Abstract
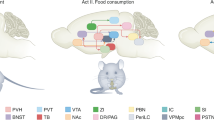
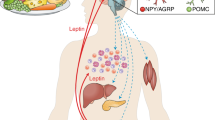
The increasing incidence of obesity and obesity-linked disease presents a serious global health threat. To develop truly effective therapies to modulate food intake and promote weight loss, we must understand the physiological regulators that underlie these processes. One crucial mediator of food intake and energy homeostasis is the adipose-derived hormone, leptin, which acts through neurons expressing the long form of the leptin receptor (LepRb). Although most investigation of leptin action has centered on the large population of LepRb neurons in the arcuate nucleus (ARC), this nucleus does not mediate all aspects of leptin action. Indeed, several hypothalamic and extrahypothalamic loci contain substantial numbers of LepRb neurons, each of which presumably mediates distinct aspects of leptin action, and the collective output of these various LepRb populations produces the totality of leptin function. This review will examine known central nervous system loci that contain LepRb neurons and the potential roles for discrete populations of LepRb neurons in the control of homeostatic and hedonic pathways by leptin. Understanding the unique neuroanatomical and functional roles for each locus of leptin action will be important to identify how specific aspects of food intake contribute to obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Pelleymounter MA, Cullen MJ, Baker MB, Hecht R, Winters D, Boone T et al. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 1995; 269: 540–543.
Chua Jr SC, Chung WK, Wu-Peng XS, Zhang Y, Liu SM, Tartaglia L et al. Phenotypes of mouse diabetes and rat fatty due to mutations in the OB (leptin) receptor. Science 1996; 271: 994–996.
Elmquist JK, Bjorbaek C, Ahima RS, Flier JS, Saper CB . Distributions of leptin receptor mRNA isoforms in the rat brain. J Comp Neurol 1998; 395: 535–547.
Elmquist JK, Coppari R, Balthasar N, Ichinose M, Lowell BB . Identifying hypothalamic pathways controlling food intake, body weight, and glucose homeostasis. J Comp Neurol 2005; 493: 63–71.
Cone RD . Anatomy and regulation of the central melanocortin system. Nat Neurosci 2005; 8: 571–578.
Dhillon H, Zigman JM, Ye C, Lee CE, McGovern RA, Tang V et al. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 2006; 49: 191–203.
Zhang R, Dhillon H, Yin H, Yoshimura A, Lowell BB, Maratos-Flier E et al. Selective inactivation of Socs3 in SF1 neurons improves glucose homeostasis without affecting body weight. Endocrinology 2008; 149: 5654–5661.
Bingham NC, Anderson KK, Reuter AL, Stallings NR, Parker KL . Selective loss of leptin receptors in the ventromedial hypothalamic nucleus results in increased adiposity and a metabolic syndrome. Endocrinology 2008; 149: 2138–2148.
Majdic G, Young M, Gomez-Sanchez E, Anderson P, Szczepaniak LS, Dobbins RL et al. Knockout mice lacking steroidogenic factor 1 are a novel genetic model of hypothalamic obesity. Endocrinology 2002; 143: 607–614.
Sanz C, Roncero I, Vazquez P, Navas MA, Blazquez E . Effects of glucose and insulin on glucokinase activity in rat hypothalamus. J Endocrinol 2007; 193: 259–267.
Minokoshi Y, Haque MS, Shimazu T . Microinjection of leptin into the ventromedial hypothalamus increases glucose uptake in peripheral tissues in rats. Diabetes 1999; 48: 287–291.
Sternson SM, Shepherd GM, Friedman JM . Topographic mapping of VMH → arcuate nucleus microcircuits and their reorganization by fasting. Nat Neurosci 2005; 8: 1356–1363.
Ellacott KL, Halatchev IG, Cone RD . Characterization of leptin-responsive neurons in the caudal brainstem. Endocrinology 2006; 147: 3190–3195.
Huo L, Maeng L, Bjorbaek C, Grill HJ . Leptin and the control of food intake: neurons in the nucleus of the solitary tract are activated by both gastric distension and leptin. Endocrinology 2007; 148: 2189–2197.
Peters JH, Simasko SM, Ritter RC . Modulation of vagal afferent excitation and reduction of food intake by leptin and cholecystokinin. Physiol Behav 2006; 89: 477–485.
Wise RA . Brain reward circuitry: insights from unsensed incentives. Neuron 2002; 36: 229–240.
Fulton S, Pissios P, Manchon RP, Stiles L, Frank L, Pothos EN et al. Leptin regulation of the mesoaccumbens dopamine pathway. Neuron 2006; 51: 811–822.
Roseberry AG, Painter T, Mark GP, Williams JT . Decreased vesicular somatodendritic dopamine stores in leptin-deficient mice. J Neurosci 2007; 27: 7021–7027.
Figlewicz DP, Evans SB, Murphy J, Hoen M, Baskin DG . Expression of receptors for insulin and leptin in the ventral tegmental area/substantia nigra (VTA/SN) of the rat. Brain Res 2003; 964: 107–115.
Hommel JD, Trinko R, Sears RM, Georgescu D, Liu ZW, Gao XB et al. Leptin receptor signaling in midbrain dopamine neurons regulates feeding. Neuron 2006; 51: 801–810.
Morrison SD, Mayer J . Adipsia and aphagia in rats after lateral subthalamic lesions. Am J Physiol 1957; 191: 248–254.
DiLeone RJ, Georgescu D, Nestler EJ . Lateral hypothalamic neuropeptides in reward and drug addiction. Life Sci 2003; 73: 759–768.
Georgescu D, Sears RM, Hommel JD, Barrot M, Bolanos CA, Marsh DJ et al. The hypothalamic neuropeptide melanin-concentrating hormone acts in the nucleus accumbens to modulate feeding behavior and forced-swim performance. J Neurosci 2005; 25: 2933–2940.
Georgescu D, Zachariou V, Barrot M, Mieda M, Willie JT, Eisch AJ et al. Involvement of the lateral hypothalamic peptide orexin in morphine dependence and withdrawal. J Neurosci 2003; 23: 3106–3111.
Zheng H, Patterson LM, Berthoud HR . Orexin signaling in the ventral tegmental area is required for high-fat appetite induced by opioid stimulation of the nucleus accumbens. J Neurosci 2007; 27: 11075–11082.
Borowsky B, Durkin MM, Ogozalek K, Marzabadi MR, DeLeon J, Lagu B et al. Antidepressant, anxiolytic and anorectic effects of a melanin-concentrating hormone-1 receptor antagonist. Nat Med 2002; 8: 825–830.
Rodgers RJ, Halford JC, Nunes de Souza RL, Canto de Souza AL, Piper DC, Arch JR et al. SB-334867, a selective orexin-1 receptor antagonist, enhances behavioural satiety and blocks the hyperphagic effect of orexin-A in rats. Eur J Neurosci 2001; 13: 1444–1452.
Sakurai T . The neural circuit of orexin (hypocretin): maintaining sleep and wakefulness. Nat Rev 2007; 8: 171–181.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Leinninger, G. Location, location, location: the CNS sites of leptin action dictate its regulation of homeostatic and hedonic pathways. Int J Obes 33 (Suppl 2), S14–S17 (2009). https://doi.org/10.1038/ijo.2009.66
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2009.66
Keywords
This article is cited by
-
Distinct Subsets of Lateral Hypothalamic Neurotensin Neurons are Activated by Leptin or Dehydration
Scientific Reports (2019)
-
Responses to drugs of abuse and non-drug rewards in leptin deficient ob/ob mice
Psychopharmacology (2016)