Abstract
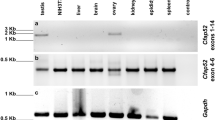
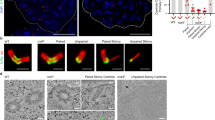
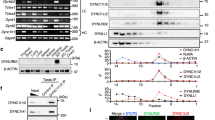
Outer dense fibre 2 (Odf2; also known as cenexin) was initially identified as a main component of the sperm tail cytoskeleton, but was later shown to be a general scaffold protein that is specifically localized at the distal/subdistal appendages of mother centrioles1,2. Here we show that Odf2 expression is suppressed in mouse F9 cells when both alleles of Odf2 genes are deleted. Unexpectedly, the cell cycle of Odf2−/− cells does not seem to be affected. Immunofluorescence and ultrathin-section electron microscopy reveals that in Odf2−/− cells, distal/subdistal appendages disappear from mother centrioles, making it difficult to distinguish mother from daughter centrioles. In Odf2−/− cells, however, the formation of primary cilia is completely suppressed, although ∼25% of wild-type F9 cells are ciliated under the steady-state cell cycle. The loss of primary cilia in Odf2−/− F9 cells can be rescued by exogenous Odf2 expression. These findings indicate that Odf2 is indispensable for the formation of distal/subdistal appendages and the generation of primary cilia, but not for other cell-cycle-related centriolar functions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lange, B. M. & Gull, K. A molecular marker for centriole maturation in the mammalian cell cycle. J. Cell Biol. 130, 919–927 (1995).
Nakagawa, Y., Yamane, Y., Okanoue, T., Tsukita, Sh. & Tsukita, Sa. Outer dense fiber 2 is a widespread centrosome scaffold component preferentially associated with mother centrioles: its identification from isolated centrosomes. Mol. Biol. Cell 12, 1687–1697 (2001).
Doxsey, S. Re-evaluating centrosome function. Nature Rev. Mol. Cell Biol. 2, 688–698 (2001).
Rieder, C. L., Faruki, S. & Khodjakov, A. The centrosome in vertebrates: more than a microtubule-organizing center. Trends Cell Biol. 11, 413–419 (2001).
Bornens, M. Centrosome composition and microtubule anchoring mechanisms. Curr. Opin. Cell Biol. 14, 25–34 (2002).
Kochanski, R. S. & Borisy, G. G. Mode of centriole duplication and distribution. J. Cell Biol. 110, 1599–1605 (1990).
Chretien, D., Buendia, B., Fuller, S. D. & Karsenti, E. Reconstruction of the centrosome cycle from cryoelectron micrographs. J. Struct. Biol. 120, 117–133 (1997).
Rieder, C. L. & Borisy, G. G. The centrosome cycle in PtK2 cells: asymmetric distribution and structural changes in the pericentriolar material. Biol. Cell 44, 117–132 (1982).
Vorobjev, I. A. & Chentsov Yu, S. Centrioles in the cell cycle. I. Epithelial cells. J. Cell Biol. 93, 938–949 (1982).
Wheatley, D. N. The Centriole: A Central Enigma of Cell Biology (Elsevier Biomedical Press, Amsterdam, USA, 1982).
Paintrand, M., Moudjou, M., Delacroix, H. & Bornens, M. Centrosome organization and centriole architecture: their sensitivity to divalent cations. J. Struct. Biol. 108, 107–128 (1992).
Mogensen, M. M., Malik, A., Piel, M., Bouckson-Castaing, V. & Bornens, M. Microtubule minus-end anchorage at centrosomal and non-centrosomal sites: the role of ninein. J. Cell Sci. 113, 3013–3023 (2000).
Chang, P., Giddings, T. H. Jr, Winey, M. & Stearns, T. ε-Tubulin is required for centriole duplication and microtubule organization. Nature Cell Biol. 5, 71–76 (2003).
Gromley, A. et al. A novel human protein of the maternal centriole is required for the final stages of cytokinesis and entry into S phase. J. Cell Biol. 161, 535–545 (2003).
Ou, Y. Y., Mack, G. J., Zhang, M. & Rattner, J. B. CEP110 and ninein are located in a specific domain of the centrosome associated with centrosome maturation. J. Cell Sci. 115, 1825–1835 (2002).
Dammermann, A. & Merdes, A. Assembly of centrosomal proteins and microtubule organization depends on PCM-1. J. Cell Biol. 159, 255–266 (2002).
Sorokin, S. P. Reconstructions of centriole formation and ciliogenesis in mammalian lungs. J. Cell Sci. 3, 207–230 (1968).
Piperno, G., LeDizet, M. & Chang, X. J. Microtubules containing acetylated α-tubulin in mammalian cells in culture. J. Cell Biol. 104, 289–302 (1987).
McGrath, J., Somlo, S., Makova, S., Tian, X. & Brueckner, M. Two populations of node monocilia initiate left-right asymmetry in the mouse. Cell 114, 61–73 (2003).
Watnick, T. & Germino, G. From cilia to cyst. Nature Genet. 34, 355–356 (2003).
The European Polycystic Kidney Disease Consortium. The polycystic kidney disease 1 gene encodes a 14 kb transcript and lies within a duplicated region on chromosome 16. Cell 77, 881–894 (1994).
Mochizuki, T. et al. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science 272, 1339–1342 (1996).
Pazour, G. J. et al. Chlamydomonas IFT88 and its mouse homologue, polycystic kidney disease gene Tg737, are required for assembly of cilia and flagella. J. Cell Biol. 151, 709–718 (2000).
Hou, X. et al. Cystin, a novel cilia-associated protein, is disrupted in the cpk mouse model of polycystic kidney disease. J. Clin. Invest. 109, 533–540 (2002).
Yoder, B. K., Hou, X. & Guay-Woodford, L. M. The polycystic kidney disease proteins, polycystin-1, polycystin-2, polaris, and cystin, are co-localized in renal cilia. J. Am. Soc. Nephrol. 13, 2508–2516 (2002).
Praetorius, H. A. & Spring, K. R. Bending the MDCK cell primary cilium increases intracellular calcium. J. Membr. Biol. 184, 71–79 (2001).
Nauli, S. M. & Zhou, J. Polycystins and mechanosensation in renal and nodal cilia. Bioessays 26, 844–856 (2004).
Rosenbaum, J. L. & Witman, G. B. Intraflagellar transport. Nature Rev. Mol. Cell Biol. 3, 813–825 (2002).
Nonaka, S. et al. Randomization of left-right asymmetry due to loss of nodal cilia generating leftward flow of extraembryonic fluid in mice lacking KIF3B motor protein. Cell 95, 829–837 (1998).
Jurczyk, A. et al. Pericentrin forms a complex with intraflagellar transport proteins and polycystin-2 and is required for primary cilia assembly. J. Cell Biol. 166, 637–643 (2004).
Acknowledgements
We thank all the members of our laboratory for helpful discussions. We are also grateful to N. Minato and Y. Hamazaki for help with FACS analysis. This work was supported in part by a Grant-in-Aid for Scientific Research (B) to Sa.T. from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Ishikawa, H., Kubo, A., Tsukita, S. et al. Odf2-deficient mother centrioles lack distal/subdistal appendages and the ability to generate primary cilia. Nat Cell Biol 7, 517–524 (2005). https://doi.org/10.1038/ncb1251
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1251
This article is cited by
-
Dual-color live imaging unveils stepwise organization of multiple basal body arrays by cytoskeletons
EMBO Reports (2024)
-
The apical ciliary adhesion complex is established at the basal foot of motile cilia and depends on the microtubule network
Scientific Reports (2022)
-
FOXA1 is a transcriptional activator of Odf2/Cenexin and regulates primary ciliation
Scientific Reports (2022)
-
The molecular dynamics of subdistal appendages in multi-ciliated cells
Nature Communications (2021)
-
Genome-Wide Association Study Identifies Genomic Loci of Sex Determination and Gonadosomatic Index Traits in Large Yellow Croaker (Larimichthys crocea)
Marine Biotechnology (2021)