Abstract
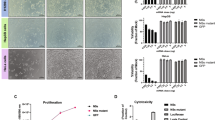
Although reovirus has been used in tests as a potential cancer therapeutic agent against a variety of cancer cells, its application to hepatocellular carcinoma cells, in which the hepatitis B virus (HBV) X (HBX) protein of HBV plays a primary role, has not yet been explored. Here, we describe experiments in which we use reovirus to treat Chang liver carcinoma cells expressing either a vector only (Chang-vec) or a vector encoding HBX protein (Chang-HBX). Although Chang-vec cells readily support reoviral proliferation and undergo apoptosis, Chang-HBX cells are highly resistant to reoviral infection and virus-induced apoptosis, even though HBX protein induces activation of Ras and inactivation of PKR, which are normally thought to enhance reoviral oncolysis. The resistance of Chang-HBX cells to reovirus may instead be explained by HBX-induced downregulation of death receptor 5 and activation of Stat1. Phosphorylated Stat1 activates interferon (IFN)-stimulated regulatory element (ISRE)- and IFN-γ-activated sequence (GAS)-mediated transcription, leading to the production of IFN-β, whereas the reduced expression of Stat1 with its siRNA results in a decrease in IFN-β production, by which Chang-HBX cells eventually succumb to reovirus infection. This result further indicates that HBX induces the establishment of an antiviral state through Stat1 activation. Thus, it appears that active Ras does not override the antiviral effect mediated by the activation of Stat1. Accordingly, we report that HBX, an oncoprotein of HBV, can prevent reoviral oncolysis of hepatocellular carcinoma. This suggests there may be limits to the practical application of reovirus in the treatment of human cancers already expressing other oncoviral proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Norman KL, Hirasawa K, Yang AD, Shields MA, Lee PW . Reovirus oncolysis: the Ras/RalGEF/p38 pathway dictates host cell permissiveness to reovirus infection. Proc Natl Acad Sci USA 2004; 101: 11099–11104.
Kim M, Chung YH, Johnston RN . Reovirus and tumor oncolysis. J Microbiol 2007; 45: 187–192.
Rosen L, Evans HE, Spickard A . Reovirus infections in human volunteers. Am J Hyg 1963; 77: 29–37.
Nibert ML, Schiff LA, Fields BN . Mammalian reoviruses contain a myristoylated structural protein. J Virol 1991; 65: 1960–1967.
Duncan MR, Stanish SM, Cox DC . Differential sensitivity of normal and transformed human cells to reovirus infection. J Virol 1978; 28: 444–449.
Hashiro G, Loh PC, Yau JT . The preferential cytotoxicity of reovirus for certain transformed cell lines. Arch Virol 1977; 54: 307–315.
Strong JE, Coffey MC, Tang D, Sabinin P, Lee PW . The molecular basis of viral oncolysis: usurpation of the Ras signaling pathway by reovirus. EMBO J 1998; 17: 3351–3362.
Hirasawa K, Nishikawa SG, Norman KL, Coffey MC, Thompson BG, Yoon CS et al. Systemic reovirus therapy of metastatic cancer in immune-competent mice. Cancer Res 2003; 63: 348–353.
Coffey MC, Strong JE, Forsyth PA, Lee PW . Reovirus therapy of tumors with activated Ras pathway. Science 1998; 282: 1332–1334.
Norman KL, Lee PW . Not all viruses are bad guys: the case for reovirus in cancer therapy. Drug Discov Today 2005; 10: 847–855.
Yang WQ, Lun X, Palmer CA, Wilcox ME, Muzik H, Shi ZQ et al. Efficacy and safety evaluation of human reovirus type 3 in immunocompetent animals: racine and nonhuman primates. Clin Cancer Res 2004; 10: 8561–8576.
Ring CJ . Cytolytic viruses as potential anti-cancer agents. J Gen Virol 2002; 83 (Part 3): 491–502.
Russel FG, Masereeuw R, van Aubel RA . Molecular aspects of renal anionic drug transport. Annu Rev Physiol 2002; 64: 563–594.
Ikeda Y, Nishimura G, Yanoma S, Kubota A, Furukawa M, Tsukuda M . Reovirus oncolysis in human head and neck squamous carcinoma cells. Auris Nasus Larynx 2004; 31: 407–412.
Janssen HL, Higuchi H, Abdulkarim A, Gores GJ . Hepatitis B virus enhances tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) cytotoxicity by increasing TRAIL-R1/death receptor 4 expression. J Hepatol 2003; 39: 414–420.
Clarke P, Meintzer SM, Spalding AC, Johnson GL, Tyler KL . Caspase 8-dependent sensitization of cancer cells to TRAIL-induced apoptosis following reovirus-infection. Oncogene 2001; 20: 6910–6919.
Tiollais P, Charnay P, Vyas GN . Biology of hepatitis B virus. Science 1981; 213: 406–411.
Maguire HF, Hoeffler JP, Siddiqui A . HBV X protein alters the DNA binding specificity of CREB and ATF-2 by protein–protein interactions. Science 1991; 252: 842–844.
Benn J, Schneider RJ . Hepatitis B virus HBx protein activates Ras-GTP complex formation and establishes a Ras, Raf, MAP kinase signaling cascade. Proc Natl Acad Sci USA 1994; 91: 10350–10354.
Becker SA, Lee TH, Butel JS, Slagle BL . Hepatitis B virus X protein interferes with cellular DNA repair. J Virol 1998; 72: 266–272.
Chung TW, Lee YC, Ko JH, Kim CH . Hepatitis B virus X protein modulates the expression of PTEN by inhibiting the function of p53, a transcriptional activator in liver cells. Cancer Res 2003; 63: 3453–3458.
Waris G, Huh KW, Siddiqui A . Mitochondrially associated hepatitis B virus X protein constitutively activates transcription factors STAT-3 and NF-kappa B via oxidative stress. Mol Cell Biol 2001; 21: 7721–7730.
Kekule AS, Lauer U, Weiss L, Luber B, Hofschneider PH . Hepatitis B virus transactivator HBx uses a tumour promoter signalling pathway. Nature 1993; 361: 742–745.
Benn J, Su F, Doria M, Schneider RJ . Hepatitis B virus HBx protein induces transcription factor AP-1 by activation of extracellular signal-regulated and c-Jun N-terminal mitogen-activated protein kinases. J Virol 1996; 70: 4978–4985.
Chirillo P, Falco M, Puri PL, Artini M, Balsano C, Levrero M et al. Hepatitis B virus pX activates NF-kappa B-dependent transcription through a Raf-independent pathway. J Virol 1996; 70: 641–646.
Bouchard MJ, Schneider RJ . The enigmatic X gene of hepatitis B virus. J Virol 2004; 78: 12725–12734.
Um HR, Lim WC, Chae SY, Park S, Park JH, Cho H . Raf-1 and protein kinase B regulate cell survival through the activation of NF-kappaB in hepatitis B virus X-expressing cells. Virus Res 2007; 125: 1–8.
Kim M, Egan C, Alain T, Urbanski SJ, Lee PW, Forsyth PA et al. Acquired resistance to reoviral oncolysis in Ras-transformed fibrosarcoma cells. Oncogene 2007; 26: 4124–4134.
McDermott U, Longley DB, Galligan L, Allen W, Wilson T, Johnston PG . Effect of p53 status and STAT1 on chemotherapy-induced, Fas-mediated apoptosis in colorectal cancer. Cancer Res 2005; 65: 8951–8960.
Cho IR, Jeong S, Jhun BH, An WG, Lee B, Kwak YT et al. Activation of non-canonical NF-kappaB pathway mediated by STP-A11, an oncoprotein of Herpesvirus saimiri. Virology 2007; 359: 37–45.
Ayroldi E, Zollo O, Macchiarulo A, Di Marco B, Marchetti C, Riccardi C . Glucocorticoid-induced leucine zipper inhibits the Raf-extracellular signal-regulated kinase pathway by binding to Raf-1. Mol Cell Biol 2002; 22: 7929–7941.
Strong JE, Lee PW . The v-erbB oncogene confers enhanced cellular susceptibility to reovirus infection. J Virol 1996; 70: 612–616.
Klein NP, Schneider RJ . Activation of Src family kinases by hepatitis B virus HBx protein and coupled signaling to Ras. Mol Cell Biol 1997; 17: 6427–6436.
Tarn C, Lee S, Hu Y, Ashendel C, Andrisani OM . Hepatitis B virus X protein differentially activates RAS-RAF-MAPK and JNK pathways in X-transforming versus non-transforming AML12 hepatocytes. J Biol Chem 2001; 276: 34671–34680.
Clarke P, Meintzer SM, Gibson S, Widmann C, Garrington TP, Johnson GL et al. Reovirus-induced apoptosis is mediated by TRAIL. J Virol 2000; 74: 8135–8139.
Lee YH, Yun Y . HBx protein of hepatitis B virus activates Jak1-STAT signaling. J Biol Chem 1998; 273: 25510–25515.
Lee YI, Kang-Park S, Do SI, Lee YI . The hepatitis B virus-X protein activates a phosphatidylinositol 3-kinase-dependent survival signaling cascade. J Biol Chem 2001; 276: 16969–16977.
Uddin S, Yenush L, Sun XJ, Sweet ME, White MF, Platanias LC . Interferon-alpha engages the insulin receptor substrate-1 to associate with the phosphatidylinositol 3′-kinase. J Biol Chem 1995; 270: 15938–15941.
Uddin S, Fish EN, Sher D, Gardziola C, Colamonici OR, Kellum M et al. The IRS-pathway operates distinctively from the Stat-pathway in hematopoietic cells and transduces common and distinct signals during engagement of the insulin or interferon-alpha receptors. Blood 1997; 90: 2574–2582.
McKay DM, Botelho F, Ceponis PJ, Richards CD . Superantigen immune stimulation activates epithelial STAT-1 and PI 3-K: PI 3-K regulation of permeability. Am J Physiol Gastrointest Liver Physiol 2000; 279: G1094–G1103.
Navarro A, Anand-Apte B, Tanabe Y, Feldman G, Larner AC . A PI-3 kinase-dependent, Stat1-independent signaling pathway regulates interferon-stimulated monocyte adhesion. J Leukoc Biol 2003; 73: 540–545.
Mulrooney-Cousins PM, Michalak TI . Persistent occult hepatitis B virus infection: experimental findings and clinical implications. World J Gastroenterol 2007; 13: 5682–5686.
Lupberger J, Hildt E . Hepatitis B virus-induced oncogenesis. World J Gastroenterol 2007; 13: 74–81.
Su F, Schneider RJ . Hepatitis B virus HBx protein sensitizes cells to apoptotic killing by tumor necrosis factor alpha. Proc Natl Acad Sci USA 1997; 94: 8744–8749.
Diao J, Khine AA, Sarangi F, Hsu E, Iorio C, Tibbles LA et al. X protein of hepatitis B virus inhibits Fas-mediated apoptosis and is associated with up-regulation of the SAPK/JNK pathway. J Biol Chem 2001; 276: 8328–8340.
Barton ES, Forrest JC, Connolly JL, Chappell JD, Liu Y, Schnell FJ et al. Junction adhesion molecule is a receptor for reovirus. Cell 2001; 104: 441–451.
Guglielmi KM, Kirchner E, Holm GH, Stehle T, Dermody TS . Reovirus binding determinants in junctional adhesion molecule-A. J Biol Chem 2007; 282: 17930–17940.
Garcia R, Bowman TL, Niu G, Yu H, Minton S, Muro-Cacho CA et al. Constitutive activation of Stat3 by the Src and JAK tyrosine kinases participates in growth regulation of human breast carcinoma cells. Oncogene 2001; 20: 2499–2513.
Bromberg JF, Wrzeszczynska MH, Devgan G, Zhao Y, Pestell RG, Albanese C et al. Stat3 as an oncogene. Cell 1999; 98: 295–303.
Catlett-Falcone R, Dalton WS, Jove R . STAT proteins as novel targets for cancer therapy. Signal transducer an activator of transcription. Curr Opin Oncol 1999; 11: 490–496.
Srivastava SP, Kumar KU, Kaufman RJ . Phosphorylation of eukaryotic translation initiation factor 2 mediates apoptosis in response to activation of the double-stranded RNA-dependent protein kinase. J Biol Chem 1998; 273: 2416–2423.
Wek RC . eIF-2 kinases: regulators of general and gene-specific translation initiation. Trends Biochem Sci 1994; 19: 491–496.
Stewart MJ, Blum MA, Sherry B . PKR's protective role in viral myocarditis. Virology 2003; 314: 92–100.
Zhang P, Samuel CE . Protein kinase PKR plays a stimulus- and virus-dependent role in apoptotic death and virus multiplication in human cells. J Virol 2007; 81: 8192–8200.
Chang KS, Cai Z, Zhang C, Sen GC, Williams BR, Luo G . Replication of hepatitis C virus (HCV) RNA in mouse embryonic fibroblasts: protein kinase R (PKR)-dependent and PKR-independent mechanisms for controlling HCV RNA replication and mediating interferon activities. J Virol 2006; 80: 7364–7374.
Acknowledgements
This study was supported for two years by Pusan National University Research Grants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, EH., Koh, S., Srisuttee, R. et al. Expression of HBX, an oncoprotein of hepatitis B virus, blocks reoviral oncolysis of hepatocellular carcinoma cells. Cancer Gene Ther 16, 453–461 (2009). https://doi.org/10.1038/cgt.2008.95
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2008.95
Keywords
This article is cited by
-
Hepatitis B virus X protein impedes the DNA repair via its association with transcription factor, TFIIH
BMC Microbiology (2011)
-
CUG2, a novel oncogene confers reoviral replication through Ras and p38 signaling pathway
Cancer Gene Therapy (2010)
-
Down-regulation of HIF-1α by oncolytic reovirus infection independently of VHL and p53
Cancer Gene Therapy (2010)