Abstract
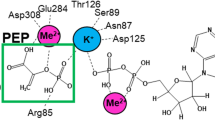
This review summarizes the recent developments on the regulation of human pyruvate dehydrogenase complex (PDC) by site-specific phosphorylation by four kinases. Mutagenic analysis of the three phosphorylation sites of human pyruvate dehydrogenase (E1) showed the site-independent mechanism of phosphorylation as well as site-independent dephosphorylation of the three phosphorylation sites and the importance of each phosphorylation site for the inactivation of E1. Both the negative charge and size of the group introduced at site 1 were involved in human E1 inactivation. Mechanism of inactivation of E1 was suggested to be site-specific. Phosphorylation of site 1 affected E1 interaction with the lipoyl domain of dihydrolipoamide acetyltransferase, whereas phosphorylation site 3 appeared to be closer to the thiamine pyrophosphate (TPP)-binding region affecting coenzyme interaction with human E1. Four isoenzymes of pyruvate dehydrogenase kinase (PDK) showed different specificity for the three phosphorylation sites of E1. All four PDKs phosphorylated sites 1 and 2 in PDC with different rates, and only PDK1 phosphorylated site 3. PDK2 was maximally stimulated by the reduction/acetylation of the lipoyl groups of E2. Presence of the multiple phosphorylation sites and isoenzymes of PDK is important for the tissue-specific regulation of PDC under different physiological conditions.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Patel, M., Korotchkina, L. Regulation of mammalian pyruvate dehydrogenase complex by phosphorylation: complexity of multiple phosphorylation sites and kinases. Exp Mol Med 33, 191–197 (2001). https://doi.org/10.1038/emm.2001.32
Published:
Issue Date:
DOI: https://doi.org/10.1038/emm.2001.32
Keywords
This article is cited by
-
SIRT5 functions as a tumor suppressor in renal cell carcinoma by reversing the Warburg effect
Journal of Translational Medicine (2021)
-
TGF-β1 is a regulator of the pyruvate dehydrogenase complex in fibroblasts
Scientific Reports (2020)
-
Functional Consequences of PDK4 Deficiency in Doberman Pinscher Fibroblasts
Scientific Reports (2020)
-
Real-time hyperpolarized 13C magnetic resonance detects increased pyruvate oxidation in pyruvate dehydrogenase kinase 2/4–double knockout mouse livers
Scientific Reports (2019)
-
Training state and fasting-induced PDH regulation in human skeletal muscle
Pflügers Archiv - European Journal of Physiology (2018)