Abstract
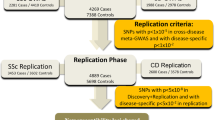
Multiple sclerosis (MS) is a T-cell-mediated disease of the central nervous system, characterized by damage to myelin and axons, resulting in progressive neurological disability. Genes may influence susceptibility to MS, but results of association studies are inconsistent, aside from the identification of HLA class II haplotypes. Whole-genome linkage screens in MS have both confirmed the importance of the HLA region and uncovered non-HLA loci that may harbor susceptibility genes. In this two-stage analysis, we determined genotypes, in up to 672 MS patients and 672 controls, for 123 single-nucleotide polymorphisms (SNPs) in 66 genes. Genes were chosen based on their chromosomal positions or biological functions. In stage one, 22 genes contained at least one SNP for which the carriage rate for one allele differed significantly (P<0.08) between patients and controls. After additional genotyping in stage two, two genes—each containing at least three significantly (P<0.05) associated SNPs—conferred susceptibility to MS: LAG3 on chromosome 12p13, and IL7R on 5p13. LAG3 inhibits activated T cells, while IL7R is necessary for the maturation of T and B cells. These results imply that germline allelic variation in genes involved in immune homeostasis—and, by extension, derangement of immune homeostasis—influence the risk of MS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kurtzke JF . Epidemiologic evidence for multiple sclerosis as an infection. Clin Microbiol Rev 1993; 6: 382–427.
Lassmann H . Neuropathology in multiple sclerosis: new concepts. Multiple Sclerosis 1998; 4: 93–98.
Ebers GC, Sadovnick AD . The role of genetic factors in multiple sclerosis susceptibility. J Neuroimmunol 1994; 54: 1–17.
Oksenberg JR, Baranzini SE, Barcellos LF, Hauser SL . Multiple sclerosis: genomic rewards. J Neuroimmunol 2001; 113: 171–184.
GAMES and Transatlantic Multiple Sclerosis Genetics Cooperative. A meta-analysis of whole genome linkage screens in multiple sclerosis. J Neuroimmunol 2003; 143: 39–46.
Becker KG, Simon RM, Bailey-Wilson JE et al. Clustering of non-major histocompatibility complex susceptibility candidate loci in human autoimmune diseases. Proc Natl Acad Sci USA 1998; 95: 9979–9984.
Ogura Y, Bonen DK, Inohara N et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature 2001; 411: 603–606.
Prokunina L, Castillejo-Lopez C, Oberg F et al. A regulatory polymorphism in PDCD1 is associated with susceptibility to systemic lupus erythematosus in humans. Nat Genet 2002; 32: 666–669.
Florez JC, Hirschhorn J, Altshuler D . The inherited basis of diabetes mellitus: implications for the genetic analysis of complex traits. Annu Rev Genomics Hum Genet 2003; 4: 257–291.
Kenealy SJ, Pericak-Vance MA, Haines JL . The genetic epidemiology of multiple sclerosis. J Neuroimmunol 2003; 143: 7–12.
Ronaghi M, Uhlen M, Nyren P . A sequencing method based on real-time pyrophosphate. Science 1998; 281: 363–365.
Schrijver HM, Crusius JB, Uitdehaag BM et al. Association of interleukin-1beta and interleukin-1 receptor antagonist genes with disease severity in MS. Neurology 1999; 52: 595–599.
Feakes R, Sawcer S, Broadley S et al. Interleukin 1 receptor antagonist (IL-1ra) in multiple sclerosis. J Neuroimmunol 2000; 105: 96–101.
Giedraitis V, He B, Huang WX, Hillert J . Cloning and mutation analysis of the human IL-18 promoter: a possible role of polymorphisms in expression regulation. J Neuroimmunol 2001; 112: 146–152.
Kotze MJ, de Villiers JN, Rooney RN et al. Analysis of the NRAMP1 gene implicated in iron transport: association with multiple sclerosis and age effects. Blood Cells Mol Dis 2001; 27: 44–53.
Hensiek AE, Roxburgh R, Meranian M et al. Osteopontin gene and clinical severity of multiple sclerosis. J Neurol 2003; 250: 943–947.
Teutsch SM, Booth DR, Bennetts BH et al. Identification of 11 novel and common single nucleotide polymorphisms in the interleukin-7 receptor-alpha gene and their associations with multiple sclerosis. Eur J Hum Genet 2003; 11: 509–515.
Jacobsen M, Schweer D, Ziegler A et al. A point mutation in PTPRC is associated with the development of multiple sclerosis. Nat Genet 2000; 26: 495–499.
Kantarci OH, Atkinson EJ, Hebrink DD et al. Association of two variants in IL-1beta and IL-1 receptor antagonist genes with multiple sclerosis. J Neuroimmunol 2000; 106: 220–227.
Vorechovsky I, Kralovicova J, Tchilian E et al. Does 77C → G in PTPRC modify autoimmune disorders linked to the major histocompatibility locus? Nat Genet 2001; 29: 22–23.
Giess R, Maurer M, Linker R et al. Association of a null mutation in the CNTF gene with early onset of multiple sclerosis. Arch Neurol 2002; 59: 407–409.
Martin ER, Lai EH, Gilbert JR et al. SNPing away at complex diseases: analysis of single-nucleotide polymorphisms around APOE in Alzheimer disease. Am J Hum Genet 2000; 67: 383–394.
Shifman S, Bronstein M, Sternfeld M et al. A highly significant association between a COMT haplotype and schizophrenia. Am J Hum Genet 2002; 71: 1296–1302.
Bruniquel D, Borie N, Hannier S, Triebel F . Regulation of expression of the human lymphocyte activation gene-3 (LAG-3) molecule, a ligand for MHC class II. Immunogenetics 1998; 48: 116–124.
Workman CJ, Cauley LS, Kim IJ et al. Lymphocyte activation gene-3 (CD223) regulates the size of the expanding T cell population following antigen activation in vivo. J Immunol 2004; 172: 5450–5455.
Andreae S, Piras F, Burdin N, Triebel F . Maturation and activation of dendritic cells induced by lymphocyte activation gene-3 (CD223). J Immunol 2002; 168: 3874–3880.
Fry TJ, Mackall CL . Interleukin-7: from bench to clinic. Blood 2002; 99: 3892–3904.
Ye SK, Maki K, Kitamura T et al. Induction of germline transcription in the TCRgamma locus by Stat5: implications for accessibility control by the IL-7 receptor. Immunity 1999; 11: 213–223.
Lai SY, Molden J, Goldsmith MA . Shared gamma(c) subunit within the human interleukin-7 receptor complex. A molecular basis for the pathogenesis of X-linked severe combined immunodeficiency. J Clin Invest 1997; 99: 169–177.
Ramanathan M, Weinstock-Guttman B, Nguyen LT et al. In vivo gene expression revealed by cDNA arrays: the pattern in relapsing-remitting multiple sclerosis patients compared with normal subjects. J Neuroimmunol 2001; 116: 213–219.
Poser CM, Paty DW, Scheinberg L et al. New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol 1983; 13: 227–231.
National Biosciences Inc. Oligo 5.0. Plymouth: Maine, USA, 1994.
Nordfors L, Jansson M, Sandberg G et al. Large-scale genotyping of single nucleotide polymorphisms by Pyrosequencing trade mark and validation against the 5′ nuclease (Taqman) assay. Hum Mutat 2002; 19: 395–401.
Zhao JH, Curtis D, Sham PC . Model-free analysis and permutation tests for allelic associations. Hum Hered 2000; 50: 133–139.
Sham PC, Curtis D . Monte Carlo tests for associations between disease and alleles at highly polymorphic loci. Ann Hum Genet 1995; 59: 97–105.
Slatkin M, Excoffier L . Testing for linkage disequilibrium in genotypic data using the Expectation-Maximization algorithm. Heredity 1996; 76: 377–383.
Abecasis GR, Cardon LR, Cookson WO . A general test of association for quantitative traits in nuclear families. Am J Hum Genet 2000; 66: 279–292.
Epstein MP, Satten GA . Inference on haplotype effects in case–control studies using unphased genotype data. Am J Hum Genet 2003; 73: 1316–1329.
Acknowledgements
This study was supported by grants from the European Commission, the Swedish Medical Research Council, the Swedish Association of Neurologically Disabled, the Swedish Society of Medicine, the Swedish Brain Foundation, the Lars Hierta Foundation, the Swedish Foundation for Strategic Research, and Karolinska Institutet. We thank Nancy Pedersen of the Swedish Twin Registry and all the patients and volunteers who participated in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Genes and Immunity website (http://www.nature.com/gene).
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, Z., Duvefelt, K., Svensson, F. et al. Two genes encoding immune-regulatory molecules (LAG3 and IL7R) confer susceptibility to multiple sclerosis. Genes Immun 6, 145–152 (2005). https://doi.org/10.1038/sj.gene.6364171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gene.6364171
Keywords
This article is cited by
-
Evaluation of the possible association of PDCD-1 and LAG3 gene polymorphisms with hepatocellular carcinoma risk
BMC Medical Genomics (2023)
-
Advances in immune checkpoint-based immunotherapies for multiple sclerosis: rationale and practice
Cell Communication and Signaling (2023)
-
Novel human immunomodulatory T cell receptors and their double-edged potential in autoimmunity, cardiovascular disease and cancer
Cellular & Molecular Immunology (2021)
-
Association of LAG3 genetic variation with an increased risk of PD in Chinese female population
Journal of Neuroinflammation (2019)
-
IL-7/IL-7R gene variants impact circulating IL-7/IL-7R homeostasis and ART-associated immune recovery status
Scientific Reports (2019)