Abstract
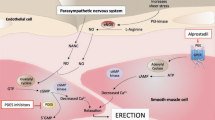
In two multicenter, placebo controlled, phase 2 studies, patients with mild-to-moderate (n=161, Study 1) or severe (n=142, Study 2) erectile dysfunction (ED) were randomized to receive placebo, 0.05, 0.1, or 0.2 mg (Study 1) or placebo, 0.1, 0.2, or 0.3 mg (Study 2) of topically applied alprostadil (containing a proprietary skin permeation enhancer). The primary efficacy end point in both studies was the change in erectile function (EF) score from baseline to final visit. The changes from baseline for EF scores were −0.8±1.1, 1.8±1.1, 0.7±1.2, and 3.7±1.2 (P<0.01; Study 1) and 2.7±1.3, 6.29±1.4, 6.49±1.5, and 9.44±1.5 (P<0.001; Study 2) for ascending dose groups in each study. Topical alprostadil was well tolerated with the most common adverse event being urogenital pain. These results suggest this topical alprostadil formulation may be a potentially useful agent for the treatment of ED.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
McKinlay JB . The worldwide prevalence and epidemiology of erectile dysfunction. Int J Impot Res 2000; 12(Suppl 4): S6–S11.
Feldman HA et al. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol 1994; 151: 54–61.
Goldstein I, Lue TF, Padma-Nathan H et al. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N Engl J Med 1988; 338: 1397–1404.
Linet O, Ogrinc FG, Alprostadil Study Group. Efficacy and safety of intracavernosal Alprostadil in men with erectile dysfunction. N Engl J Med 1996; 334: 873–877.
Porst H, Buvat J, Meuleman E, Michal V, Wagner G . Intracavernous alprostadil alfadex—an effective and well tolerated treatment for erectile dysfunction. Results of a long-term European study. Int J Impot Res 1998; 10: 225–231.
Padma-Nathan H, Hellstrom WJG, Kaiser FE et al. Treatment of men with erectile dysfunction with transurethral alprostadil. N Engl J Med 1997; 336: 1–7.
Porst H . Transurethral Alprostadil with MUSETM (medicated urethral system for erection) vs intracavernosal Alprostadil—a comparative study of 103 patients with erectile dysfunction. Int J Impot Res 1997; 9: 187–192.
Porst H . Review Article. The rationale for prostaglandin E1 in erectile failure: a survey of worldwide experience. J Urol 1996; 155: 802–815.
Adaikin PG, Kottegoda SR, Ratnam SS . A possible role for prostaglandin E1 in human penile erection. In: Abstract Book Second World Meeting on Impotence. Prague, 1986; Abstract 2.6.
Shabsigh R, Padma-Nathan H, Gittleman M et al. Intracavernous alprostadil alfadex (edex/viridal) is effective and safe in patients with erectile dysfunction after failing sildenafil (Viagra). Urology 2000; 55: 477.
Kim ED, McVary KT . Topical prostaglandin-E1 for the treatment of erectile dysfunction. J Urol 1995; 153: 1828.
Montorsi F, Guazzoni C, Barbieri L et al. Clinical and hemodynamic effects of transdermal alprostadil for mild arteriogenic impotence: a double-blind placebo controlled study. Int J Impot Res 1995; 7: 10.
Goldstein I, Payton TR, Schechter PJ . A double-blind, placebo-controlled, efficacy and safety study of topical gel formulation of 1% alprostadil (topiglan) for the in-office treatment of erectile dysfunction. Urology 2001; 57: 301.
Rosen RC, Riley A, Wagner G et al. The International Index of Erectile Function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997; 49: 822.
Padma-Nathan H, McMurray JG, Pullman WE et al. On-demand IC351 (Cialis™) enhances erectile function in patients with erectile dysfunction. Int J Imp Res 2001; 13: 2.
Padma-Nathan H et al. Hemodynamic effects of intraurethral Alprostadil: The Medicated Urethral System for Erection (MUSE). J Urol 1994; 151: 345A (469).
Porst HRR, Padma-Nathan H et al. The efficacy and tolerability of vardenafil, a new, oral, selective phosphodiesterase type 5 inhibitor, in patients with erectile dysfunction: the first at-home clinical trial. Int J Impot Res 2001; 13: 192–199.
Acknowledgements
These studies were supported by a grant from NexMed Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Padma-Nathan, H., Steidle, C., Salem, S. et al. The efficacy and safety of a topical alprostadil cream, Alprox-TD®, for the treatment of erectile dysfunction: two phase 2 studies in mild-to-moderate and severe ED. Int J Impot Res 15, 10–17 (2003). https://doi.org/10.1038/sj.ijir.3900940
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3900940
Keywords
This article is cited by
-
Combination therapy with topical alprostadil and phosphodiesterase-5 inhibitors after failure of oral therapy in patients with erectile dysfunction: a prospective, two-arm, open-label, non-randomized study
International Journal of Impotence Research (2022)
-
The intra-meatal application of alprostadil cream (Vitaros®) improves drug efficacy and patient’s satisfaction: results from a randomized, two-administration route, cross-over clinical trial
International Journal of Impotence Research (2019)
-
Combination therapy for erectile dysfunction involving a PDE5 inhibitor and alprostadil
International Journal of Impotence Research (2018)
-
Efficacy of PDE-5-inhibitors for erectile dysfunction. A comparative meta-analysis of fixed-dose regimen randomized controlled trials administering the International Index of Erectile Function in broad-spectrum populations
International Journal of Impotence Research (2006)
-
Retention and migration of alprostadil cream applied topically to the glans meatus for erectile dysfunction
International Journal of Impotence Research (2005)