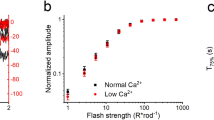
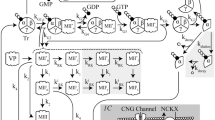
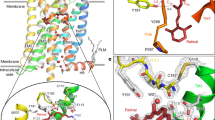
Abstract
IT has recently been shown in several laboratories that if isolated rod outer segments (ROS) are incubated in the presence of MgATP, rhodopsin is phosphorylated (see ref. 1). The reaction is much faster after the ROS have been exposed to light as the enzyme, ‘opsin kinase’, is specific to bleached rather than unbleached rhodopsin1. It has been suggested that the function of rhodopsin phosphorylation may be to regulate the responsiveness of ROS to light and in this paper we describe a mechanism whereby such a regulation could occur.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Weller, M., Virmaux, N., and Mandel, P., Proc. natn. Acad. Sci. U.S.A., 72, 381–385 (1974).
Tomita, T., Miller, W. H., Hashimoto, Y., and Saito, T., Expl Eye Res., 16, 327–341 (1973).
Hagins, W. A., A. Rev. Biophys. Bioeng, 1, 131–158 (1972).
Korenbrot, J. I., Expl Eye Res., 16, 343–355 (1973).
Penn, R. D., and Hagins, W. A., Biophys. J., 12, 1073–1094 (1972).
Cohen, A. I., Vision Res., 10, 445–453 (1970).
Yoshikami, S., and Hagins, W. A., in Biochemistry and Physiology of the Visual Pigments (edit. by Langer, H.), 246–255 (Springer, Berlin, 1973).
Hagins, W. A., and Yoshikami, S., Expl Eye Res., 18, 299–305 (1974).
Bownds, D., Gordon-Walker, A., Gaide-Huguenin, A. D., and Robinson, W. E., J. gen. Physiol., 58, 225–237 (1971).
Hendriks, Th., Daemen, F. J. M., and Bonting, S. L., Biochim. biophys. Acta, 345, 468–473 (1974).
Poo, M. M., and Cone, R. A., Expl Eye Res., 17, 503–510 (1973).
Mason, W. T., Fager, R. S., and Abrahamson, E. W., Nature, 247, 562–563 (1974).
Szuts, E. Z., and Cone, R. A., Fedn Proc., 33, 1471 (1974).
Shlatz, L., and Marinetti, G. V., Biochem. biophys. Res. Commun., 45, 41–50 (1971).
Will, H., Schirpke, B., and Wollenberger, A., Acta Biol. med. Germ., 31 K, 45–52 (1973).
Kirchberger, M. A., Tada, M., Repke, D. I., and Latz, A. M., J. molec. Cell. Cardiol., 4, 673–680 (1972).
Sulakhe, P. V., and Drummond, G. I., Archs Biochem. Biophys., 161, 448–455 (1974).
Virmaux, N., Urban, P. F., and Waenheldt, T. V., FEBS Lett., 12, 325–328 (1971).
Meyer-Schultz, F., Porte, A., Nullans, G., and Virmaux, N., Cytobiologie, 7, 393–405 (1973).
Kuhn, H., Nature, 250, 588–590 (1974).
Weller, M., and Rodnight, R., Nature, 225, 187–188 (1970).
Weller, M., and Rodnight, R., Biochem. J., 132, 483–492 (1973).
Rodnight, R., and Weller, M., in Effects of Drugs on Cellular Control Mechanism (edit. by Rabin, B. R., and Friedman, R. B.), 175–192 (Macmillan, London, 1972).
Krause, E. G., Will, H., Pelough, V., and Wollenberger, A., Acta Biol. med. Germ., 31 K, 37–43 (1973).
Aurbach, G. D., Marcus, R., Heersche, J., Marx, S., Niall, H., Tregear, C. W., Keutmann, H. T., and Potts, J. T., Ann. N.Y. Acad. Sci., 185, 386–394 (1971).
Rasmussen, H., Goodman, D. B. P., and Tenenhouse, A., Crit. Rev. Biochemistry, 1, 95–148 (CRC, Cleveland, Ohio, 1972).
Friedman, N., and Park, C. R., Proc. natn. Acad. Sci. U.S.A., 61, 504–508 (1968).
Friedman, N., Fedn Proc., 30, 261 (1971).
Entman, M. L., Levey, G. L., and Epstein, S. E., Circulation Res., 25, 429–438 (1969).
Shinebourne, E. A., and White, R. J., Cardiovasc. Res., 4, 194–200 (1970).
Schwartz, I. L., and Walter, R., Excerpta Medica Int. Congr. Ser., no. 16, 264 (1968).
Delorenzo, R., and Greengard, P., Proc. natn. Acad. Sci. U.S.A., 70, 1831–1835 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WELLER, M., VIRMAUX, N. & MANDEL, P. Role of light and rhodopsin phosphorylation in control of permeability of retinal rod outer segment disks to Ca2+. Nature 256, 68–70 (1975). https://doi.org/10.1038/256068a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/256068a0
This article is cited by
-
The role of bleaching and phosphorylation of rhodopsin in the control of Ca2+ permeability
Molecular and Cellular Biochemistry (1980)
-
Interaction of bovine rhodopsin with calcium ions
Biophysics of Structure and Mechanism (1979)
-
Phosphorylation of acetylcholine receptor by endogenous membrane protein kinase in receptor-enriched membranes of Torpedo californica
Nature (1977)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.