Abstract
ASPM (known as Asp in fly and ASPM-1 in worm) is a microcephaly-associated protein family that regulates spindle architecture, but the underlying mechanism is poorly understood. Here, we show that ASPM forms a complex with another protein linked to microcephaly, the microtubule-severing ATPase katanin. ASPM and katanin localize to spindle poles in a mutually dependent manner and regulate spindle flux. X-ray crystallography revealed that the heterodimer formed by the N- and C-terminal domains of the katanin subunits p60 and p80, respectively, binds conserved motifs in ASPM. Reconstitution experiments demonstrated that ASPM autonomously tracks growing microtubule minus ends and inhibits their growth, while katanin decorates and bends both ends of dynamic microtubules and potentiates the minus-end blocking activity of ASPM. ASPM also binds along microtubules, recruits katanin and promotes katanin-mediated severing of dynamic microtubules. We propose that the ASPM–katanin complex controls microtubule disassembly at spindle poles and that misregulation of this process can lead to microcephaly.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
22 May 2017
In the original version of this Article, the name of author A. F. Maarten Altelaar was coded wrongly, resulting in it being incorrect when exported to citation databases. This has now been corrected, though no visible changes will be apparent.
References
Morris-Rosendahl, D. J. & Kaindl, A. M. What next-generation sequencing (NGS) technology has enabled us to learn about primary autosomal recessive microcephaly (MCPH). Mol. Cell Probes 29, 271–281 (2015).
Dammermann, A., Desai, A. & Oegema, K. The minus end in sight. Curr. Biol. 13, R614–R624 (2003).
Rogers, G. C., Rogers, S. L. & Sharp, D. J. Spindle microtubules in flux. J. Cell Sci. 118, 1105–1116 (2005).
Saunders, R. D., Avides, M. C., Howard, T., Gonzalez, C. & Glover, D. M. The Drosophila gene abnormal spindle encodes a novel microtubule-associated protein that associates with the polar regions of the mitotic spindle. J. Cell Biol. 137, 881–890 (1997).
Wakefield, J. G., Bonaccorsi, S. & Gatti, M. The Drosophila protein asp is involved in microtubule organization during spindle formation and cytokinesis. J. Cell Biol. 153, 637–648 (2001).
Fish, J. L., Kosodo, Y., Enard, W., Paabo, S. & Huttner, W. B. Aspm specifically maintains symmetric proliferative divisions of neuroepithelial cells. Proc. Natl Acad. Sci. USA 103, 10438–10443 (2006).
van der Voet, M. et al. NuMA-related LIN-5, ASPM-1, calmodulin and dynein promote meiotic spindle rotation independently of cortical LIN-5/GPR/Gα. Nat. Cell Biol. 11, 269–277 (2009).
Higgins, J. et al. Human ASPM participates in spindle organisation, spindle orientation and cytokinesis. BMC Cell Biol. 11, 85 (2010).
Ito, A. & Goshima, G. Microcephaly protein Asp focuses the minus ends of spindle microtubules at the pole and within the spindle. J. Cell Biol. 211, 999–1009 (2015).
Schoborg, T., Zajac, A. L., Fagerstrom, C. J., Guillen, R. X. & Rusan, N. M. An Asp–CaM complex is required for centrosome-pole cohesion and centrosome inheritance in neural stem cells. J. Cell Biol. 211, 987–998 (2015).
Ripoll, P., Pimpinelli, S., Valdivia, M. M. & Avila, J. A cell division mutant of Drosophila with a functionally abnormal spindle. Cell 41, 907–912 (1985).
do Carmo Avides, M. & Glover, D. M. Abnormal spindle protein, Asp, and the integrity of mitotic centrosomal microtubule organizing centers. Science 283, 1733–1735 (1999).
Morales-Mulia, S. & Scholey, J. M. Spindle pole organization in Drosophila S2 cells by dynein, abnormal spindle protein (Asp), and KLP10A. Mol. Biol. Cell 16, 3176–3186 (2005).
Bond, J. et al. ASPM is a major determinant of cerebral cortical size. Nat. Genet. 32, 316–320 (2002).
Roll-Mecak, A. & McNally, F. J. Microtubule-severing enzymes. Curr. Opin. Cell Biol. 22, 96–103 (2010).
McNally, F. J. & Vale, R. D. Identification of katanin, an ATPase that severs and disassembles stable microtubules. Cell 75, 419–429 (1993).
Hartman, J. J. et al. Katanin, a microtubule-severing protein, is a novel AAA ATPase that targets to the centrosome using a WD40-containing subunit. Cell 93, 277–287 (1998).
Mishra-Gorur, K. et al. Mutations in KATNB1 cause complex cerebral malformations by disrupting asymmetrically dividing neural progenitors. Neuron 84, 1226–1239 (2014).
Hu, W. F. et al. Katanin p80 regulates human cortical development by limiting centriole and cilia number. Neuron 84, 1240–1257 (2014).
McNally, K. P., Bazirgan, O. A. & McNally, F. J. Two domains of p80 katanin regulate microtubule severing and spindle pole targeting by p60 katanin. J. Cell Sci. 113, 1623–1633 (2000).
Srayko, M., Buster, D. W., Bazirgan, O. A., McNally, F. J. & Mains, P. E. MEI-1/MEI-2 katanin-like microtubule severing activity is required for Caenorhabditis elegans meiosis. Genes Dev. 14, 1072–1084 (2000).
McNally, K., Audhya, A., Oegema, K. & McNally, F. J. Katanin controls mitotic and meiotic spindle length. J. Cell Biol. 175, 881–891 (2006).
McNally, K. P. & McNally, F. J. The spindle assembly function of Caenorhabditis elegans katanin does not require microtubule-severing activity. Mol. Biol. Cell 22, 1550–1560 (2011).
McNally, K. et al. Katanin maintains meiotic metaphase chromosome alignment and spindle structure in vivo and has multiple effects on microtubules in vitro. Mol. Biol. Cell 25, 1037–1049 (2014).
Loughlin, R., Wilbur, J. D., McNally, F. J., Nedelec, F. J. & Heald, R. Katanin contributes to interspecies spindle length scaling in Xenopus. Cell 147, 1397–1407 (2011).
Bailey, M. E., Sackett, D. L. & Ross, J. L. Katanin severing and binding microtubules are inhibited by tubulin carboxy tails. Biophys. J. 109, 2546–2561 (2015).
Cheung, K. et al. Proteomic analysis of the mammalian katanin family of microtubule-severing enzymes defines KATNBL1 as a regulator of mammalian katanin microtubule-severing. Mol. Cell Proteomics 15, 1658–1669 (2016).
Elting, M. W., Hueschen, C. L., Udy, D. B. & Dumont, S. Force on spindle microtubule minus ends moves chromosomes. J. Cell Biol. 206, 245–256 (2014).
Sikirzhytski, V. et al. Direct kinetochore-spindle pole connections are not required for chromosome segregation. J. Cell Biol. 206, 231–243 (2014).
Gai, M. et al. ASPM and CITK regulate spindle orientation by affecting the dynamics of astral microtubules. EMBO Rep. 17, 1396–1409 (2016).
Zhang, D., Rogers, G. C., Buster, D. W. & Sharp, D. J. Three microtubule severing enzymes contribute to the ‘Pacman-flux’ machinery that moves chromosomes. J. Cell Biol. 177, 231–242 (2007).
Ganem, N. J., Upton, K. & Compton, D. A. Efficient mitosis in human cells lacking poleward microtubule flux. Curr. Biol. 15, 1827–1832 (2005).
Iwaya, N. et al. A common substrate recognition mode conserved between katanin p60 and VPS4 governs microtubule severing and membrane skeleton reorganization. J. Biol. Chem. 285, 16822–16829 (2010).
Jiang, K. et al. Microtubule minus-end stabilization by polymerization-driven CAMSAP deposition. Dev. Cell 28, 295–309 (2014).
Brouhard, G. J. & Rice, L. M. The contribution of αβ-tubulin curvature to microtubule dynamics. J. Cell Biol. 207, 323–334 (2014).
Janosi, I. M., Chretien, D. & Flyvbjerg, H. Modeling elastic properties of microtubule tips and walls. Eur. Biophys. J. 27, 501–513 (1998).
Wang, H., Brust-Mascher, I., Civelekoglu-Scholey, G. & Scholey, J. M. Patronin mediates a switch from kinesin-13-dependent poleward flux to anaphase B spindle elongation. J. Cell Biol. 203, 35–46 (2013).
Rogers, G. C. et al. Two mitotic kinesins cooperate to drive sister chromatid separation during anaphase. Nature 427, 364–370 (2004).
Gaetz, J. & Kapoor, T. M. Dynein/dynactin regulate metaphase spindle length by targeting depolymerizing activities to spindle poles. J. Cell Biol. 166, 465–471 (2004).
Desai, A., Verma, S., Mitchison, T. J. & Walczak, C. E. Kin I kinesins are microtubule-destabilizing enzymes. Cell 96, 69–78 (1999).
Montenegro Gouveia, S. et al. In vitro reconstitution of the functional interplay between MCAK and EB3 at microtubule plus ends. Curr. Biol. 20, 1717–1722 (2010).
Tanenbaum, M. E. et al. A complex of Kif18b and MCAK promotes microtubule depolymerization and is negatively regulated by Aurora kinases. Curr. Biol. 21, 1356–1365 (2011).
Zhang, D. et al. Drosophila katanin is a microtubule depolymerase that regulates cortical-microtubule plus-end interactions and cell migration. Nat. Cell Biol. 13, 361–370 (2011).
Taverna, E., Gotz, M. & Huttner, W. B. The cell biology of neurogenesis: toward an understanding of the development and evolution of the neocortex. Annu. Rev. Cell Dev. Biol. 30, 465–502 (2014).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Hockemeyer, D. et al. Efficient targeting of expressed and silent genes in human ESCs and iPSCs using zinc-finger nucleases. Nat. Biotechnol. 27, 851–857 (2009).
Scholz, J., Besir, H., Strasser, C. & Suppmann, S. A new method to customize protein expression vectors for fast, efficient and background free parallel cloning. BMC Biotechnol. 13, 12 (2013).
Schuck, P. Size-distribution analysis of macromolecules by sedimentation velocity ultracentrifugation and lamm equation modeling. Biophys. J. 78, 1606–1619 (2000).
Kabsch, W. Xds. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Adams, P. D. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Orchard, S. et al. The MIntAct project–IntAct as a common curation platform for 11 molecular interaction databases. Nucleic Acids Res. 42, D358–D363 (2014).
Acknowledgements
We thank A. Prota for the help in refining the crystal structures, M. Boxem, C. Janke and M. Mikhaylova for the gift of materials, and the beamline scientists at beamlines X06DA of the Swiss Light Source (Paul Scherrer Institut, Villigen, Switzerland) for technical assistance with the X-ray data collection. This work was supported by the European Research Council Synergy grant 609822 and Netherlands Organization for Scientific Research (NWO) CW ECHO grant (711.011.005) to A.A., the EMBO long-term and Marie Curie IEF fellowships to L.R., grants from the Swiss National Science Foundation (31003A_166608 to M.O.S. and 31003A_163449 to R.A.K.), a NWO VIDI grant (723.012.102) to A.F.M.A. and as part of the National Roadmap Large-scale Research Facilities of the Netherlands (project number 184.032.201) to A.F.M.A. and A.J.R.H. The structural data reported in this paper are available in PDB (PDB code 5LB7).
Author information
Authors and Affiliations
Contributions
K.J., L.R., S.H., R.A.K., M.O.S. and A.A. designed experiments, analysed data and wrote the paper. A.A. coordinated the project. K.J. and S.H. performed cellular and in vitro reconstitution experiments. L.R. performed biophysical experiments. L.R. and G.C. performed crystallography experiments. Q.L., A.F.M.A. and A.J.R.H. performed and analysed mass spectrometry experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Characterization of ASPM knock-in cell lines.
(a) Genotyping of GFP and strep-GFP-ASPM knock-in cells used in this study. (b) Immunostaining for α-tubulin and DNA (DAPI) in GFP-ASPM knock-in HeLa cells during mitosis. White arrows indicate the localization of ASPM to the microtubule minus ends in the central spindle in telophase. Scale bar, 5 μm.
Supplementary Figure 2 ASPM and katanin regulate spindle architecture.
(a) western blotting with the indicated antibodies in control, ASPM knockout and p80 knockout cells (left, HeLa; right, U2OS PA-GFP-α-tubulin). Note that the knockout of p80 caused the concomitant loss of p60, in agreement with the fact that the two subunits form a tight complex. (b) Sequencing results of ASPM knockout cell lines used in this study. 1 nt insertion will result in p.N71KfsX31 or p.E72RfsX30. (c–d) Immunostaining of γ-tubulin, DNA (DAPI), and NuMA (c) or p150Glued (d) in control, ASPM knockout, and p80 knockout HeLa cells. Scale bar, 5 μm. In HeLa cells, the intensity of NuMA and p150Glued at the spindle pole remained largely unchanged in both ASPM and p80 knockout cells. (e) Quantification of spindle pole intensities of NuMA, and p150Glued as shown in panels c, d. For NuMA intensity, n = 164 spindle poles, control; n = 172, ASPM knockout; n = 176, p80 knockout. For p150Glued intensity, n = 160, control; n = 166, ASPM knockout; n = 170, p80 knockout. (f,g) Immunostaining for α-tubulin, CEP135 and DNA in control, ASPM knockout and p80 knockout HeLa cells (f, bipolar spindles with two centrosomes; g, bipolar and multipolar spindles with >2 centrosomes in p80 knockout cells). Scale bar, 5 μm. (h) Quantification of the distance between two centrosomes in control, ASPM knockout and p80 knockout HeLa cells as shown in f. n = 67 cells, control; n = 61, ASPM knockout, n = 90, p80 knockout. (i) Quantification of centrosome numbers as shown in f,g. 470 cells, control; 469, ASPM knockout; 572, p80 knockout (2 experiments). (j) Quantification of the average distance between two spindle poles in photoactivation experiments in U2OS cells as shown in Fig. 2e. n = 26 cells, control; n = 29, ASPM knockout; n = 26, p80 knockout. The standard deviations of the distance between the two poles during imaging were small (∼0.3 μm), which means that the spindle length kept constant during imaging. Compared to control, spindle length in ASPM and p80 knockout HeLa (h) and U2OS (j) cells shows 6 ∼ 10% reduction. Data represent mean ± s.d.∗, P < 0.05, ∗∗∗, P < 0.001, Mann-Whitney U test. Unprocessed original scans of blots are shown in Supplementary Fig. 8. Source data for panels e,h,i and j can be found in Supplementary Table 5.
Supplementary Figure 3 The interaction between ASPM and katanin requires a conserved repeat sequence of ASPM and the p60N/p80C heterodimer.
(a) Alignment of katanin-binding linear motifs of ASPM from several vertebrate species. Note the complete conservation of the phenylalanine residue corresponding to F352 in the third repeat of mouse ASPM (red arrowhead below the alignment). (b) Streptavidin pull down assays with extracts of HEK293T cells expressing Biotinylation tagged (Bio)-GFP-tagged wild-type (WT) p80 or its indicated mutants together with GFP-tagged WT p60 or the indicated mutants. None of the analyzed mutations perturbed the p60–p80 interaction. See also Supplementary Fig. 8e. (c) Electron density maps of ASPMp in the p60N/p80C/ASPMp complex structure. Only residues S351, F352 and L353 of the ASPMp peptide (LSPDSFLND, residues 347–355 of mouse ASPM) are visible. The SigmaA-weighted 2mFo-DFc (left) and mFo-DFc (right) omit maps (green mash) are contoured at +1.0σ and +3.0σ, respectively.
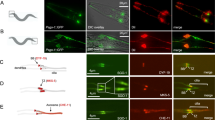
Supplementary Figure 4 The CH1-CH3 fragment of C. elegans ASPM-1 binds microtubule minus-end in cells.
(a,b) The worm ASPM-1 CH1–CH3 fragment associates with minus ends of free microtubules (a) or minus ends freshly generated by photoablation (b) in interphase MRC5 cells. Green lightning bolts indicate the sites of photoablation. Scale bars, 2 μm.
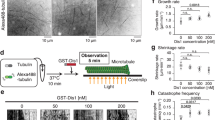
Supplementary Figure 5 The katanin p60/p80 heterodimer decorates and bends microtubule ends.
(a) TIRFM live cell imaging and kymographs of colocalization of GFP-p60/p80C and EB3-TagRFP in MRC5 cells. Scale bars: horizontal, 2 μm; vertical, 10 s. (b) Live cell images of a single microtubule plus end bound to GFP-p60/p80C and EB3-TagRFP in MRC5 cells. White arrow, breaking of a bent microtubule end. Scale bar, 1 μm. (c) Live imaging of MRC5 cells expressing individual katanin subunits together with EB3-TagRFP. Scale bar, 2 μm. (d) TIRFM time lapse images showing the complete severing of dynamic microtubules by p60/SNAP-Alexa647-p80C at 750 nM in a flow-in experiment. Time represented as min:sec. Scale bar, 2 μm. (e–f) Maximum intensity projections of time lapse images of GFP-p60/p80 WT or mutants in HeLa cells. Imaging was performed using TIRFM with 500 ms exposure in a stream mode. 100–200 images representing consecutive frames were used to make projections. Mutations of R615A, K618A, G607A, V608A, D609A and I610A in p80 completely abolish the end-binding activity of p60/p80 in cells. Scale bar, 2 μm. (g) Protein sequence alignment of p60N and p80C from Caenorhabditis elegans, Chlamydomonas reinhardtii, Tetrahymena thermophila, Arabidopsis thaliana, Drosophila melanogaster , Danio rerio, Xenopus tropicalis, Homo sapiens and Mus musculus. The residues that are essential for microtubule end binding are indicated with red dots. Mutating charged residues denoted by black dots to alanine had no effect on end binding in vivo. Red triangles indicate the residues interacting with ASPM, which are conserved in vertebrates (red dashed rectangle). Numbers displayed on top of the sequence alignment are based on mouse katanin sequences.
Supplementary Figure 6 Characterization of wild-type katanin p60/p80C and its mutants, ASPM mutant and the ASPM–katanin complex at different concentrations.
(a) The p60/p80C R615A mutant does not bind microtubule ends at 25 or 75 nM. Time represented as min:sec. Scale bars, 2 μm. (b) The p60/p80C Y574A mutant can decorate, bend and break microtubule ends at 75 nM but not at 25 nM. White arrow, breaking of a bent microtubule end. Time represented as min:sec. Scale bars, 2 μm. (c) Wild-type (WT) p60/p80C and the indicated mutants can efficiently sever GMPCPP-stabilized microtubules at 75 nM but not at 25 nM. See Fig. 7a for quantification. Scale bar, 2 μm. (d–g) The p60/p80C R615A mutant but not the Y574A mutant at 25 nM can be recruited onto microtubule lattice and perform severing in the assay with dynamic microtubules in the presence of 30 nM ASPM. White arrows indicate the sites where severing events occur. Time represented as min:sec. f, n = 64 (WT), 59 (R615A) and 59 (Y574A) microtubules; g, Data are the mean of 2 experiments. Scale bar, 2 μm. (h–j) 30 nM ASPM F302A/F377A mutant does not recruit p60/p80C to microtubule lattice and does not promote severing. i, n = 62 (WT) and 60 (F302A/F377A) microtubules; j, data are the mean of 2 experiments. Severing frequency data for WT in panels g and j were replotted from Fig. 7f (30 nM). (k–m) 5 nM ASPM F302A/F377A mutant does not recruit p60/p80C to dynamic microtubule minus ends and does not induce their blocking. l, n = 19 (WT) and 29 (F302A/F377A) microtubules; m, n = 19 (WT) and 33 (F302A/F377A) microtubules. Scale bars, horizontal, 2 μm; vertical, 1 min. Data represent mean ± s.d. Source data for panels f, g, i, j, l and m can be found in Supplementary Table 5.
Supplementary Figure 7 Purified proteins used for in vitro reconstitution experiments.
(a) Coomassie blue stained gels with strep-GFP tagged ASPM, Asp and ASPM-1 proteins purified from transiently transfected HEK293T cells. The bands corresponding to purified full-length proteins are marked with red asterisks. (b) Coomassie blue stained gels with p60/strep-SNAP-p80C wild-type and mutants, p60N + L/strep-GFP-p80C and p60N + L/strep-SNAP-p80C purified from HEK293T cells and strep-p60/6xHis-GFP-p80C purified from E. coli.
Supplementary information
Supplementary Information
Supplementary Information (PDF 6176 kb)
Supplementary Table 1
Supplementary Information (XLSX 9 kb)
Supplementary Table 2
Supplementary Information (XLSX 14 kb)
Supplementary Table 3
Supplementary Information (XLSX 13 kb)
Supplementary Table 4
Supplementary Information (XLSX 130 kb)
Supplementary Table 5
Supplementary Information (XLSX 106 kb)
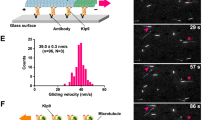
Reduced spindle flux in ASPM and katanin knockout cells.
Photoactivation of PA-GFP-α-tubulin in control (left), ASPM knockout (middle) and katanin knockout (right) U2OS metaphase cells. Images were collected using a spinning disk microscope at 2 s interval. Video is sped up 60 times. Time is shown in the format min:sec. (MOV 5999 kb)
Overexpressed katanin p60/p80 complex tracks and bends microtubule ends in cells.
MRC5 cells were co-transfected with GFP-p60, p80 (dark) and EB3-TagRFP. Images were collected using a TIRF microscope in stream mode (2 frames/s). Video is sped up 15 times. Time is shown in the format min:sec. (MOV 1481 kb)
Katanin p60/p80C severs dynamic microtubules at 300 nM in vitro.
Microtubules were first polymerized in the tubulin polymerization reaction mix (20 μM unlabeled tubulin and 0.5 μM X-rhodamine-tubulin in MRB80 buffer supplemented with 50 mM KCl). Subsequently, the tubulin polymerization reaction mix supplemented with 300 nM p60/SNAP-Alexa647-p80C and 1 mM ATP was flowed into the reaction chamber. Images were collected using a TIRF microscope at a 3 s interval. Video is sped up 15 times. Time is shown in the format min:sec. (MOV 3936 kb)
Katanin p60N + L/p80C bends and breaks a microtubule end in vitro.
Microtubules were polymerized in the presence of 1 μM p60N + L/GFP-p80C, 20 μM unlabeled tubulin and 0.5 μM X-rhodamine-tubulin in MRB80 buffer supplemented with 50 mM KCl. Images were collected using a TIRF microscope at 2 s interval. Video is sped up 30 times. Time is shown in the format min:sec. (MOV 181 kb)
30 nM ASPM and 25 nM katanin p60/p80C sever dynamic microtubules in vitro.
Microtubules were first polymerized in the tubulin polymerization reaction mix (20 μM unlabeled tubulin and 0.5 μM X-rhodamine-tubulin in MRB80 buffer supplemented with 50 mM KCl). Subsequently, tubulin polymerization reaction mix supplemented with 30 nM ASPM, 25 nM p60/SNAP-Alexa647-p80C and 1 mM ATP was flowed into the reaction chamber. Images were collected using a TIRF microscope at a 3 s interval. Video is sped up 15 times. Time is shown in the format min:sec. (MOV 3526 kb)
Rights and permissions
About this article
Cite this article
Jiang, K., Rezabkova, L., Hua, S. et al. Microtubule minus-end regulation at spindle poles by an ASPM–katanin complex. Nat Cell Biol 19, 480–492 (2017). https://doi.org/10.1038/ncb3511
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3511
This article is cited by
-
Microtubule polarity determines the lineage of embryonic neural precursor in zebrafish spinal cord
Communications Biology (2024)
-
CAMSAPs and nucleation-promoting factors control microtubule release from γ-TuRC
Nature Cell Biology (2024)
-
Mechanisms of microtubule organization in differentiated animal cells
Nature Reviews Molecular Cell Biology (2022)
-
Defective replication stress response linked to microcephaly
Genome Instability & Disease (2022)
-
Identification of potential markers for differentiating epithelial ovarian cancer from ovarian low malignant potential tumors through integrated bioinformatics analysis
Journal of Ovarian Research (2021)