Abstract
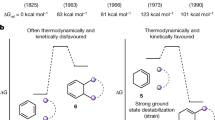
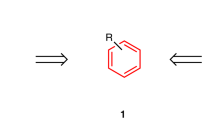
CONCERTED rearrangements of the carbonium ion kind (1 → 3) are usually supposed to go with stereochemical inversion at the migration terminus (C1) and retention in the migrating group (R) (ref. 1). Many chemists have recorded one or other of these results but no one has shown both in one and the same acyclic molecule, largely because  it is very difficult to find a reaction in which both centres are chiral and both survive as chiral in the product. Rigid cyclic systems do show both results at once but this is not very informative as no other outcome is conceivable in a molecule which has no opportunity for internal rotation.
it is very difficult to find a reaction in which both centres are chiral and both survive as chiral in the product. Rigid cyclic systems do show both results at once but this is not very informative as no other outcome is conceivable in a molecule which has no opportunity for internal rotation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Eliel, E. L., Stereochemistry of Carbon Compounds, 142–146 (McGraw Hill, New York, 1962).
Howells, D., and Warren, S., J. chem. Soc. Perkin II, 1472–1475 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ALLEN, F., KENNARD, O., NASSIMBENI, L. et al. Stereochemistry of a carbonium ion rearrangement. Nature 248, 670–671 (1974). https://doi.org/10.1038/248670b0
Received:
Issue Date:
DOI: https://doi.org/10.1038/248670b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.