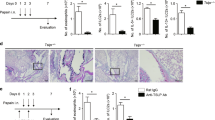
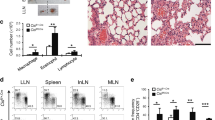
Abstract
Type 2 helper T cells (TH2) are critically involved in allergies and asthma. Here we demonstrate that extracellular matrix protein-1 (ECM1) is highly and selectively expressed in TH2 cells. ECM1 deficiency caused impaired TH2 responses and reduced allergic airway inflammation in vivo. Functional analysis demonstrated that although the TH2 polarization of ECM1-deficient cells was unimpaired, these cells had a defect in migration and were retained in peripheral lymphoid organs. This was associated with reduced expression of KLF2 and S1P1. We also found that ECM1 could directly bind the interleukin-2 (IL-2) receptor to inhibit IL-2 signaling and activate S1P1 expression. Our data identify a previously unknown function of ECM1 in regulating TH2 cell migration through control of KLF2 and S1P1 expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Scharton, T.M. & Scott, P. Natural killer cells are a source of interferon gamma that drives differentiation of CD4+ T cell subsets and induces early resistance to Leishmania major in mice. J. Exp. Med. 178, 567–577 (1993).
Mosmann, T.R. & Coffman, R.L. TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties. Annu. Rev. Immunol. 7, 145–173 (1989).
Weaver, C.T., Harrington, L.E., Mangan, P.R., Gavrieli, M. & Murphy, K.M. Th17: an effector CD4 T cell lineage with regulatory T cell ties. Immunity 24, 677–688 (2006).
Zhu, J. & Paul, W.E. CD4 T cells: fates, functions, and faults. Blood 112, 1557–1569 (2008).
Dong, C. TH17 cells in development: an updated view of their molecular identity and genetic programming. Nature Rev. Immunol. 8, 337–348 (2008).
Murphy, K.M. & Reiner, S.L. The lineage decisions of helper T cells. Nature Rev. Immunol. 2, 933–944 (2002).
Rosen, H. et al. Modulating tone: the overture of S1P receptor immunotherapeutics. Immunol. Rev. 223, 221–235 (2008).
Schwab, S.R. & Cyster, J.G. Finding a way out: lymphocyte egress from lymphoid organs. Nat. Immunol. 8, 1295–1301 (2007).
Rivera, J., Proia, R.L. & Olivera, A. The alliance of sphingosine-1-phosphate and its receptors in immunity. Nature Rev. Immunol. 8, 753–763 (2008).
Graeler, M. & Goetzl, E.J. Activation-regulated expression and chemotactic function of sphingosine 1-phosphate receptors in mouse splenic T cells. FASEB J. 16, 1874–1878 (2002).
Mandala, S. et al. Alteration of lymphocyte trafficking by sphingosine-1-phosphate receptor agonists. Science 296, 346–349 (2002).
Brinkmann, V. et al. The immune modulator FTY720 targets sphingosine 1-phosphate receptors. J. Biol. Chem. 277, 21453–21457 (2002).
Matloubian, M. et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 427, 355–360 (2004).
Allende, M.L., Dreier, J.L., Mandala, S. & Proia, R.L. Expression of the sphingosine 1-phosphate receptor, S1P1, on T-cells controls thymic emigration. J. Biol. Chem. 279, 15396–15401 (2004).
Chi, H. & Flavell, R.A. Cutting edge: regulation of T cell trafficking and primary immune responses by sphingosine 1-phosphate receptor 1. J. Immunol. 174, 2485–2488 (2005).
Gräler, M.H., Huang, M.C., Watson, S. & Goetzl, E.J. Immunological effects of transgenic constitutive expression of the type 1 sphingosine 1-phosphate receptor by mouse lymphocytes. J. Immunol. 174, 1997–2003 (2005).
Cyster, J.G. Chemokines, sphingosine-1-phosphate, and cell migration in secondary lymphoid organs. Annu. Rev. Immunol. 23, 127–159 (2005).
Bhalerao, J., Tylzanowski, P., Filie, J.D., Kozak, C.A. & Merregaert, J. Molecular cloning, characterization, and genetic mapping of the cDNA coding for a novel secretory protein of mouse. Demonstration of alternative splicing in skin and cartilage. J. Biol. Chem. 270, 16385–16394 (1995).
Mongiat, M. et al. Perlecan protein core interacts with extracellular matrix protein 1 (ECM1), a glycoprotein involved in bone formation and angiogenesis. J. Biol. Chem. 278, 17491–17499 (2003).
Hamada, T. Lipoid proteinosis. Clin. Exp. Dermatol. 27, 624–629 (2002).
Han, Z. et al. Extracellular matrix protein 1 (ECM1) has angiogenic properties and is expressed by breast tumor cells. FASEB J. 15, 988–994 (2001).
Wang, L. et al. Extracellular matrix protein 1 (ECM1) is over-expressed in malignant epithelial tumors. Cancer Lett. 200, 57–67 (2003).
Liu, Z. et al. Dec2 promotes Th2 cell differentiation by enhancing IL-2R signaling. J. Immunol. 183, 6320–6329 (2009).
Luger, D. et al. Either a Th17 or a Th1 effector response can drive autoimmunity: conditions of disease induction affect dominant effector category. J. Exp. Med. 205, 799–810 (2008).
Carlson, C.M. et al. Kruppel-like factor 2 regulates thymocyte and T-cell migration. Nature 442, 299–302 (2006).
Kerdiles, Y.M. et al. Foxo1 links homing and survival of naive T cells by regulating L-selectin, CCR7 and interleukin 7 receptor. Nat. Immunol. 10, 176–184 (2009).
Sinclair, L.V. et al. Phosphatidylinositol-3-OH kinase and nutrient-sensing mTOR pathways control T lymphocyte trafficking. Nat. Immunol. 9, 513–521 (2008).
Leonard, W.J. & O′Shea, J.J. Jaks and STATs: biological implications. Annu. Rev. Immunol. 16, 293–322 (1998).
Miller, J.D., Stevens, E.T., Smith, D.R., Wight, T.N. & Wrenshall, L.E. Perlecan: a major IL-2-binding proteoglycan in murine spleen. Immunol. Cell Biol. 86, 192–199 (2008).
Kuznetsova, S.A. & Roberts, D.D. Functional regulation of T lymphocytes by modulatory extracellular matrix proteins. Int. J. Biochem. Cell Biol. 36, 1126–1134 (2004).
Medoff, B.D., Thomas, S.Y. & Luster, A.D. T cell trafficking in allergic asthma: the ins and outs. Annu. Rev. Immunol. 26, 205–232 (2008).
Dang, X., Raffler, N.A. & Ley, K. Transcriptional regulation of mouse L-selectin. Biochim. Biophys. Acta 1789, 146–152 (2009).
Le Gros, G., Ben-Sasson, S.Z., Seder, R., Finkelman, F.D. & Paul, W.E. Generation of interleukin 4 (IL-4)-producing cells in vivo and in vitro: IL-2 and IL-4 are required for in vitro generation of IL-4-producing cells. J. Exp. Med. 172, 921–929 (1990).
Hou, W. et al. Pertussis toxin enhances Th1 responses by stimulation of dendritic cells. J. Immunol. 170, 1728–1736 (2003).
Wang, Y. et al. Trichosanthin functions as Th2-type adjuvant in induction of allergic airway inflammation. Cell Res. 19, 962–972 (2009).
Wu, X. et al. Novel function of IFN-gamma: negative regulation of dendritic cell migration and T cell priming. J. Immunol. 177, 934–943 (2006).
Acknowledgements
We thank R. Caspi, Y. He, Y. Zhuang, H. Gu, W. Zhang, Y. Liu and D. Li for helpful comments about this paper, S. Skinner for reviewing the manuscript, and X. Zhu for help with the experiments. This work was supported by grants from the National Natural Science Foundation of China (30623003, 31030029, 30721065, 30801011, 90713044, 30870126, 30950002), a Chinese Academy of Sciences project (KSCX2-YW-R-161, KSCX2-YW-R-169), the Technology Commission of Shanghai Municipality (08DZ2291703, 088014199, 08431903004), the National 973 key project (2007CB512404), the National 863 project (2006AA02A247), an EU project (SP5B-CT-2006-044161), a China National Science and Technology Major Project (2008ZX10002-014, 2008ZX10004-002, 2009ZX10004-105, 2009ZX10004-016), the China National Ministry of Science and Technology (20072714), the Shanghai Pasteur Health Research Foundation (SPHRF2008001, SPHRF2009001), the E-institutes of the Shanghai Universities Immunology Division and the Li Kha Shing Foundation.
Author information
Authors and Affiliations
Contributions
Z. Li designed (with help from Z. Liu) and performed mouse experiments and in vitro cell culture experiments, analyzed data and wrote the manuscript; Y. Zhang did all western blotting and real-time PCR experiments and helped in mouse experiments and analyzing data; X.W., K.M. and Y. Zheng helped in mouse experiments; Z.T. bred mice and confirmed the Ecm1−/− genotype; J.W. helped with immunoprecipitation experiments; G.L., L.T. and Y.J. made two ECM1 antibodies; M.Q. supplied reagents; S.S. performed flow cytometry; X.Z. helped design experiments and edit the manuscript; and B.S. conceived of the research, directed the study and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 (PDF 631 kb)
Rights and permissions
About this article
Cite this article
Li, Z., Zhang, Y., Liu, Z. et al. ECM1 controls TH2 cell egress from lymph nodes through re-expression of S1P1. Nat Immunol 12, 178–185 (2011). https://doi.org/10.1038/ni.1983
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1983
This article is cited by
-
Plasma host protein signatures correlating with Mycobacterium tuberculosis activity prior to and during antituberculosis treatment
Scientific Reports (2022)
-
Extracellular matrix protein 1 regulates cell proliferation and trastuzumab resistance through activation of epidermal growth factor signaling
Breast Cancer Research (2014)
-
Expression and clinical significance of extracellular matrix protein 1 and vascular endothelial growth factor-C in lymphatic metastasis of human breast cancer
BMC Cancer (2012)
-
Highlights of the advances in basic immunology in 2011
Cellular & Molecular Immunology (2012)
-
Travel agents for two
Nature Reviews Immunology (2011)