Abstract
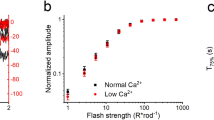
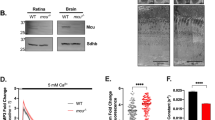
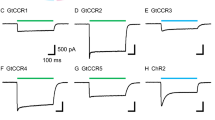
Photons absorbed in vertebrate rods and cones probably cause electrochemical changes at the photoreceptor plasma membrane by changing the cytoplasmic concentration of a diffusible transmitter substance, reducing the Na+ current flowing into the outer segment of the cell in the dark, to produce the observed membrane hyperpolarization that is the initial excitatory response1–4. Cyclic GMP has been proposed as the transmitter because a light-activated cyclic GMP phosphodiesterase (PDE) has been found in rod disk membranes5,6 and because intracellularly injected cyclic GMP7 reduces rod membrane potentials. Free Ca2+ has also been proposed because increasing external [Ca2+] quickly and reversibly reduces the dark current and divalent cationophores increase the Ca2+ sensitivity8. Ca2+ efflux from rod outer segments (ROS) of intact retinas occurs simultaneously with light responses9,10. Vesicles prepared from ROS disk membranes become more permeable on illumination, releasing trapped ions or molecules11–13, but intact outer segment disks have not previously been found to store sufficient Ca2+ in darkness and to release enough in light to meet the theoretical requirements for control of the dark current by varying cytoplasmic Ca2+ (refs 14–18). We now report experiments that show the required Ca2+ storage and release from rod disk membranes suspended in media containing high-energy phosphate esters and electrolytes approximating the cytoplasmic composition of live rod cells. Cyclic GMP stimulates Ca2+ uptake by ROS disks in such media.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bortoff, A. & Norton, A. L. Vision Res. 7, 253 (1967).
Baylor, D. A. & Fuortes, M. G. F. J. Physiol., Lond. 207, 77–92 (1970).
Penn, R. D. & Hagins, W. A. Biophys. J. 12, 1073–1094 (1972).
Yau, K.-W., McNaughton, P. A. & Hodgkin, A. L. Nature 292, 502–505 (1981).
Miki, N., Baraban, J. M., Keirns, J. J., Boyce, J. & Bitensky, M. W. J. biol. Chem. 250, 6320–6327 (1975).
Liebman, P. A. & Pugh, E. N. Jr Vision Res. 19, 375–380 (1979).
Miller, W. H. & Nicol, G. D. Nature 280, 64–66 (1979).
Hagins, W. A. A. Rev. Biophys. Bioengng 1, 131–158 (1972).
Yoshikami, S., George, J. S. & Hagins, W. A. Nature 286, 395–598 (1980).
Gold, G. H. & Korenbrot, J. I. Proc. natn. Acad. Sci. U.S.A. 77, 5557–5561 (1980).
Montal, M., Darzon, A. & Trissl, H. W. Nature 267, 221–225 (1977).
Smith, H. G. & Bauer, P. J. Biochemistry 18, 5067–5073 (1979).
O'Brien, D. F., Zumbulyadis, N., Michaels, P. M. & Ott, R. A. Proc. natn. Acad. Sci. U.S.A. 74, 5222–5226 (1977).
Szuts, E. Z. & Cone, R. A. Biochim. biophys. Acta 468, 194–208 (1977).
Kaupp, U. B., Schnetkamp, P. P. M. & Junge, W. Nature 286, 638–640 (1980).
Szuts, E. J. gen. Physiol. 76, 253–286 (1980).
Schnetkamp, P. P. M., Daemen, F. J. M. & Bonting, S. L. Biochim. biophys. Acta 468, 259–270 (1977).
Schnetkamp, P. P. M. Biochim. biophys. Acta 554, 441–459 (1979).
Robinson, W. E. & Hagins, W. A. Nature 28, 398–400 (1979).
Hagins, W. A., Foster, M. C., George, J. S. & Trus, B. Microbeam Analysis, 139–142 (San Francisco Press, 1982).
Cavaggioni, A. & Sorbi, R. T. Proc. natn. Acad. Sci. U.S.A. 78, 3964–3968 (1981).
Vasington, R., Gazetti, P., Tiozzo, R. & Carofoli, E. Biochim. biophys. Acta 256, 43–54 (1972).
Yee, R. & Liebman, P. A. J. biol. Chem. 253, 8902–8909 (1979).
Cohen, A. I., Hall, I. A. & Ferrendelli, J. A. J. gen. Physiol. 71, 595–612 (1978).
Kilbride, K. P. & Ebrey, T. G. J. gen. Physiol. 74, 415–426 (1979).
Robinson, W. E. & Hagins, W. A. Fedn Proc. 39, (1980).
George, J. S. & Hagins, W. A. Biophys. J. 33, 288a (1981).
Baylor, D. A., Lamb, T. D. & Yau, K.-W. J. Physiol., Lond. 288, 589–611 (1979).
Hagins, W. A. & Yoshikami, S. in Vertebrate Photoreception (eds Fatt, P. & Barlow, H.) 97–137 (Academic, New York, 1977).
Yoshikami, S. & Hagins, W. A. Fedn Proc. 39, 1092 (1980).
Caretta, A., Cavaggioni, A. & Sorbi, R. T. J. Physiol., Lond. 295, 171–178 (1979).
Lipton, S. A., Rasmussen, H. & Dowling, J. E. J. gen. Physiol. 70, 771–791 (1977).
Polans, A. S., Kawamura, S. & Bownds, M. D. J. gen. Physiol. 77, 41–48 (1981).
Kilbride, P. J. gen. Physiol. 75, 457–465 (1980).
Yoshikami, S., Robinson, W. E. & Hagins, W. A. Science 185, 1176–1179 (1977).
Amman, D., Guggi, M., Pretsch, E. & Simon, W. Analyt. Lett. 8, 709–720 (1975).
Marban, E., Rink, T. J., Tsien, R. W. & Tsien, R. Y. Nature 286, 845–850 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
George, J., Hagins, W. Control of Ca2+ in rod outer segment disks by light and cyclic GMP. Nature 303, 344–348 (1983). https://doi.org/10.1038/303344a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/303344a0
This article is cited by
-
Size and chirality of intracellularly applied anions affect the function of isolated photoreceptors
Pfl�gers Archiv European Journal of Physiology (1992)
-
Ultrastructural localization of endogenous calcium in the teleost retina
The Histochemical Journal (1990)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.