Abstract
Site-selective functionalizations of complex small molecules can generate targeted derivatives with exceptional step efficiency, but general strategies for maximizing selectivity in this context are rare. Here, we report that site-selectivity can be tuned by simply modifying the electronic nature of the reagents. A Hammett analysis is consistent with linking this phenomenon to the Hammond postulate: electronic tuning to a more product-like transition state amplifies site-discriminating interactions between a reagent and its substrate. This strategy transformed a minimally site-selective acylation reaction into a highly selective and thus preparatively useful one. Electronic tuning of both an acylpyridinium donor and its carboxylate counterion further promoted site-divergent functionalizations. With these advances, we achieve a range of modifications to just one of the many hydroxyl groups appended to the ion channel-forming natural product amphotericin B. Thus, electronic tuning of reagents represents an effective strategy for discovering and optimizing site-selective functionalization reactions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lewis, C. A. & Miller, S. J. Site-selective derivatization and remodeling of erythromycin A by using simple peptide-based chiral catalysts. Angew. Chem. Int. Ed. 45, 5616–5619 (2006).
Chen, M. S. & White, M. C. A predictably selective aliphatic C–H oxidation reaction for complex molecule synthesis. Science 318, 783–787 (2007).
Wender, P. A., Hilinski, M. K. & Mayweg, A. V. W. Late-stage intermolecular C–H activation for lead diversification: a highly chemoselective oxyfunctionalization of the C-9 position of potent bryostatin analogues. Org. Lett. 7, 79–82 (2005).
Peddibhotla, S., Dang, Y., Liu, J. O. & Romo, D. Simultaneous arming and structure/activity studies of natural products employing O–H insertions: an expedient and versatile strategy for natural products-based chemical genetics. J. Am. Chem. Soc. 129, 12222–12231 (2007).
Snyder, S. A., Gollner, A. & Chiriac, M. I. Regioselective reactions for programmable resveratrol oligomer synthesis. Nature 474, 461–466 (2011).
Pathak, T. P. & Miller, S. J. Site-selective bromination of vancomycin. J. Am. Chem. Soc. 134, 6120–6123 (2012).
Afagh, N. A. & Yudin, A. K. Chemoselectivity and the curious reactivity preferences of functional groups. Angew. Chem. Int. Ed. 49, 262–310 (2010).
Griswold, K. S. & Miller, S. J. A peptide-based catalyst approach to regioselective functionalization of carbohydrates. Tetrahedron 59, 8869–8875 (2003).
Sanchez-Rosello, M., Puchlopek, A. L., Morgan, A. J. & Miller, S. J. Site-selective cataylsis of phenyl thionoformate transfer as a tool for regioselective deoxygenation of polyols. J. Org. Chem. 73, 1774–1782 (2008).
Lewis, C. A., Merkel, J. & Miller, S. J. Catalytic site-selective synthesis and evaluation of a series of erythromycin analogs. Bioorg. Med. Chem. Lett. 18, 6007–6011 (2008).
Lewis, C. A., Longcore, K. E., Miller, S. J. & Wender, P. A. An approach to the site-selective diversification of apoptolidin A with peptide-based catalysts. J. Nat. Prod. 72, 1864–1869 (2009).
Kurahashi, T., Mizutani, T. & Yoshida, J. Functionalized DMAP catalysts for regioselective acetylation of carbohydrates. Tetrahedron 58, 8669–8677 (2002).
Kawabata, T. & Furuta, T. Nonenzymatic regioselective acylation of carbohydrates. Chem. Lett. 38, 640–647 (2009).
Kurahashi, T., Mizutani, T. & Yoshida, J. Effect of intramolecular hydrogen-bonding network on the relative reactivities of carbohydrate OH groups. J. Chem. Soc. Perkin Trans. I 465–473 (1999).
Luning, U. et al. Concave pyridines for selective acylations of polyols. Eur. J. Org. Chem. 1077–1084 (1998).
Katting, E. & Albert, M. Counterion-directed regioselective acetylation of octyl β-D-glucopyranoside. Org. Lett. 6, 945–948 (2004).
Gu, J., Ruppen, M. E. & Cai, P. Lipase-catalyzed regioselective esterification of rapamycin: synthesis of temsirolimus (CCI-779). Org. Lett. 7, 3945–3948 (2005).
Park, H. G., Do, J. H. & Chang, H. N. Regioselective enzymatic acylation of multi-hydroxyl compounds in organic synthesis. Biotechnol. Bioprocess Eng. 8, 1–8 (2003).
Hammond, G. S. A correlation of reaction rates. J. Am. Chem. Soc. 77, 334–338 (1955).
Leffler, J. E. & Grunwald, E. Rates and Equilibria of Organic Reactions (Wiley, 1963).
Jacobsen, E. N., Zhang, W. & Guler, M. L. Electronic tuning of asymmetric catalysts. J. Am. Chem. Soc. 113, 6703–6704 (1991).
Palucki, M. et al. The mechanistic basis for electronic effects on enantioselectivity in the (salen)Mn(III)-catalyzed epoxidation reaction. J. Am. Chem. Soc. 120, 948–954 (1998).
RajanBabu, T. V. & Casalnuovo, A. L. Role of electronic asymmetry in the design of new ligands: the asymmetric hydrocyanation reaction. J. Am. Chem. Soc. 118, 6325–6326 (1996).
Harper, K. C. & Sigman, M. S. Three-dimensional correlation of steric and electronic free energy relationships guides asymmetric propargylation. Science 333, 1875–1878 (2011).
Xu, S. et al. The DMAP-catalyzed acetylation of alcohols — a mechanistic study. Chem. Eur. J. 11, 4751–4757 (2005).
Lutz, V. et al. Structural analyses of N-acylated 4-(dimethylamino)pyridine (DMAP) salts. Chem. Eur. J. 15, 8548–8557 (2009).
Cheong, P. H., Legault, C. Y., Um, J. M., Celebi-Olcum, N. & Houk, K. N. Quantum mechanical investigations of organocataysis: mechanisms, reactivities, and selectivities. Chem. Rev. 111, 5042–5137 (2011).
Palacios, D. S., Anderson, T. M. & Burke, M. D. A post-PKS oxidation of the amphotericin B skeleton predicted to be critical for channel formation is not required for potent antifungal activity. J. Am. Chem. Soc. 129, 13804–13805 (2007).
Palacios, D. S., Dailey, I., Siebert, D. M., Wilcock, B. C. & Burke, M. D. Synthesis-enabled functional group deletions reveal key underpinnings of amphotericin B ion channel and antifungal activities. Proc. Natl Acad. Sci. USA 108, 6733–6738 (2011).
Gray, K. C. et al. Amphotericin primarily kills yeast by simply binding ergosterol. Proc. Natl Acad. Sci. USA 109, 2234–2239 (2012).
Silberstein, A. Conformational analysis of amphotericin B-cholesterol channel complex. J. Membr. Biol. 162, 117–126 (1998).
Baran, M. & Mazerski, M. Molecular modelling of amphotericin B–ergosterol primary complex in water. Biophys. Chem. 95, 125–133 (2002).
Neumann, A., Czub, J. & Baginski, M. On the possibility of the amphotericin B–sterol complex formation in cholesterol- and ergosterol-containing lipid bilayers: a molecular dynamics study. J. Phys. Chem. B 113, 15875–15885 (2009).
Croatt, M. P. & Carreira, E. M. Probing the role of the mycosamine C2′-OH on the activity of amphotericin B. Org. Lett. 13, 1390–1393 (2011).
Matsumori, N., Sawada, Y. & Murata, M. Mycosamine orientation of amphotericin B controlling interaction with ergosterol: sterol-dependent activity of conformation-restricted derivatives with an amino-carbonyl bridge. J. Am. Chem. Soc. 127, 10667–10675 (2005).
De Kruijff, B., Gerritsen, W. J., Oerlemans, A., Demel, R. A. & van Deenen, L. L. M. Polyene antibiotic–sterol interactions in membranes of Acholeplasma laidlawii cells and lecthin liposomes. III. Molecular structure of the polyene antibiotic–cholesterol complexes. Biochim. Biophys. Acta 339, 57–70 (1974).
Murata, M. et al. Ion channel complex of antibiotics as viewed by NMR. Pure Appl. Chem. 81, 1123–1129 (2009).
Volmer, A. A., Szpilman, A. M. & Carreira, E. M. Synthesis and biological evaluation of amphotericin B derivatives. Nat. Prod. Rep. 27, 1329–1349 (2010).
Pandey, R. C. & Rinehart, K. L. Polyene antibiotics. IX. An improved method for the preparation of methyl esters of polyene antibiotics. J. Antibiot. 30, 158–162 (1977).
Taylor, A. W., Costello, B. J., Hunter, P. A., MacLachlan, W. S. & Shanks, C. T. Synthesis and antifungal selectivity of new derivatives of amphotericin B modified at the C-13 position. J. Antibiot. 46, 486–493 (1993).
Matsushita, N. et al. Synthesis of 25-13C-amphotericin B methyl ester: a molecular probe for solid-state NMR measurements. Chem. Lett. 38, 114–115 (2009).
Jiang, L. & Chan, T. Regioselective acylation of hexopyranosides with pivaloyl chloride. J. Org. Chem. 63, 6035–6038 (1998).
Nicolaou, K. C. & Webber, S. E. Stereocontrolled total synthesis of lipoxins B. Synthesis 453–461 (1986).
Philippe, M. et al. New fatty monoesters of erythromycin A. Chem. Pharm. Bull. 38, 1672–1674 (1990).
Hammett, L. P. Some relations between reaction rates and equilibrium constants. Chem. Rev. 17, 125–136 (1935).
Garegg, P. & Samuelsson, B. Novel reagent system for converting a hydroxy-group into an iodo-group in carbohydrates with inversion of configuration. J. Chem. Soc. Perkin Trans. I 2866–2869 (1980).
Hutchins, R. et al. Nucleophilic borohydride: selective reductive displacement of halides, sulfonate esters, tertiary amines, and N,N-disulfonimides with borohydride reagents in polar aprotic solvents. J. Org. Chem. 43, 2259–2267 (1977).
Matsumori, N. et al. An amphotericin B-ergosterol covalent conjugate with powerful membrane permeabilizing activity. Chem. Biol. 11, 673–679 (2004).
Mayr, H. & Ofial, A. R. The reactivity-selectivity principle: an imperishable myth in organic chemistry. Angew. Chem. Int. Ed. 45, 1844–1854 (2006).
Fiori, K. W., Puchlopek, A. L. A. & Miller, S. J. Enantioselective sulfonylation reactions mediated by a tetrapeptide catalyst. Nature Chem. 1, 630–634 (2009).
Jordan, P. A., Kayser-Bricker, K. J. & Miller, S. J. Asymmetric phosphorylation through catalytic P(III) phosphoramidite transfer: enantioselective synthesis of D-myo-inositol-6-phosphate. Proc. Natl Acad. Sci. USA 107, 20620–20624 (2010).
Acknowledgements
The authors thank M.C. White for helpful discussions and P. Beak for a thoughtful review of the manuscript. The authors acknowledge Bristol-Myers Squibb for the gift of AmB, and the National Institutes of Health (GM080436) for financial support. M.D.B. is an HHMI Early Career Scientist.
Author information
Authors and Affiliations
Contributions
B.C.W., B.E.U. and M.D.B. designed experiments. B.C.W. performed acylation experiments. B.C.W., B.E.U., G.L.B., M.J.C. and T.M.A. contributed to the synthesis of intermediates and derivatives. B.C.W., B.E.U. and M.D.B. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 6878 kb)
Rights and permissions
About this article
Cite this article
Wilcock, B., Uno, B., Bromann, G. et al. Electronic tuning of site-selectivity. Nature Chem 4, 996–1003 (2012). https://doi.org/10.1038/nchem.1495
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1495
This article is cited by
-
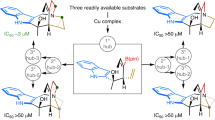
Site-selective chemical reactions by on-water surface sequential assembly
Nature Communications (2023)
-
Synergistic photoredox and copper catalysis by diode-like coordination polymer with twisted and polar copper–dye conjugation
Nature Communications (2020)
-
Site-switchable mono-O-allylation of polyols
Nature Communications (2020)
-
Late-stage chemoselective functional-group manipulation of bioactive natural products with super-electrophilic silylium ions
Nature Chemistry (2018)
-
π-Clamp-mediated cysteine conjugation
Nature Chemistry (2016)