Abstract
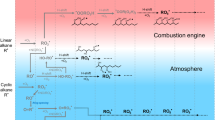
Atmospheric hydrocarbons have an important influence on the chemistry of the polluted lower atmosphere. Aromatic hydrocarbons or benzene derivatives comprise about 25–40% of gasoline in the US1 and they are widely employed as solvents. Toluene is the most commonly used aromatic hydrocarbon and is often the most abundant of all non-methane hydrocarbons in urban atmospheres. Typical urban toluene concentrations range from 1 to 50 p.p.b. (parts per 109)2,3 and clean-air concentrations up to 0.4 p.p.b. have been reported4. The aromatic hydrocarbons are destroyed by reaction with atmospheric hydroxyl radical (HO) and toluene remains in the atmosphere for about 50 daylight hours before reacting. Despite extensive study, toluene's reaction products are poorly understood5. Many reaction products have been identified, but most of these are ring-addition or side-chain oxidation products which retain the aromatic character of toluene. We have conducted a thorough study of toluene's oxidation products in simulated atmospheric conditions and present here an account of the products found and outline their probable formation and destruction mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
O'Brien, R. J., Hightower, J. & Crocker, T. D. in The Alkylbenzenes Ch. 1 (ed. Peter, F. M.) (National Academy of Sciences, Washington DC, 1981).
Pellizzari, E. D. Information on the Characterization of Ambient Organic Vapors in Areas of High Chemical Pollution Control No. 68-02-2721 (US Environmental Protection Agency, North Carolina, 1979).
Sexton, K. & Westberg, H. Envir. Sci. Technol. 14, 329–332 (1980).
Rasmussen, R. A. & Khalil, M. A. K. Geophys. Res. Lett. 10, 1096–1099 (1983).
O'Brien, R. J., Johnson, J. E. & Wilkerson, C. in The Alkylbenzenes Ch. 4 (ed. Peter, F. M.) (National Academy of Sciences, Washington DC, 1981).
O'Brien, R. J., Green, P. J., Nguyen, N-L., Doty, R. A. & Dumdei, B. E. Envir. Sci. Technol. 17, 183–186 (1983).
Slayback, J. R. B., Story, M. S. Ind. Res. Devl February 129–139 (1981).
O'Brien, R. J., Dumdei, B. E., Hummel, S. V. & Yost, R. A. Analyt. Chem. (in the press).
Nojima, K., Fukaya, K., Fukui, S., Kanno, S. Chemosphere 5, 247–252 (1974).
Schwartz, W. Chemical Characterization of Model Aerosols (Rep. to U.S. Environmental Protection Agency, NTIS PB-238 557, 1974).
Akimoto, H., Hosbino, J., Inone, G., Okuda, M. & Washida, N. Bull. chem. Soc. Jap. 51, 2496–2502 (1978).
Hoshino, J., Akimoto, J. & Okuda, M. Bull chem. Soc. Jap. 51, 718–724 (1978).
Ishikawa, J., Watanabe, K. & Ando, W. Bull. chem. Soc. Jap. 51, 2174–2178 (1978).
O'Brien, R. J. et al. in Nitrogenous Air Pollutants Ch. 11 (ed. Grosjean, D.) (Ann Arbor Press, 1979).
Besemer, A. C. Atmos. Envir. 16, 1599–1602 (1982).
Leone, J. A. & Seinfeld, J. H. Int. J. Chem. Kinet. 16, 159–174 (1984).
Killus, J. P. & Whitten, G. Z. Atmos. Envir. 16, 1973–1988 (1982).
Atkinson, R., Carter, W. P. L., Darnall, K. R., Winer, A. M. & Pitts, J. N. Jr Int. J. chem. Kinet. 12, 799–836 (1980).
O'Brien, R. J., Green, P. J. & Doty, R. A. J. phys. Chem. 83, 3302 (1979).
O'Brien, R. J., Dumdei, B. E., Hummel, S. V. & Yost, R. A. 38th Northwest Regional Meet. Am. chem. Soc. Honolulu (submitted for publication).
Lee, E. K. C. & Lewis, R. S. Adv. Photochem. 12, 53–55 (1980).
Takamuku, S., Matsimoto, H., Hori, A. & Sakurai, H. J. Am. chem. Soc. 102, 1441–1443 (1980).
Calvert, J. G. & Stockwell, W. R. Envir. Sci. Technol. 17, 428A–439 (1983).
Atkinson, R., Aschmann, S. M., Winer, A. M. & Pitts, J. N. Jr Envir. Sci. Technol. 18, 370–374 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dumdei, B., O'Brien, R. Toluene degradation products in simulated atmospheric conditions. Nature 311, 248–250 (1984). https://doi.org/10.1038/311248a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/311248a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.