Abstract
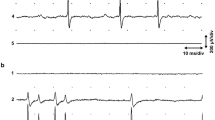
WHEN the motor axons innervating an insect muscle are severed, the peripheral axon stumps degenerate and neuromuscular synaptic transmission fails, that is, the muscle no longer responds to indirect stimulation1. At 20° C transmission at excitatory synapses on locust metathoracic extensor tibiae and retractor unguis muscle fibres fails 9–24 days after sectioning of the innervating nerve in the metathorax of this insect1. At 30° C, the muscle fails to respond to indirect stimulation after only 2 days of denervation. The isolated peripheral portions of the severed motor axons are, however, still able to conduct impulses for some time after synaptic transmission has failed1. The spontaneous miniature excitatory post-synaptic potentials (m.e.p.s.p.s)2,3 also survive for some time, thus precluding the possibility that transmission at the excitatory synapses fails initially because of depletion of transmitter from the terminals of the excitatory axons1. It seems that the properties of the postsynaptic membrane are also unaffected at this stage as witnessed by the persistence of the miniature activity1 and the continued response of the muscle fibres to L-glutamate (L-glutamate mimics the transmitter at locust excitatory neuromuscular synapses4,5). It is concluded that synaptic transmission failure results initially from a breakdown in the mechanism which couples excitation of the nerve terminals by the invading nerve impulse to the release of transmitter from the nerve endings. It is perhaps significant that after transmission at the synapses has failed the spontaneous miniature discharge is unaffected during depolarization of the nerve endings with potassium1. In normal preparations potassium depolarization causes a large increase in the frequency of the m.e.p.s.p.s3.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Usherwood, P. N. R., J. Insect Physiol., 9, 811 (1963).
Usherwood, P. N. R., Nature, 191, 814 (1961).
Usherwood, P. N. R., J. Physiol., 169, 149 (1963).
Usherwood, P. N. R., and Machili, P., Nature, 210, 634 (1966).
Usherwood, P. N. R., Amer. Zool., 7, 553 (1967).
Usherwood, P. N. R., J. Insect Physiol., 9, 247 (1963).
Liley, A. W., J. Physiol., 136, 595 (1957).
Miledi, R., J. Physiol., 151, 1 (1960).
Del Castillo, J., and Katz, B., Prog. Biophys., 6, 121 (1956).
Katz, B., Proc. Roy. Soc., B, 155, 455 (1962).
Axelsson, J., and Thesleff, S., J. Physiol., 147, 178 (1959).
Usherwood, P. N. R., and Machili, P., J. Exp. Biol. (in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
USHERWOOD, P., COCHRANE, D. & REES, D. Changes in Structural, Physiological and Pharmacological Properties of Insect Excitatory Nerve-muscle Synapses after Motor Nerve Section. Nature 218, 589–591 (1968). https://doi.org/10.1038/218589a0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1038/218589a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.