Abstract
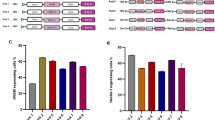
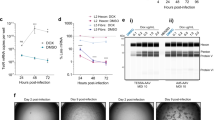
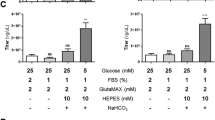
Recombinant herpes simplex virus type 1 (rHSV)-assisted recombinant adeno-associated virus (rAAV) vector production provides a highly efficient and scalable method for manufacture of clinical grade rAAV vectors. Here, we present an rHSV co-infection system for rAAV production, which uses two ICP27-deficient rHSV constructs, one bearing the rep2 and cap (1, 2 or 9) genes of rAAV, and the second bearing an AAV2 ITR-gene of interest (GOI) cassette. The optimum rAAV production parameters were defined by producing rAAV2/GFP in HEK293 cells, yielding greater than 9000 infectious particles per cell with a 14:1 DNase resistance particle to infectious particle (DRP/ip) ratio. The optimized co-infection parameters were then used to generate large-scale stocks of rAAV1/AAT, which encode the human α-1-antitrypsin (hAAT) protein, and purified by column chromatography. The purified vector was extensively characterized by rAAV- and rHSV-specific assays and compared to transfection-made vector for in vivo efficacy in mice through intramuscular injection. The co-infection method was also used to produce rAAV9/AAT for comparison to rAAV1/AAT in vivo. Intramuscular administration of 1 × 1011 DRP per animal of rHSV-produced rAAV1/AAT and rAAV9/AAT resulted in hAAT protein expression of 5.4 × 104 and 9.4 × 105 ng ml−1 serum respectively, the latter being clinically relevant.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hermonat PL, Muzyczka N . Use of adeno-associated virus as a mammalian DNA cloning vector: transduction of neomycin resistance into mammalian tissue culture cells. Proc Natl Acad Sci USA 1984; 81: 6466–6470.
Tratschin JD, West MH, Sandbank T, Carter BJ . A human parvovirus, adeno-associated virus, as a eukaryotic vector: transient expression and encapsidation of the prokaryotic gene for chloramphenicol acetyltransferase. Mol Cell Biol 1984; 4: 2072–2081.
Muzyczka N, Berns KI . Parvoviridae: the viruses and their replication. In: Knipe DM, Howley PM (eds). Fields Virology, 4th edn. Lippincott, Williams and Wilkins: New York, NY, 2001, pp 2327–2360.
Kotin RM, Siniscalco M, Samulski RJ, Zhu XD, Hunter L, Laughlin CA et al. Site-specific integration by adeno-associated virus. Proc Natl Acad Sci USA 1990; 87: 2211–2215.
Kotin RM, Menninger JC, Ward DC, Berns KI . Mapping and direct visualization of a region-specific viral DNA integration site on chromosome 19q13-qter. Genomics 1991; 10: 831–834.
Samulski RJ, Zhu X, Xiao X, Brook JD, Housman DE, Epstein N et al. Targeted integration of adeno-associated virus (AAV) into human chromosome 19 [published erratum appears in EMBO J 1992; 11: 1228]. EMBO J 1991; 10: 3941–3950.
Kay MA, Manno CS, Ragni MV, Larson PJ, Couto LB, McClelland A et al. Evidence for gene transfer and expression of factor IX in haemophilia B patients treated with an AAV vector. Nat Genet 2000; 24: 257–261.
Jiang H, Pierce GF, Ozelo MC, de Paula EV, Vargas JA, Smith P et al. Evidence of multiyear factor IX expression by AAV-mediated gene transfer to skeletal muscle in an individual with severe hemophilia B. Mol Ther 2006; 14: 452–455.
Kaplitt MG, Feigin A, Tang C, Fitzsimons HL, Mattis P, Lawlor PA et al. Safety and tolerability of gene therapy with an adeno-associated virus (AAV) borne GAD gene for Parkinson's disease: an open label, phase I trial. Lancet 2007; 369: 2097–2105.
Worgall S, Sondhi D, Hackett NR, Kosofsky B, Kekatpure MV, Neyzi N et al. Treatment of late infantile neuronal ceroid lipofuscinosis by CNS administration of a serotype 2 adeno-associated virus expressing CLN2 cDNA. Hum Gene Ther 2008; 19: 463–474.
Stedman H, Wilson JM, Finke R, Kleckner AL, Mendell J . Phase I clinical trial utilizing gene therapy for limb girdle muscular dystrophy: α-, β-, γ-, or Δ-sarcoglycan gene delivered with intramuscular instillations of adeno-associated vectors. Hum Gene Ther 2000; 11: 777–790.
Tebbutt SJ . Technology evaluation: AAV-CFTR vector, targeted genetics. Curr Opin Mol Ther 1999; 1: 524–529.
Brantly ML, Spencer LT, Humphries M, Conlon TJ, Spencer CT, Poirier A et al. Phase I trial of intramuscular injection of a recombinant adeno-associated virus serotype 2 alpha1-antitrypsin (AAT) vector in AAT-deficient adults. Hum Gene Ther 2006; 17: 1177–1186.
Muzyczka N . Use of adeno-associated virus as a general transduction vector for mammalian cells. Curr Top Microbiol Immunol 1992; 158: 97–129.
Samulski RJ, Srivastava A, Berns KI, Muzyczka N . Rescue of adeno-associated virus from recombinant plasmids: gene correction within the terminal repeats of AAV. Cell 1983; 33: 135–143.
Berns KI . The Parvoviruses. Plenum Press: New York, 1984.
Chejanovsky N, Carter BJ . Mutagenesis of an AUG codon in the adeno-associated virus rep gene: effects on viral DNA replication. Virology 1989; 173: 120–128.
Samulski RJ, Chang LS, Shenk T . A recombinant plasmid from which an infectious adeno-associated virus genome can be excised in vitro and its use to study viral replication. J Virol 1987; 61: 3096–3110.
Chejanovsky N, Carter BJ . Mutation of a consensus purine nucleotide binding site in the adeno-associated virus rep gene generates a dominant negative phenotype for DNA replication. J Virol 1990; 64: 1764–1770.
Huang MM, Hearing P . Adenovirus early region 4 encodes two gene products with redundant effects in lytic infection. J Virol 1989; 63: 2605–2615.
Samulski RJ, Shenk T . Adenovirus E1B 55-Mr polypeptide facilitates timely cytoplasmic accumulation of adeno-associated virus mRNAs. J Virol 1988; 62: 206–210.
Xiao X, Li J, Samulski RJ . Production of high-titer recombinant adeno-associated virus vectors in the absence of helper adenovirus. J Virol 1998; 72: 2224–2232.
Clark KR . Recent advances in recombinant adeno-associated virus vector production. Kidney Int 2002; 61s: 9–15.
Qiao C, Li J, Skold A, Zhang X, Xiao X . Feasibility of generating adeno-associated virus packaging cell lines containing inducible adenovirus helper genes. J Virol 2002; 76: 1904–1913.
Clark KR, Voulgaropoulou F, Fraley DM, Johnson PR . Cell lines for the production of recombinant adeno-associated virus. Hum Gene Ther 1995; 6: 1329–1341.
Grimm D, Kern A, Rittner K, Kleinschmidt JA . Novel Tools for production and purification of recombinant adeno-associated virus vectors. Hum Gene Ther 1998; 9: 2745–2760.
Zolotukhin S, Potter M, Zolotukhin I, Sakai Y, Loiler S, Fraites Jr TJ et al. Production and purification of serotype 1, 2, and 5 recombinant adeno-associated viral vectors. Methods 2002; 28: 158–167.
Buller RML, Janik JE, Sebring ED, Rose JA . Herpes simplex virus types 1 and 2 completely help adenovirus-associated virus replication. J Virol 1981; 40: 241–247.
Weindler FW, Heilbronn R . A Subset of herpes simplex virus replication genes provides helper functions for productive adeno-associated virus replication. J Virol 1991; 65: 2476–2483.
Conway JE, ap Rhys CMJ, Zolotukhin I, Zolotukhin S, Muzyczka N, Hayward GS et al. High-titer recombinant adeno-associated virus production utilizing a recombinant herpes simplex virus type I vector expressing AAV2 Rep and Cap. Gene Therapy 1999; 6: 986–993.
Conway JE, Zolotukhin S, Muzyczka N, Hayward GS, Byrne BJ . Recombinant adeno-associated virus type 2 replication and packaging is entirely supported by a herpes simplex virus type 1 amplicon expressing rep and cap. J Virol 1997; 71: 8780–8789.
Mistry AR, De Alwis M, Feudner E, Ali RR, Thrasher AJ . High-titer stocks of adeno-associated virus from replicating amplicons and herpes vectors. Methods Mol Med 2002; 69: 445–460.
Wustner JT, Arnold S, Lock M, Richardson JC, Himes VB, Kurtzman G et al. Production of recombinant adeno-associated type 5 (rAAV5) vectors using recombinant herpes simplex viruses containing rep and cap. Mol Ther 2002; 6: 510–518.
Feudner E, de Alwis M, Thrasher AJ, Ali RR, Fauser S . Optimization of recombinant adeno-associated virus production using an herpes simplex virus amplicon system. J Virol Methods 2001; 96: 97–105.
Zhang X, de Alwis M, Hart SL, Fitzke FW, Inglis SC, Boursnell MEG et al. High-titer recombinant adeno-associated virus production from replicating amplicons and herpes vectors deleted for glycoprotein H. Hum Gene Ther 1999; 10: 2527–2537.
Booth MJ, Mistry A, Li X, Thrasher A, Coffin RS . Transfection-free and scalable recombinant AAV vector production using HSV/AAV hybrids. Gene Therapy 2004; 11: 829–837.
Toublanc E, Benraiss A, Bonnin D, Blouin V, Brument N, Cartier N et al. Identification of a replication-defective herpes simplex virus for recombinant adeno-associated virus type 2 (rAAV2) particle assembly using stable producer cell lines. J Gene Med 2004; 6: 555–564.
Sollerbrant K, Elmén J, Wahlestedt C, Acker J, Leblois-Prehaud H, Latta-Mahieu M et al. A novel method using baculovirus-mediated gene transfer for production of recombinant adeno-associated virus vectors. J Gen Virol 2001; 82: 2051–2060.
Urabe M, Ding C, Kotin RM . Insect cells as a factory to produce adeno-associated virus type 2 vectors. Hum Gene Ther 2002; 13: 1935–1943.
Meghrous J, Aucoin MG, Jacob D, Chahal PS, Arcand N, Kamen AA . Production of recombinant adeno-associated viral vectors using a baculovirus/insect cell suspension culture system: from shake flasks to a 20-L bioreactor. Biotechnol Prog 2005; 21: 154–160.
Kohlbrenner E, Aslanidi G, Nash K, Shklyaev S, Campbell-Thompson M, Byrne BJ et al. Successful production of pseudotyped rAAV vectors using a modified baculovirus expression system. Mol Ther 2005; 12: 1217–1225.
Allen JM, Debelak DJ, Reynolds TC, Miller AD . Identification and elimination of replication-competent adeno-associated virus (AAV) that can arise by nonhomologous recombination during AAV vector production. J Virol 1997; 71: 6816–6822.
Ogasawara Y, Urabe M, Kogure K, Kume A, Colosi P, Kurtzman GJ et al. Efficient production of adeno-associated virus vectors using split-type helper plasmids. Jpn J Cancer Res 1999; 90: 476–483.
Rice SA, Knipe DM . Genetic evidence for two distinct transactivation functions of the herpes simplex virus α protein ICP27. J Virol 1990; 64: 1704–1715.
Knop DR, Harrell H . Bioreactor production of recombinant herpes simplex virus vectors. Biotechnol Prog 2007; 23: 715–721.
Li J, Samulski RJ, Xiao X . Role for highly regulated rep gene expression in adeno-associated virus vector production. J Virol 1997; 71: 5236–5243.
Ogasawara Y, Urabe M, Ozawa K . The use of heterologous promoters for adeno-associated virus (AAV) protein expression in AAV vector production. Microbiol Immunol 1998; 42: 177–185.
Mendelson E, Trempe JP, Carter BJ . Identification of the trans-acting rep proteins of adeno-associated virus by antibodies to a synthetic oligopeptide. J Virol 1986; 60: 823–832.
Trempe JP, Mendelson E, Carter BJ . Characterization of adeno-associated virus rep proteins in human cells by antibodies raised against rep expressed in Escherichia coli. Virology 1987; 161: 18–28.
Buller RML, Straus SE, Rose JA . Mechanism of host restriction of adenovirus-associated virus replication in African green monkey kidney cells. J Gen Virol 1970; 43: 663–672.
Collaco RF, Cao X, Trempe JP . A Helper virus-free packaging system for recombinant adeno-associated virus vectors. Gene 1999; 238: 397–405.
Ye G et al. A novel herpes simplex virus helper based production of adeno-associated virus vectors for treatment of retinal angiogenesis. Mol Ther 2006; 13 (Suppl 1s): 194 (Abstract 503).
Aubert M, Blaho JA . The herpes simplex virus type 1 regulatory protein ICP27 is required for the prevention of apoptosis in infected human cells. J Virol 1999; 73: 2803–2813.
Sanfilippo CM, Chirimuuta FNW, Blaho JA . Herpes simplex virus type 1 immediate-early gene expression is required for the induction of apoptosis in human epithelial HEp-2 cells. J Virol 2004; 78: 224–239.
Sanfilippo CM, Blaho JA . ICP0 gene expression is a herpes simplex virus type 1 apoptotic trigger. J Virol 2006; 80: 6810–6821.
DeFilippis RA, Goodwin EC, Wu L, DiMaio D . Endogenous human papillomavirus E6 and E7 proteins differentially regulate proliferation, senescence, and apoptosis in HeLa cervical carcinoma cells. J Virol 2003; 77: 1551–1563.
Conway JE . Herpes simplex virus type I based systems for the large-scale production of recombinant adeno-associated virus type-2 vectors. A dissertation. 2001, John Hopkins University.
Clark KR, Voulgaropoulou F, Johnson PR . A stable cell line carrying adenovirus-inducible rep and cap genes allows for infectivity titration of adeno-associated virus vectors. Gene Therapy 1996; 3: 1124–1132.
Zolotukhin S, Byrne BJ, Mason E, Zolotukhin I, Potter M, Chesnut K et al. Recombinant adeno-associated virus purification using novel methods improves infectious titer and yield. Gene Therapy 1999; 6: 973–985.
Acknowledgements
We thank Dr Barry Byrne (Department of Pediatrics), Irene Zolotukhin, Dr Nicholas Muzyczka, Dr Richard O Snyder and Dr Brian Cleaver (Molecular Genetics and Microbiology Department) of the University of Florida Powell Gene Therapy Center for technical discussions and helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, W., Wang, L., Harrell, H. et al. An efficient rHSV-based complementation system for the production of multiple rAAV vector serotypes. Gene Ther 16, 229–239 (2009). https://doi.org/10.1038/gt.2008.158
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2008.158
Keywords
This article is cited by
-
Engineered CHO cells as a novel AAV production platform for gene therapy delivery
Scientific Reports (2023)
-
Molecular design for recombinant adeno-associated virus (rAAV) vector production
Applied Microbiology and Biotechnology (2018)
-
Production of Recombinant Adeno-associated Virus Vectors Using Suspension HEK293 Cells and Continuous Harvest of Vector From the Culture Media for GMP FIX and FLT1 Clinical Vector
Molecular Therapy (2016)
-
Manufacturing of recombinant adeno-associated viral vectors for clinical trials
Molecular Therapy - Methods & Clinical Development (2016)
-
A scalable method for the production of high-titer and high-quality adeno-associated type 9 vectors using the HSV platform
Molecular Therapy - Methods & Clinical Development (2016)