Abstract
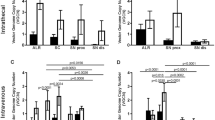
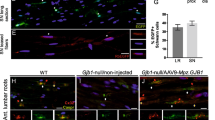
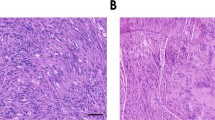
Schwannomas are benign tumors forming along peripheral nerves that can cause deafness, pain and paralysis. Current treatment involves surgical resection, which can damage associated nerves. To achieve tumor regression without damage to nerve fibers, we generated an HSV amplicon vector in which the apoptosis-inducing enzyme, caspase-1 (ICE), was placed under the Schwann cell-specific P0 promoter. Infection of schwannoma, neuroblastoma and fibroblastic cells in culture with ICE under the P0 promoter showed selective toxicity to schwannoma cells, while ICE under a constitutive promoter was toxic to all cell types. After direct intratumoral injection of the P0-ICE amplicon vector, we achieved marked regression of schwannoma tumors in an experimental xenograft mouse model. Injection of this amplicon vector into the sciatic nerve produced no apparent injury to the associated dorsal root ganglia neurons or myelinated nerve fibers. The P0-ICE amplicon vector provides a potential means of ‘knifeless resection’ of schwannoma tumors by injection of the vector into the tumor with low risk of damage to associated nerve fibers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- CCD:
-
charged-coupling device
- CMV:
-
cytomegalovirus
- CNS:
-
central nervous system
- CV:
-
conduction velocity
- DRG:
-
dorsal root ganglia
- EGFR:
-
epidermal growth factor receptor
- FBS:
-
fetal bovine serum
- Fluc:
-
firefly luciferase
- GFP:
-
green fluorescent protein
- HSV:
-
herpes simplex virus type 1
- ICE:
-
caspase-1
- MCS:
-
multiple cloning site
- MOI:
-
multiplicity of infection (tu per cell)
- NF2:
-
neurofibromatosis type 2
- PBS:
-
phosphate buffered saline
- PNS:
-
peripheral nervous system
- tu:
-
transducing units
References
Baser ME, Evans DG, Gutmann DH . Neurofibromatosis 2. Curr Opin Neurol 2003; 16: 27–33.
McClatchey AI . Neurofibromatosis. Annu Rev Pathol 2007; 2: 191–216.
MacCollin M, Chiocca EA, Evans DG, Friedman JM, Horvitz R, Jaramillo D et al. Diagnostic criteria for schwannomatosis. Neurology 2005; 64: 1838–1845.
Gutmann DH, Giovannini M . Mouse models of neurofibromatosis 1 and 2. Neoplasia 2002; 4: 279–290.
Messerli SM, Prabhakar S, Tang Y, Mahmood U, Weissleder R, Giovannini M et al. Treatment of schwannomas with an oncolytic HSV recombinant virus in murine models of neurofibromatosis type 2. Hum Gene Ther 2006; 17: 20–30.
Prabhakar S, Messerli SM, Stemmer-Rachamimov AO, Liu TC, Rabkin S, Martuza R et al. Treatment of implantable NF2 schwannoma tumor models with oncolytic herpes simplex virus G47Delta. Cancer Gene Ther 2007; 14: 460–467.
Mahller YY, Vaikunth SS, Currier MA, Miller SJ, Ripberger MC, Hsu YH et al. Oncolytic HSV and erlotinib inhibit tumor growth and angiogenesis in a novel malignant peripheral nerve sheath tumor xenograft model. Mol Ther 2007; 15: 279–286.
Oehmig A, Fraefel C, Breakefield XO . Update of herpesvirus amplicon vectors. Mol Ther 2004; 10: 630–643.
Aboody-Guterman K, Pechan P, Rainov NG, Sena-Esteves M, Jacobs A, Snyder E et al. Green fluorescent protein as a reporter for retrovirus and helper virus-free HSV-1 amplicon vector-mediated gene transfer into neural cells in culture and in vivo. Neuroreport 1997; 8: 3801–3808.
Grandi P, Bein K, Hadjipanayis CG, Wolfe D, Breakefield XO, Glorioso JC . Application of HSV-1 vectors to the treatment of cancer. In: Harrington D, Vile RG, Panda H (eds). Viral Therapy of Cancer. Wiley Publishers, 2008. Chapter 2, pp 2.1–2.8.
Shah K, Breakefield X . HSV amplicon vectors for cancer therapy. Curr Gene Ther 2006; 6: 361–370.
Grandi P, Wang S, Schuback D, Krasnykh V, Spear M, Curiel DT et al. HSV-1 virions engineered for specific binding to cell surface receptors. Mol Ther 2004; 9: 419–427.
Hung G, Li X, Faudoa R, Xeu Z, Kluwe L, Rhim JS et al. Establishment and characterization of a schwannoma cell line from a patient with neurofibromatosis 2. Int J Oncol 2002; 20: 475–482.
Seijffers R, Allchorne AJ, Woolf CJ . The transcription factor ATF-3 promotes neurite outgrowth. Mol Cell Neurosci 2006; 32: 143–154.
Sadee W, Yu VC, Richards ML, Preis PN, Schwab MR, Brodsky FM et al. Expression of neurotransmitter receptors and myc protooncogenes in subclones of a human neuroblastoma cell line. Cancer Res 1987; 47: 5207–5212.
Hewett J, Gonzalez-Agosti C, Slater D, Ziefer P, Li S, Bergeron D et al. Mutant torsinA, responsible for early-onset torsion dystonia, forms membrane inclusions in cultured neural cells. Hum Mol Genet 2000; 9: 1403–1413.
Ikeda K, Saeki Y, Gonzalez-Agosti C, Ramesh V, Chiocca EA . Inhibition of NF2-negative and NF2-positive primary human meningioma cell proliferation by overexpression of merlin due to vector-mediated gene transfer. J Neurosurg 1999; 91: 85–92.
Shah K, Tang Y, Breakefield X, Weissleder R . Real-time imaging of TRAIL-induced apoptosis of glioma tumors in vivo. Oncogene 2003; 22: 6865–6872.
Brown AM, Lemke G . Multiple regulatory elements control transcription of the peripheral myelin protein zero gene. J Biol Chem 1997; 272: 28939–28947.
Jung Y, Miura M, Yuan J . Suppression of interleukin-1 beta-converting enzyme-mediated cell death by insulin-like growth factor. J Biol Chem 1996; 271: 5112–5117.
Saeki Y, Breakefield XO, Chiocca EA . Improved HSV-1 amplicon packaging system using ICP27-deleted, oversized HSV-1 BAC DNA. In: Machida CA (ed). Viral Vectors for Gene Therapy, Methods and Protocols. Humana Press: New Jersey, 2003. vol 76, pp 51–60.
Sena-Esteves M, Tebbets JC, Steffens S, Crombleholme T, Flake AW . Optimized large-scale production of high titer lentivirus vector pseudotypes. J Virol Methods 2004; 122: 131–139.
Rizzo MA, Springer GH, Granada B, Piston DW . An improved cyan fluorescent protein variant useful for FRET. Nat Biotechnol 2004; 22: 445–449.
Galve-Roperh I, Sanchez C, Cortes ML, del Pulgar TG, Izquierdo M, Guzman M . Anti-tumoral action of cannabinoids: involvement of sustained ceramide accumulation and extracellular signal-regulated kinase activation. Nat Methods 2000; 6: 313–310.
Tannous BA, Kim DE, Fernandez JL, Weissleder R, Breakefield XO . Codon-optimized Gaussia luciferase cDNA for mammalian gene expression in culture and in vivo. Mol Ther 2005; 11: 435–443.
Messerli SM, Tang Y, Giovannini M, Bronson R, Weissleder R, Breakefield XO . Detection of spontaneous schwannomas by MRI in a transgenic murine model of neurofibromatosis type 2. Neoplasia 2002; 4: 501–509.
Gold MS, Weinreich D, Kim CS, Wang R, Treanor J, Porreca F et al. Redistribution of Na(V) 1.8 in uninjured axons enables neuropathic pain. J Neurosci 2003; 23: 158–166.
Baba H, Doubell JP, Woolf CJ . Peripheral inflammation facilitates Abeta fiber-mediated synaptic input to the substantia gelatinosa of the adult rat spinal cord. J Neurosci 1999; 19: 859–867.
Deguen B, Mérel P, Goutebroze L, Giovannini M, Reggio H, Arpin M et al. Impaired interaction of naturally occurring mutant NF2 protein with actin-based cytoskeleton and membrane. Hum Mol Genet 1998; 7: 217–226.
Giovannini M, Robanus-Maandag E, Niwa-Kawakita M, van der Valk M, Woodruff JM, Goutebroze L et al. Schwann cell hyperplasia and tumors in transgenic mice expressing a naturally occurring mutant NF2 protein. Genes Dev 1999; 13: 978–986.
Giovannini M, Robanus-Maandag E, van der Valk M, Niwa-Kawakita M, Abramowski V, Goutebroze L et al. Conditional biallelic Nf2 mutation in the mouse promotes manifestations of human neurofibromatosis type 2. Genes Dev 2000; 14: 1617–1630.
Kalamarides M, Niwa-Kawakita M, Leblois H, Abramowski V, Perricaudet M, Janin A et al. Nf2 gene inactivation in arachnoidal cells is rate-limiting for meningioma development in the mouse. Genes Dev 2002; 16: 1060–1065.
Stidham KR, Roberson JB . Human vestibular schwannoma growth in the nude mouse: evaluation of a modified subcutaneous implantation model. Am J Otol 1997; 18: 622–626.
Todo T, Martuza RL, Rabkin SD, Johnson PA . Oncolytic herpes simplex virus vector with enhanced MHC class I presentation and tumor cell killing. Proc Natl Acad Sci USA 2001; 98: 6396–6401.
Plotkin SR, Singh MA, O'Donnell CC, Harris GJ, McClatchey AI, Halpin C . Audiologic and radiographic response of NF2-related vestibular schwannoma to erlotinib therapy. Nat Clin Pract Oncol 2008; 5: 487–491.
Saydam O, Senol O, Tannous BA, Prabhakar S, Stemmer-Rachamimov AO, Breakefield XO et al. A novel imaging-compatible sciatic nerve schwannoma model, submitted.
Markert JM, Medlock MD, Rabkin SD, Gillespie GY, Todo T, Hunter WD et al. Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a phase I trial. Gene Ther 2000; 7: 867–874.
Hu JC, Coffin RS, Davis CJ, Graham NJ, Groves N, Guest PJ et al. A phase I study of OncoVEXGM-CSF, a second-generation oncolytic herpes simplex virus expressing granulocyte macrophage colony-stimulating factor. Clin Cancer Res 2006; 12: 6737–6747.
Kemeny N, Brown K, Covey A, Kim T, Bhargava A, Brody L et al. Phase I, open-label, dose-escalating study of a genetically engineered herpes simplex virus, NV1020, in subjects with metastatic colorectal carcinoma to the liver. Hum Gene Ther 2006; 17: 1214–1224.
Bragg DC, Camp SM, Kaufman CA, Wilbur JD, Boston H, Schuback DE et al. Perinuclear biogenesis of mutant torsin-A inclusions in cultured cells infected with tetracycline-regulated herpes simplex virus type 1 amplicon vectors. Neuroscience 2004; 125: 651–661.
Wang GY, Ho IA, Sia KC, Miao L, Hui KM, Lam PY . Engineering an improved cell cycle-regulatable herpes simplex virus type 1 amplicon vector with enhanced transgene expression in proliferating cells yet attenuated activities in resting cells. Hum Gene Ther 2007; 18: 222–231.
Reinblatt M, Pin RH, Federoff HJ, Fong Y . Utilizing tumor hypoxia to enhance oncolytic viral therapy in colorectal metastases. Ann Surg 2004; 239: 892–899.
Ho IA, Hui KM, Lam PY . Glioma-specific and cell cycle-regulated herpes simplex virus type 1 amplicon viral vector. Hum Gene Ther 2004; 15: 495–508.
Wang S, Vos J . A hybrid herpesvirus infectious vector based on Epstein-Barr virus and herpes simplex virus type 1 for gene transfer into human cells in vitro and in vivo. J Virol 1996; 70: 8422–8430.
Acknowledgements
We thank Ms. Suzanne McDavitt for skilled editorial assistance. This work was supported by grants from NINDS NS024279, Department of the Army, US Army Research Medical Research and Material Command Award W81XWH-04-1-0237, the P41 RR001395 NCRR grant, the Terrill Family and Texas Neurofibromatosis Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prabhakar, S., Brenner, G., Sung, B. et al. Imaging and therapy of experimental schwannomas using HSV amplicon vector-encoding apoptotic protein under Schwann cell promoter. Cancer Gene Ther 17, 266–274 (2010). https://doi.org/10.1038/cgt.2009.71
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2009.71
Keywords
This article is cited by
-
Schwannoma gene therapy by adeno-associated virus delivery of the pore-forming protein Gasdermin-D
Cancer Gene Therapy (2019)
-
Genetically Engineered Microvesicles Carrying Suicide mRNA/Protein Inhibit Schwannoma Tumor Growth
Molecular Therapy (2013)