Abstract
Self-assembly offers the possibility to organize molecules in a given architecture through a subtle interplay between different noncovalent interactions. Although the kind of molecular association can often be predicted from information present in the individual molecules, the synthesis of supramolecular assemblies having a perfectly defined size and shape remains challenging. Here, we introduce the use of Coulombic interactions to control the supramolecular synthesis of finite, well-defined nanostructures. In particular, we demonstrate that the energy associated with the separation of ion pairs can regulate very precisely guanosine self-assembly into discrete G-quadruplexes. Assemblies comprising 8, 12, 16 or 24 guanosine molecules can be selectively and quantitatively obtained simply by tuning the stabilization of the dissociated anions in the solvent environment. Thus, factors such as solvent polarity, the nature of the anion and the cation–anion distance are shown to have a decisive role in the growth of G-quadruplexes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Whitesides, G. M. & Grzybowski, B. Self-assembly at all scales. Science 295, 2418–2421 (2002).
Schmittel, M. & Kalsani, V. Functional, discrete, nanoscale supramolecular assemblies. Top. Curr. Chem. 245, 1–53 (2005).
Prins, L. J., De Jong, F., Timmerman, P. & Reinhoudt, D. N. An enantiomerically pure hydrogen-bonded assembly. Nature 408, 181–184 (2000).
Sato, S. et al. Fluorous nanodroplets structurally confined in an organopalladium sphere. Science 313, 1273–1276 (2006).
Sugimoto, T., Suzuki, T., Shinkai, S. & Sada, K. A double-stranded helix by complexation of two polymer chains with a helical supramolecular assembly. J. Am. Chem. Soc. 129, 270–271 (2007).
Janssen, P. G. A., Vandenbergh, J., van Dongen, J. L., Meijer, E.W. & Schenning, A. P. H. J. ssDNA templated self-assembly of chromophores. J. Am. Chem. Soc. 129, 6078–6079 (2007).
Ikeda, A., Numata, M. & Shinkai, S. A novel attempt to control the aggregation number of dendrons with a saccharide. Chem. Lett. 9, 929–930 (1999).
Michelsen, U. & Hunter, C. A. Self-assembled porphyrin polymers. Angew. Chem. Int. Ed. 39, 764–767 (2000).
Brunsveld, L., Folmer, B. J. B., Meijer, E. W. & Sijbesma, R. Supramolecular polymers. Chem. Rev. 101, 4071–4098 (2001).
Sundquist, W. I. & Klug, A. Telomeric DNA dimerizes by formation of guanine tetrads between hairpin loops. Nature 342, 825–829 (1989).
Huppert, J. L. Four-stranded nucleic acids: structure, function and targeting of G-quadruplexes. Chem. Soc. Rev. 37, 1375–1384 (2008).
Davis, J. T. G-quartets 40 years later: From 5′-GMP to molecular biology and supramolecular chemistry. Angew. Chem. Int. Ed. 43, 668–698 (2004).
Davis, J. T. & Spada, G. P. Supramolecular architectures generated by self-assembly of guanosine derivatives. Chem. Soc. Rev. 36, 296–313 (2007).
Gu, J., Leczczynski, J. & Banal, M. A new insight into the structure and stability of Hoogsteen hydrogen-bonded G-tetrad: An ab initio SCF study. Chem. Phys. Lett. 311, 209–214 (1999).
García-Arriaga, M., Hobley, G. & Rivera, J. M. Isostructural self-assembly of 2′-deoxyguanosine derivatives in aqueous and organic media. J. Am. Chem. Soc. 130, 10492–10493 (2008)
Guschlbauer, W., Chantot, J. F. & Thiele, D. Four-stranded nucleic acid structures 25 years later: From guanosine gels to telomer DNA. J. Biomol. Struct. Dyn. 8, 491–511 (1990).
Gottarelli, G. et al. The self-assembly of lipophilic guanosine derivatives in solution and on solid surfaces. Chem. Eur. J. 6, 3242–3248 (2000).
Marlow, A. L. et al. Cation-templated self-assembly of a lipophilic deoxyguanosine: Solution structure of a K+-dG8 octamer. J. Org. Chem. 64, 5116–5123 (1999).
Forman, S. L., Fettinger, J. C., Pieraccini, S., Gottarelli, G. & Davis, J. T. Toward artificial ion channels: A lipophilic G-quadruplex. J. Am. Chem. Soc. 122, 4060–4067 (2000).
Shi, X., Fettinger, J. C. & Davis, J. T. Homochiral G-quadruplexes with Ba2+ but not with K+: The cation programs enantiomeric self-recognition. J. Am. Chem. Soc. 123, 6738–6739 (2001).
Shi, X. et al. Lipophilic G-quadruplexes are self-assembled ion pair receptors, and the bound anion modulates the kinetic stability of these complexes. J. Am. Chem. Soc. 125, 10830–10841 (2003).
Ma, L., Iezzi, M., Kaucher, M. S., Lam, Y.-F. & Davis, J. T. Cation exchange in lipophilic G-quadruplexes: Not all ion binding sites are equal. J. Am. Chem. Soc. 128, 15269–15277 (2006).
Kaucher, M. S., Lam, Y.-F., Pieraccini, S., Gottarelli, G. & Davis, J. T. Using diffusion NMR to characterize guanosine self-association: insights into structure and mechanism. Chem. Eur. J. 11, 164–173 (2005).
Prins, L. J., De Jong, F., Timmerman, P. & Reinhoudt, D. N. Noncovalent synthesis using hydrogen bonding. Angew. Chem. Int. Ed. 40, 2382 (2001).
Krossing, I. & Raabe, I. Noncoordination anions—Fact or fiction? A survey of likely candidates. Angew. Chem. Int. Ed. 43, 2066–2090 (2004).
Kotch, F. W. et al. Water-mediated association provides an ion pair receptor. J. Am. Chem. Soc. 125, 15140–15150 (2003).
Wüthrich, K. NMR of Proteins and Nucleic Acids 208 (Wiley, 1986).
Sessler, J. L., Sathiosatham, M., Doerr, K., Lynch, V. & Abboud, K. A. A G-quartet formed in the absence of a templating metal cation: A new 8-(N,N-dimethylaniline)guanosine derivative. Angew. Chem. Int. Ed. 39, 1300–1303 (2000).
Giorgi, T. et al. Supramolecular helices via self-assembly of 8-oxoguanosines. J. Am. Chem. Soc. 125, 14741–14749 (2002).
Mezzina, E. et al. The self-assembly of a lipophilic guanosine nucleoside into polymeric columnar aggregates: the nucleoside structure contains sufficient information to drive the process towards a strikingly regular polymer. Chem. Eur. J. 7, 388–395 (2001).
Mariani, P., Mazabard, C., Garbesi, A. & Spada, G. P. A study of the structure of the lyomesophases formed by the dinucleoside phosphate d(GpG). An approach by x-ray diffraction and optical microscopy. J. Am. Chem. Soc. 111, 6369–6373 (1989).
Acknowledgements
The authors are grateful for financial support from the Council for the Chemical Sciences of the Netherlands Organization for Scientific Research (CW-NWO). D.G.-R. would like to acknowledge a Marie Curie Intraeuropean Fellowship. We would also like to thank A. de la Escosura for his help with the transmission electron microscopy, P.G.A. Janssen and T. de Greef for discussions, and G.P. Spada and G. Gottarelli for discussions and support at the beginning of this project.
Author information
Authors and Affiliations
Contributions
D.G.-R. designed and performed the synthesis and experiments and wrote the paper. J.L.J.v.D. contributed to the MS experiments. M.L. and A.L.S. contributed to the X-ray structural analysis. A.P.H.J.S. and E.W.M. supervised the work.
Corresponding author
Supplementary information
Supplementary information
Supplementary information (PDF 5953 kb)
Supplementary information
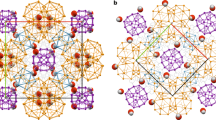
Crystallographic data for the 16-mer complex formed from compound 3 and KI (CIF 102 kb)
Rights and permissions
About this article
Cite this article
González-Rodríguez, D., van Dongen, J., Lutz, M. et al. G-quadruplex self-assembly regulated by Coulombic interactions. Nature Chem 1, 151–155 (2009). https://doi.org/10.1038/nchem.177
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.177