Abstract
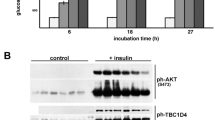
In adipocytes, insulin stimulates glucose transport primarily by promoting the translocation of GLUT4 to the plasma membrane. Requirements for Ca2+/ calmodulin during insulin-stimulated GLUT4 translocation have been demonstrated; however, the mechanism of action of Ca2+ in this process is unknown. Recently, myosin II, whose function in non-muscle cells is primarily regulated by phosphorylation of its regulatory light chain by the Ca2+/calmodulin-dependent myosin light chain kinase (MLCK), was implicated in insulin-stimulated GLUT4 translocation. The present studies in 3T3- F442A adipocytes demonstrate the novel finding that insulin significantly increases phosphorylation of the myosin II RLC in a Ca2+-dependent manner. In addition, ML-7, a selective inhibitor of MLCK, as well as inhibitors of myosin II, such as blebbistatin and 2,3-butanedione monoxime, block insulin- stimulated GLUT4 translocation and subsequent glucose transport. Our studies suggest that MLCK may be a regulatory target of Ca2+/calmodulin and may play an important role in insulin-stimulated glucose transport in adipocytes.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Choi, Y., Ryu, H., Kim, H. et al. Implication of phosphorylation of the myosin II regulatory light chain in insulin-stimulated GLUT4 translocation in 3T3-F442A adipocytes. Exp Mol Med 38, 180–189 (2006). https://doi.org/10.1038/emm.2006.22
Published:
Issue Date:
DOI: https://doi.org/10.1038/emm.2006.22
Keywords
This article is cited by
-
ALPK1 phosphorylates myosin IIA modulating TNF-α trafficking in gout flares
Scientific Reports (2016)
-
Rac-mediated actin remodeling and myosin II are involved in KATP channel trafficking in pancreatic β-cells
Experimental & Molecular Medicine (2015)
-
The proteomic signature of insulin-resistant human skeletal muscle reveals increased glycolytic and decreased mitochondrial enzymes
Diabetologia (2012)
-
Proteomic Analysis of Human Adipose Tissue After Rosiglitazone Treatment Shows Coordinated Changes to Promote Glucose Uptake
Obesity (2010)