Abstract
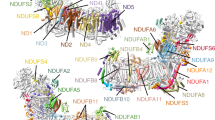
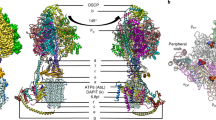
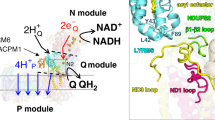
Complex I is the first and largest enzyme of the respiratory chain, coupling electron transfer between NADH and ubiquinone to the translocation of four protons across the membrane. It has a central role in cellular energy production and has been implicated in many human neurodegenerative diseases. The L-shaped enzyme consists of hydrophilic and membrane domains. Previously, we determined the structure of the hydrophilic domain. Here we report the crystal structure of the Esherichia coli complex I membrane domain at 3.0 Å resolution. It includes six subunits, NuoL, NuoM, NuoN, NuoA, NuoJ and NuoK, with 55 transmembrane helices. The fold of the homologous antiporter-like subunits L, M and N is novel, with two inverted structural repeats of five transmembrane helices arranged, unusually, face-to-back. Each repeat includes a discontinuous transmembrane helix and forms half of a channel across the membrane. A network of conserved polar residues connects the two half-channels, completing the proton translocation pathway. Unexpectedly, lysines rather than carboxylate residues act as the main elements of the proton pump in these subunits. The fourth probable proton-translocation channel is at the interface of subunits N, K, J and A. The structure indicates that proton translocation in complex I, uniquely, involves coordinated conformational changes in six symmetrical structural elements.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Walker, J. E. The NADH:ubiquinone oxidoreductase (complex I) of respiratory chains. Q. Rev. Biophys. 25, 253–324 (1992)
Yagi, T. & Matsuno-Yagi, A. The proton-translocating NADH–quinone oxidoreductase in the respiratory chain: the secret unlocked. Biochemistry 42, 2266–2274 (2003)
Brandt, U. Energy converting NADH:quinone oxidoreductase (complex I). Annu. Rev. Biochem. 75, 69–92 (2006)
Ohnishi, T. Iron-sulfur clusters/semiquinones in complex I. Biochim. Biophys. Acta 1364, 186–206 (1998)
Sazanov, L. A. Respiratory complex I: mechanistic and structural insights provided by the crystal structure of the hydrophilic domain. Biochemistry 46, 2275–2288 (2007)
Schapira, A. H. Human complex I defects in neurodegenerative diseases. Biochim. Biophys. Acta 1364, 261–270 (1998)
Dawson, T. M. & Dawson, V. L. Molecular pathways of neurodegeneration in Parkinson’s disease. Science 302, 819–822 (2003)
Balaban, R. S., Nemoto, S. & Finkel, T. Mitochondria, oxidants, and aging. Cell 120, 483–495 (2005)
Carroll, J. et al. Bovine complex I is a complex of 45 different subunits. J. Biol. Chem. 281, 32724–32727 (2006)
Yip, C. Y., Harbour, M. E., Jayawardena, K., Fearnley, I. M. & Sazanov, L. A. Evolution of respiratory complex I: “supernumerary” subunits are present in the alpha-proteobacterial enzyme. J. Biol. Chem. 286, 5023–5033 (2011)
Efremov, R. G., Baradaran, R. & Sazanov, L. A. The architecture of respiratory complex I. Nature 465, 441–445 (2010)
Sazanov, L. A. & Hinchliffe, P. Structure of the hydrophilic domain of respiratory complex I from Thermus thermophilus . Science 311, 1430–1436 (2006)
Berrisford, J. M. & Sazanov, L. A. Structural basis for the mechanism of respiratory complex I. J. Biol. Chem. 284, 29773–29783 (2009)
Fearnley, I. M. & Walker, J. E. Conservation of sequences of subunits of mitochondrial complex I and their relationships with other proteins. Biochim. Biophys. Acta 1140, 105–134 (1992)
Mathiesen, C. & Hagerhall, C. Transmembrane topology of the NuoL, M and N subunits of NADH:quinone oxidoreductase and their homologues among membrane-bound hydrogenases and bona fide antiporters. Biochim. Biophys. Acta 1556, 121–132 (2002)
Friedrich, T. Complex I: a chimaera of a redox and conformation-driven proton pump? J. Bioenerg. Biomembr. 33, 169–177 (2001)
Hunte, C., Zickermann, V. & Brandt, U. Functional modules and structural basis of conformational coupling in mitochondrial complex I. Science 329, 448–451 (2010)
Baranova, E. A., Holt, P. J. & Sazanov, L. A. Projection structure of the membrane domain of Escherichia coli respiratory complex I at 8 Å resolution. J. Mol. Biol. 366, 140–154 (2007)
von Heijne, G. Membrane protein structure prediction. Hydrophobicity analysis and the positive-inside rule. J. Mol. Biol. 225, 487–494 (1992)
Yau, W. M., Wimley, W. C., Gawrisch, K. & White, S. H. The preference of tryptophan for membrane interfaces. Biochemistry 37, 14713–14718 (1998)
Vik, S. B. The transmembrane helices of the L, M, and N subunits of Complex I from E. coli can be assigned on the basis of conservation and hydrophobic moment analysis. FEBS Lett. 585, 1180–1184 (2011)
Vinothkumar, K. R. & Henderson, R. Structures of membrane proteins. Q. Rev. Biophys. 43, 65–158 (2010)
Screpanti, E. & Hunte, C. Discontinuous membrane helices in transport proteins and their correlation with function. J. Struct. Biol. 159, 261–267 (2007)
Luecke, H., Schobert, B., Richter, H. T., Cartailler, J. P. & Lanyi, J. K. Structure of bacteriorhodopsin at 1.55 Å resolution. J. Mol. Biol. 291, 899–911 (1999)
Cooley, R. B., Arp, D. J. & Karplus, P. A. Evolutionary origin of a secondary structure: π-helices as cryptic but widespread insertional variations of α-helices that enhance protein functionality. J. Mol. Biol. 404, 232–246 (2010)
Morino, M. et al. Single site mutations in the hetero-oligomeric Mrp antiporter from alkaliphilic Bacillus pseudofirmus OF4 that affect Na+/H+ antiport activity, sodium exclusion, individual Mrp protein levels, or Mrp complex formation. J. Biol. Chem. 285, 30942–30950 (2010)
Nakamaru-Ogiso, E. et al. the membrane subunit NuoL (ND5) is involved in the indirect proton pumping mechanism of Escherichia coli complex I. J. Biol. Chem. 285, 39070–39078 (2010)
Amarneh, B. & Vik, S. B. Mutagenesis of subunit N of the Escherichia coli complex I. Identification of the initiation codon and the sensitivity of mutants to decylubiquinone. Biochemistry 42, 4800–4808 (2003)
Mathiesen, C. & Hagerhall, C. The ‘antiporter module’ of respiratory chain complex I includes the MrpC/NuoK subunit—a revision of the modular evolution scheme. FEBS Lett. 549, 7–13 (2003)
Euro, L., Belevich, G., Verkhovsky, M. I., Wikstrom, M. & Verkhovskaya, M. Conserved lysine residues of the membrane subunit NuoM are involved in energy conversion by the proton-pumping NADH:ubiquinone oxidoreductase (complex I). Biochim. Biophys. Acta 1777, 1166–1172 (2008)
Torres-Bacete, J., Nakamaru-Ogiso, E., Matsuno-Yagi, A. & Yagi, T. Characterization of the NuoM (ND4) subunit in Escherichia coli NDH-1: conserved charged residues essential for energy-coupled activities. J. Biol. Chem. 282, 36914–36922 (2007)
Kao, M. C., Nakamaru-Ogiso, E., Matsuno-Yagi, A. & Yagi, T. Characterization of the membrane domain subunit NuoK (ND4L) of the NADH-quinone oxidoreductase from Escherichia coli . Biochemistry 44, 9545–9554 (2005)
Kervinen, M., Patsi, J., Finel, M. & Hassinen, I. E. A pair of membrane-embedded acidic residues in the NuoK subunit of Escherichia coli NDH-1, a counterpart of the ND4L subunit of the mitochondrial complex I, are required for high ubiquinone reductase activity. Biochemistry 43, 773–781 (2004)
Fisher, N. & Rich, P. R. A motif for quinone binding sites in respiratory and photosynthetic systems. J. Mol. Biol. 296, 1153–1162 (2000)
Ohnishi, T., Nakamaru-Ogiso, E. & Ohnishi, S. T. A new hypothesis on the simultaneous direct and indirect proton pump mechanisms in NADH-quinone oxidoreductase (complex I). FEBS Lett. 584, 4131–4137 (2010)
Nakamaru-Ogiso, E., Sakamoto, K., Matsuno-Yagi, A., Miyoshi, H. & Yagi, T. The ND5 subunit was labeled by a photoaffinity analogue of fenpyroximate in bovine mitochondrial complex I. Biochemistry 42, 746–754 (2003)
Steimle, S. et al. The role of subunit NuoL for proton translocation by the respiratory complex I. Biochemistry 50, 3386–3393 (2011)
Michel, J., Deleon-Rangel, J., Zhu, S., Van Ree, K. & Vik, S. B. mutagenesis of the L, M, and N subunits of complex I from Escherichia coli indicates a common role in function. PLoS ONE 6, e17420 (2011)
Krishnamoorthy, G. & Hinkle, P. C. Studies on the electron transfer pathway, topography of iron-sulfur centers, and site of coupling in NADH-Q oxidoreductase. J. Biol. Chem. 263, 17566–17575 (1988)
Kao, M. C. et al. Characterization of the membrane domain subunit NuoJ (ND6) of the NADH-quinone oxidoreductase from Escherichia coli by chromosomal DNA manipulation. Biochemistry 44, 3562–3571 (2005)
Pätsi, J., Kervinen, M., Finel, M. & Hassinen, I. E. Leber hereditary optic neuropathy mutations in the ND6 subunit of mitochondrial complex I affect ubiquinone reduction kinetics in a bacterial model of the enzyme. Biochem. J. 409, 129–137 (2008)
Galkin, A. S., Grivennikova, V. G. & Vinogradov, A. D. H+/2e− stoichiometry in NADH-quinone reductase reactions catalyzed by bovine heart submitochondrial particles. FEBS Lett. 451, 157–161 (1999)
Treberg, J. R. & Brand, M. D. A model of the proton translocation mechanism of complex I. J. Biol. Chem. 286, 17579–17584 (2011)
Ohnishi, S. T., Salerno, J. C. & Ohnishi, T. Possible roles of two quinone molecules in direct and indirect proton pumps of bovine heart NADH-quinone oxidoreductase (complex I). Biochim. Biophys. Acta 1797, 1891–1893 (2010)
Sazanov, L. A., Carroll, J., Holt, P., Toime, L. & Fearnley, I. M. A role for native lipids in the stabilization and two-dimensional crystallization of the Escherichia coli NADH-ubiquinone oxidoreductase (Complex I). J. Biol. Chem. 278, 19483–19491 (2003)
Van Duyne, G. D., Standaert, R. F., Karplus, P. A., Schreiber, S. L. & Clardy, J. Atomic structures of the human immunophilin FKBP-12 complexes with FK506 and rapamycin. J. Mol. Biol. 229, 105–124 (1993)
Leif, H., Sled, V. D., Ohnishi, T., Weiss, H. & Friedrich, T. Isolation and characterization of the proton-translocating NADH: ubiquinone oxidoreductase from Escherichia coli . Eur. J. Biochem. 230, 538–548 (1995)
Collaborative Computational Project 4.. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Strong, M. et al. Toward the structural genomics of complexes: crystal structure of a PE/PPE protein complex from Mycobacterium tuberculosis . Proc. Natl Acad. Sci. USA 103, 8060–8065 (2006)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Cryst. 40, 658–674 (2007)
de La Fortelle, E. & Bricogne, G. Maximum-likelihood heavy-atom parameter refinement for multiple isomorphous replacement and multiwavelength anomalous diffraction methods. Methods Enzymol. 276, 472–494 (1997)
Brunger, A. T. Version 1.2 of the crystallography and NMR system. Nature Protocols 2, 2728–2733 (2007)
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002)
Jones, T. A. & Kjeldgaard, M. Electron-density map interpretation. Methods Enzymol. 277, 173–208 (1997)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr.D 60, 2126–2132 (2004)
Cowtan, K. The Buccaneer software for automated model building. 1. Tracing protein chains. Acta Crystallogr. D 62, 1002–1011 (2006)
Laskowski, R. A., MacArthur, M. W., Moss, D. S. & Thornton, J. M. PROCHECK: a program to check the stereochemical quality of protein structures. J. Appl. Cryst. 26, 283–291 (1993)
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010)
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994)
Eswar, N. et al. Comparative protein structure modeling using Modeller. Curr. Protoc. Bioinformatics Ch. 5, Unit 5.6. (2006)
Acknowledgements
This work was funded by the Medical Research Council. We thank the ESRF and the SLS for provision of synchrotron radiation facilities. We are grateful to the staff of beamlines ID29 (ESRF) and X06SA (SLS) for assistance.
Author information
Authors and Affiliations
Contributions
R.G.E. performed research and analysed data; L.A.S. designed the project, analysed data and wrote the manuscript, with contributions from R.G.E.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, Supplementary Tables 1-8, Supplementary Figures 1-8 with legends and additional references. (PDF 7855 kb)
Supplementary Movie 1
This movie illustrates the overall architecture of the membrane domain, showing arrangement of subunits, connecting elements, key helices and key charged residues. Towards the end, the arrangement of peripheral domain is illustrated using T. thermophilus structure (PDB 3I9V) shown in grey with Fe-S clusters as spheres. (MOV 22129 kb)
Supplementary Movie 2
This movie illustrates the fold of antiporter-like subunits (using subunit NuoM), showing the fold from N- to C-terminus, key helices, key charged residues and internal symmetry. Two repeating domains are shown in magenta and green. (MOV 25622 kb)
Rights and permissions
About this article
Cite this article
Efremov, R., Sazanov, L. Structure of the membrane domain of respiratory complex I. Nature 476, 414–420 (2011). https://doi.org/10.1038/nature10330
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10330
This article is cited by
-
Mitochondrial genomic analyses provide new insights into the “missing” atp8 and adaptive evolution of Mytilidae
BMC Genomics (2022)
-
The coupling mechanism of mammalian mitochondrial complex I
Nature Structural & Molecular Biology (2022)
-
Positive selection over the mitochondrial genome and its role in the diversification of gentoo penguins in response to adaptation in isolation
Scientific Reports (2022)
-
Mutations in respiratory complex I promote antibiotic persistence through alterations in intracellular acidity and protein synthesis
Nature Communications (2022)
-
Mechanisms of membrane protein crystallization in ‘bicelles’
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.