Abstract
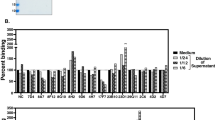
We have compared the in vivo therapeutic potential of anti-tetanus toxin (TT) human Fab antibodies derived from a combinatorial phage display library to established polyclonal and monoclonal reagents. The oligoclonality and fine specificity distribution of the synthetic anti-TT Fab preparations was comparable to the antibody spectrum present in the donor serum and the affinities determined for the synthetic phage-bound Fab (Phab) and soluble Fab were in the same range as their monoclonal and polyclonal counterparts. On a weight basis, the protective capacity of the new oligoclonal preparations in vivo (16.4 IU/100 μg Fab) was comparable to those of the best combinations of hybridoma derived human monoclonal antibodies, and far better than those exhibited by the polyclonal serum antibodies of the donor (0.29 IU/100 μg IgG) or by a standard commercial human tetanus immunoglobulin preparation. These data suggest that recombinant antibodies may become a safe and effective alternative to human plasma-derived immunoglobulins for passive immunization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
WHO Technical Report Series, No.840,1994.
Cross, A.S. 1991. Immunotherapy, p. 97–127. In: Immunotherapy and Vaccines, S. J. Cryz (Ed.). VCH Verlagsgesellschaft, Berlin.
Hammarström, L. and Smith, C.I.E. 1990. The use of intravenous IgG as prophylaxis and for treatment of infections. Infection 18: 314–324.
Masuho, Y. 1988. Human monoclonal antibodies: Prospects for use as passive immunotherapy. Serodiag. Immunother. Infect. Dis. 2: 319–340.
Larrick, J.W. 1991. Human monoclonal antibodies for the prevention and treatment of infectious diseases, p. 453–463. In: Vaccines and Immunotherapy, S. J. Cryz (Ed.). Pergamon Press, Inc. New York.
Winter, G. 1989. Antibody engineering. Phil. Trans. R. Soc. Lond. B324: 537–547.
Winter, G. and Milstein, C. 1991. Man-made antibodies. Nature 349: 293–299.
Plückthun, A. 1991. Antibody engineering: Advances from the use of Escherichia coli expression systems. Bio/Technology 9: 545–551.
Lerner, R.A., Kang, A.S., Bain, J.D., Burton, D.R. and Barbas, C.F. III 1992. Antibodies without immunization. Science 258: 1313–1314.
Hoogenboom, H.R., Marks, J.D., Griffiths, A.D. and Winter, G. 1992. Building antibodies from their genes. Immun. Rev. 130: 41–68.
Owens, R.J. and Young, R.J. 1994. The genetic engineering of monoclonal antibodies. J. Immunol. Methods 168: 149–165.
Lang, A.B., Cryz, S.J., Schürch, U., Ganss, M.T. and Bruderer, U. 1993. Immunotherapy with human monoclonal antibodies: Fragment A specificity of polyclonal and monoclonal antibodies is crucial for full protection against tetanus toxin. J. Immunol. 151: 466–473.
McCafferty, J., Griffiths, A.D., Winter, G. and Chiswell, D.J. 1990. Phage antibodies: filamentous phage displaying antibody variable domains. Nature 348: 552–554.
Kang, A.S., Barbas, C.F., Janda, K.D., Benkovic, S.J. and Lerner, R.A. 1991. Linkage of recognition and replication functions by assembling combinatorial antibody Fab libraries along phage surfaces. Proc. Natl. Acad. Sci. USA 88: 4363–4366.
Clackson, T., Hoogenboom, H.R., Griffiths, A.D. and Winter, G. 1991. Making antibody fragments using phage display libraries. Nature 352: 624–628.
Barbas, C.F. III and Lerner, R.A. 1991. Combinatorial immunoglobulin libraries on the surface of phage (Phabs): rapid selection of antigen-specific Fabs. Methods Comp. Methods Enzymol. 2: 119–124.
Barbas, C.F. III, Kang, A.S., Lerner, R.A. and Benkovic, S.J. 1991. Assembly of combinatorial antibody libraries on phage surfaces: the gene III site. Proc. Natl. Acad. Sci. USA 88: 7978–7982.
Burton, D.R., Barbas, C.F. III, Persson, M.A.A., Koenig, S., Chaock, R.M. and Lerner, R.A. 1991. A large array of human monoclonal antibodies to type 1 human immunodeficiency virus from combinatorial libraries of asymptomatic seropositive individuals. Proc. Natl. Acad. Sci. USA 88: 10134–10137.
Barbas, C.F. III, Björling, E., Chiodi, F., Dunlop, N., Cababa, D., Jones, T.M., Zebedee, S.L., Persson, M.A.A., Nara, P.L., Norrby, E. and Burton, D.R. 1992. Recombinant human Fab fragments neutralize human type 1 immunodeficiency virus in vitro. Proc. Natl. Acad. Sci. USA 89: 9339–9343.
Williamson, R.A., Burioni, R., Sanna, P.P., Partridge, L.J., Barbas, C.F. III and Burton, D.R. 1993. Human monoclonal antibodies against a plethora of viral pathogens from single combinatorial libraries. Proc. Natl. Acad. Sci. USA 90: 4141–4145.
Zebedee, S.L., Barbas, C.F. III, Hom, Y.L., Caothien, R.H., Graff, R., DeGraw, J., Pyati, J., LaPolla, R., Burton, D., Lerner, R.A. and Thorton, G.B. 1992. Human combinatorial antibody libraries to hepatitis B surface antigen. Proc. Natl. Acad. Sci. USA 89: 3175–3179.
Barbas, C.F. III, Crowe, J.E., Cababa, D., Jones, T.M., Zebedee, S.L., Murphy, B.R., Chanock, R.M. and Burton, D.R. 1992. Human monoclonal Fab fragments from a combinatorial library bind to respiratory syncytial virus F glycoprotein and neutralize infectivity. Proc. Natl. Acad. Sci. USA 89: 10164–10168.
Crowe, J.E., Murphy, B.R., Chanock, R.M., Williamson, R.A., Barbas, C.F. III and Burton, D.R. 1994. Recombinant human respiratory syncytial virus (RSV) monoclonal antibody Fab is effective therapeutically when introduced directly into the lungs of RSV-infected mice. Proc. Natl. Acad. Sci. USA 91: 1386–1390.
Bender, E., Woof, J.M., Atkin, J.D., Barker, M.D., Bebbington, C.R. and Burton, D.R. 1993. Recombinant human antibodies: linkage of an Fab fragment from a combinatorial library to an Fc fragment for expression in mammalian cell culture. Hum. Antibod. Hybridomas 4: 74–79.
Vogel, M., Miescher, S., Biaggi, C. and Stadler, B.M. 1994. Human anti-IgE antibodies by repertoire cloning. Eur. J. Immunol. 24: 1200–1207.
Bruderer, U., Deusinger, M., Schürch, U. and Lang, A.B. 1992. Analyses of affinity distributions within polyclonal populations of antigen specific antibodies: Evaluation of accuracy in population detection using monoclonal antibodies. J. Immunol. Methods 151: 157–164.
Bruderer, U., Fürer, E., Cryz, S.J. and Lang, A.B. 1990. Qualitative analysis of antibody binding. An in vitro assay for the evaluation and development of vaccines. J. Immunol. Methods 133: 263–268.
European Pharmacopeia. 1981. Tetanus antitoxin for human use, 2nd ed. Sainte Ruffine, France: Maisonneuve S. A., Monograph No. 91.
Lang, A.B., Fürer, E., Senyk, G., Larrick, J.W. and Cryz, S.J. 1990. Systematic generation of antigen specific human monoclonal antibodies with therapeutical activities using active immunization. Hum. Antibod. Hybridomas 1: 96–103.
Kamei, M., Hashizume, S., Sugimoto, N., Ozutsumi, K. and Matsuda, M. 1990. Establishment of stable mouse/human-human hybrid cell lines producing large amounts of anti-tetanus human monoclonal antibodies with high neutralizing activity. Eur. J. Epidemiol. 6: 386–397.
Volk, W.A., Bizzini, B., Snyder, R.M., Bernhard, E. and Wagner, R.R. 1984. Neutralization of tetanus toxin by distinct monoclonal antibodies binding to multiple epitopes on the toxin molecule. Infect. Immun. 45: 604–609.
Tempest, P.R., Bremner, P., Lambert, M., Taylor, G., Furze, J.M., Carr, F.J. and Harris, W.J. 1991. Reshaping a human monoclonal antibody to inhibit human respiratory syncytial virus infection in vivo. Bio/Technology 9: 266–271.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lang, A., Vogel, M., Viret, JF. et al. Polyclonal Preparations of Anti-Tetanus Toxoid Antibodies Derived from a Combinatorial Library Confer Protection. Nat Biotechnol 13, 683–685 (1995). https://doi.org/10.1038/nbt0795-683
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0795-683