after hapten application. The images were taken every 7 min for 24 h. (WMV 41200 kb)
High magnification view of leukocyte cluster in the elicitation phase of CHS
CMTMR-labeled DNFB-sensitized T cells were transferred into CD11c-YFP mice and then challenged with DNFB to the ear. Sixteen hours later, the established DC–T cell cluster was observed in high magnification view for 2 h every 1 min. In this leukocyte cluster, some of T cells (red) interacted with dermal DCs (green) for more than 2 h. The pale yellow debris are melanin granules. Fragmented red and green debris seems to be indicative of dead T cells and DCs engulfed by macrophages, respectively. (WMV 28045 kb)
T cell division in the skin
CMTMR-labeled DNFB-sensitized T cells divided in DNFB-challenged site. The mean frequency of T cell division was 1.67±1.81 /h/mm2 (calculated from 5 movies which recorded more than an hour). (WMV 3003 kb)
Macrophages attracted dermal DCs
TRITC-conjugated dextran was intravenously injected to DNFB-sensitized CD11c-YFP mice to label skin macrophages. The next day, ear skin was challenged with DNFB and examined using two-photon microscopy. In this representative movie, a dermal DC (green) migrated toward TRITC-positive macrophages (red). (WMV 9892 kb)
Rights and permissions
About this article
Cite this article
Natsuaki, Y., Egawa, G., Nakamizo, S. et al. Perivascular leukocyte clusters are essential for efficient activation of effector T cells in the skin. Nat Immunol 15, 1064–1069 (2014). https://doi.org/10.1038/ni.2992
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2992